Common menu bar links
Breadcrumb Trail
ARCHIVED - Assisted Human Reproduction Canada - Report
 This page has been archived.
This page has been archived.
Archived Content
Information identified as archived on the Web is for reference, research or recordkeeping purposes. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats on the "Contact Us" page.
Minister's Message
 I am pleased to present the 2009-10 Departmental Performance Report for Assisted Human Reproduction Canada (AHRC), which presents the Agency's progress in furthering its mandate of protecting and promoting the health and safety of Canadians in relation to reproductive
technologies.
I am pleased to present the 2009-10 Departmental Performance Report for Assisted Human Reproduction Canada (AHRC), which presents the Agency's progress in furthering its mandate of protecting and promoting the health and safety of Canadians in relation to reproductive
technologies.
The issues and impacts in this area are of great importance to Canadians, and to people around the world. The Government of Canada decided, out of respect to the Court, not to pre-publish regulations while constitutional issues relating to the Assisted Human Reproduction Act are before the Supreme Court of Canada.
The Agency continues its enforcement activities of those provisions of the Act which are in force. It continues to monitor compliance with the Act and to respond to allegations of contraventions by taking appropriate action. While further regulations await release, AHRC has prepared the systems and processes required to establish a licensing framework and continues to promote the health and safety of Canadians who use reproductive technologies to build their families. AHRC continued to do extensive education and outreach as well as to build networks with stakeholders involved in assisted human reproduction, both across Canada and around the world. This outreach increases the inclusion of, and gives voice to, all Canadians with an interest in this issue.
I thank the Board of AHRC for their continued engagement in this important work.
The Honourable Leona Aglukkaq, P.C., M.P.
Minister of Health
President's Message
Continued developments in the field of assisted human reproduction (AHR) underline the importance of Assisted Human Reproduction Canada's (AHRC) role in protecting and promoting the health, safety, dignity and rights of Canadians who use, or are born of, assisted human reproduction technologies.
This Departmental Performance Report for 2009-10 demonstrates the Agency's continued diligence and commitment to these responsibilities.
Under its compliance and enforcement mandate, the Agency continued to promote compliance with the Assisted Human Reproduction Act (the "Act") and its regulations. In addition, the Agency focused on monitoring developments in the field and formalizing its process for responding to complaints brought to its attention about possible violations of the Act and regulations. Appropriate enforcement actions were taken as necessary to promote and achieve compliance. As we await the release of further regulations of the Act, the Agency continues to engage stakeholders proactively for education or information on the implications and requirements of the Act, to provide reliable information on assisted human reproduction to Canadians and to seek the perspectives of patient groups and health professionals in the field.
Under the direction of the Board of Directors, the Agency completed many key activities in support of its mandate including: the development of the 2009 Inventory of Health Surveillance Databases to assist policy makers and health practitioners to gauge the safety and effectiveness of various AHR options; the inclusion of questions related to the incidence of infertility among the Canadian population in the Statistics Canada 2009 Canadian Community Health Survey; and the sponsoring of a symposium on oncofertility in Canada to support the establishment of new networks among researchers, medical practitioners and patient advocates to encourage further research in this field. As well, the Agency gathered researchers, clinicians and infertility experts at the first Scientific Roundtable on the Prevention of Multiple Births Associated with Infertility Treatments to explore the challenges associated with multiple births related to AHR. These efforts culminated in the development of a Canadian framework. Much of this work was informed by the Agency's Science Advisory Panel as well as AHRC policy analysts and research experts.
I remain confident that the Agency will continue to build upon these achievements and strengthen its capacity as we move forward.
Dr. Elinor Wilson, President
Assisted Human Reproduction Canada
Health Portfolio Overview
The Minister of Health is responsible for maintaining and improving the health of Canadians. These efforts are supported by the Health Portfolio, which includes Health Canada , the Public Health Agency of Canada, the Canadian Institutes of Health Research, the Hazardous Materials Information Review Commission, the Patented Medicine Prices Review Board and Assisted Human Reproduction Canada. Each member of the Portfolio prepares its own Report on Plans and Priorities.
Section I: Departmental Overview
1.1 Summary Information
Raison d'�tre
Assisted Human Reproduction Canada (AHRC) was established under the authority of the Assisted Human Reproduction Act (AHR Act). The Act seeks to protect and promote the health, safety, dignity and rights of those who use assisted human reproduction (AHR) technologies; prohibits unacceptable activities such as human cloning and commercialization of human reproductive capabilities, and places controls over AHR-related research. The Agency is responsible for issuing and reviewing licences, developing and managing a health reporting information registry, establishing a health surveillance system, and carrying out inspections and compliance and enforcement activities related to activities controlled under the Act. The Agency is also a centre of expertise and a focal point of AHR information for policy makers, health professionals and all Canadians.
Responsibilities
Responsibilities
Assisted Human Reproduction Canada (AHRC) is the federal regulatory agency responsible for protecting and promoting the health, safety, dignity and rights of Canadians who use or are born of assisted human reproduction technologies.
AHRC is also responsible for fostering an environment in which ethical principles are applied in all matters relating to assisted human reproduction, while allowing scientific advances that benefit Canadians.
AHRC's mandate and responsibilities are set out in the AHR Act. The Agency's key responsibilities include:
- implementing and administering the licensing framework for controlled activities, including AHR procedures and related research;
- developing an inspection strategy to ensure compliance with the AHR Act and its regulations;
- developing and maintaining a national Personal Health Information Registry (PHIR) on AHR that can become a key component of a more comprehensive AHR health surveillance strategy;
- becoming a centre of expertise on AHR by collecting and disseminating public information;
- communicating with and engaging stakeholders on AHR issues; and
- advising the Minister of Health on AHR issues.
Strategic Outcome(s) and Program Activity Architecture (PAA)
In order to effectively pursue its mandate, the Agency aims to achieve the following strategic outcome:
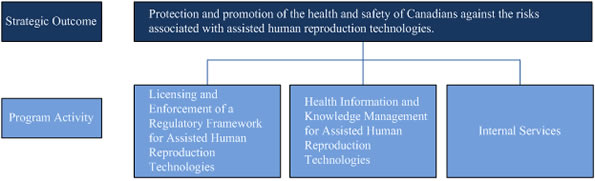
Protection and promotion of the health and safety of Canadians against the risks associated with assisted human reproduction technologies.
The chart bellow illustrates Assisted Human Reproduction Canada's complete framework of program activities, which roll up and contribute to progress toward the Agency's strategic outcome.
1.2 Summary of Performance
| Planned Spending | Total Authorities | Actual Spending |
|---|---|---|
| 10.5 | 10.5 | 5.2 |
The table above summarizes AHRC's total spending for the 2009-10 fiscal year. The Agency's actual spending was $5.3 million less than planned spending. Note that the Agency received no funding under Canada's Economic Action Plan.
| Planned | Actual | Difference |
|---|---|---|
| 44 | 13 | 31 |
Staffing was undertaken at a pace consistent with the incremental implementation of the Agency's mandate.
| Performance Indicators | Targets | 2009-10 Performance |
|---|---|---|
| As the regulations come into force, develop operational guidelines to administer the Assisted Human Reproduction Act and its associated regulations. |
In advance of the regulations coming into force:
|
Completed defining its draft Regulatory Implementation Framework and initiated drafting the guidance documentation in support of the licensing and inspection activities of the framework. Formally defined, documented and implemented the complaint management process for dealing with allegations of non-compliance with the AHR Act and its regulations. In support of its compliance and enforcement mandate the Agency also formalized its Compliance and Enforcement Policy. Stakeholder Outreach Strategy and the Information Dissemination Plan continued to evolve in the past year with a focus on establishing new networks among researchers, medical practitioners, and patient advocates as well as identifying, producing and disseminating various products. Specifically AHRC:
Brochures published and disseminated:
|
| Program Activity | 2008-09 Actual Spending |
2009-101 | Alignment to Government of Canada Outcomes | |||
|---|---|---|---|---|---|---|
| Main Estimates |
Planned Spending |
Total Authorities |
Actual Spending |
|||
| 1) Licensing and Enforcement of a Regulatory Framework for Assisted Human Reproduction Technologies | 2.8 | 4.2 | 4.2 | 4.2 | 0.8 | Healthy Canadians |
| 2) Health Information and Knowledge Management for Assisted Human Reproduction Technologies | 2.5 | 2.6 | 2.6 | 2.6 | 0.8 | Healthy Canadians |
| 3) Internal Services 2 | 3.7 | 3.7 | 3.7 | 3.6 | ||
| Total | 5.3 | 10.5 | 10.5 | 10.5 | 5.2 | |
1Commencing in the 2009-10 Estimates cycle, the resources for Program Activity: Internal Service is displayed separately from other program activities; they are no longer distributed among the remaining program activities, as was the case in previous Main Estimates. This has affected the comparability of spending and FTE information by Program Activity between fiscal years.
2Internal Services includes expenditures related to the Deputy Head's Office, Chief Science Advisor's Office and the Corporate Secretariat. In addition, it includes a Memorandum of Understanding (MOU) with Health Canada (HC) for Human Resources, Finance and IT support, a MOU with HC Inspectorate for Inspection services and a MOU with the Department of Justice for legal services.
Contribution of Priorities to Strategic Outcome(s)
| Operational Priorities | Type | Status | Links to Strategic Outcome(s) |
|---|---|---|---|
| 1. To contribute to the development of AHR regulations by Health Canada | Ongoing | (Met All) Health Canada is accountable for developing the policy and regulations under the AHR Act that AHRC will administer. The Agency actively contributes to Health Canada 's regulatory development process. | SO 1 |
| 2. To increase the awareness of Canadians and health professionals about AHR and the Agency's role | Ongoing | (Met All) Through its outreach, education and communication activities, Assisted Human Reproduction Canada (AHRC) continues to build awareness of its mandate and of understanding of the AHR Act and regulations. Through its Web site and written materials, AHRC informs Canadians about AHR. | SO 1 |
| 3. To create the capacity in AHRC to implement the regulations (once developed) | Ongoing | (Somewhat met) The Agency continues to prepare, in synchronization with the policy and regulatory development timelines of Health Canada , the systems and processes required to implement the regulations. The preparation to date will ensure that the regulations are implemented as they come into force, particularly those related to licencing, inspections, and the Personal Health Information Registry. | SO 1 |
| Management Priorities | Type | Status | Links to Strategic Outcome(s) |
|---|---|---|---|
| 1. Internal Management | Ongoing | (Mostly Met) The Agency develops and integrates its planning and reporting instruments, including Board of Directors strategic planning, integrated Agency-level business, and human resources planning and reporting. Planning and reporting is aligned with the Program Activity Architecture, with increased emphasis on performance measurement, evaluation and data collection. | SO 1 |
| 2. Human Resources | Ongoing | (Somewhat Met) AHRC continues to build its capacity in specific areas based on short-term needs, and will, as the Agency evolves and regulations come into force, need to attract additional qualified resources in specialized areas such as privacy, systems maintenance, inspection, licencing, communications, etc. | SO 1 |
| 3. Risk Management | Previously Committed to |
(Met All) The Agency built a corporate risk management framework to manage its key risks. This included the development and implementation of an ongoing process for monitoring, reporting and updating the risk management framework. This framework will also be reviewed and updated once additional regulations come into force. | SO 1 |
Risk Analysis
AHRC continues to monitor and manage its risks. Further to a systematic risk assessment, a risk management plan was developed, and risk monitoring and reporting processes were integrated into the Agency's planning cycle. The plan is reviewed on a semi-annual basis. This will serve as the Agency's corporate risk management framework, which will be updated and revised as the full regulatory framework is implemented.
Few fields are changing faster than assisted human reproduction. Each technological innovation brings additional health, ethical, social and economic implications. AHRC has established the resources and mechanisms to enable it to keep abreast of a rapidly changing environment, in terms of both science and international developments. One such mechanism is the Science Advisory Panel which brings together recognized experts in the AHR field to advise the Board of Directors and identify priorities.
AHRC continued to build upon the infrastructure (i.e., policies, procedures and systems) to effectively and efficiently manage its operations, and the specialized systems that will be required to implement the regulatory process, once the remaining regulations come into force. Work to develop the important systems required to administer the regulations and Personal Health Information Registry continued on a modular basis, using a best practices approach to ensure alignment with the regulations being developed by Health Canada. Specialized resources were retained to ensure the appropriate analysis and safeguarding of the personal health information that the Agency will eventually be collecting.
From the outset, AHRC has recognized the importance of collaborating with various stakeholder groups to deliver on its mandate. The Agency continued to conduct a comprehensive stakeholder outreach strategy that builds on its successful efforts to identify and ensure appropriate engagement of stakeholders. Fostering public and stakeholder awareness and understanding will be important for the Agency to meets its objectives.
Resource planning continues to pose a challenge for the Agency, as this planning must be aligned to the timing of the regulations coming into force and to plans to move remaining operations to the Vancouver headquarters. Key positions were staffed with indeterminate employees, and integrated business and human resource plans were reviewed and updated regularly.
Expenditure Profile
Spending Trends
AHRC spent $5.2 million to meet the expected results of its program activities and contribute to its strategic outcome in 2009 -10. The figure below illustrates the Agency's spending trend from 2006-07 to 2009-10.
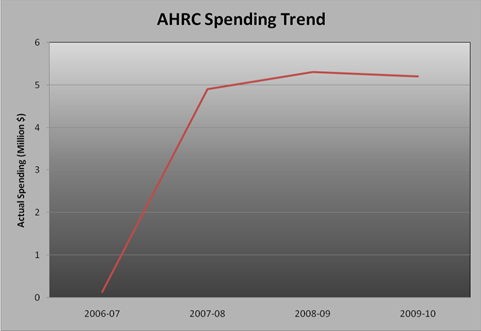
AHRC began operations in February 2007, and spending over its first few months of existence in 2006-07 totalled $134,000. In 2007-08 and 2008-09. AHRC's spending increased to $4.9 million and $5.3 million respectively as the Agency began to create the infrastructure and hire staff to fulfill its mandate. Spending remained consistent in 2009-10 at $5.2 million.
| Vote # or Statutory Item (S) | Truncated Vote or Statutory Wording | 2007-08 Actual Spending |
2008-09 Actual Spending |
2009-10 Main Estimates |
2009-10 Actual Spending |
|---|---|---|---|---|---|
| 15 | Program expenditures | 4.6 | 4.9 | 9.9 | 4.8 |
| (S) | Contributions to employee benefit plans | 0.3 | 0.4 | 0.6 | 0.4 |
| Total | 4.9 | 5.3 | 10.5 | 5.2 | |
Parliament approved resources of $10.5 million for the Agency, as per its Report on Plans and Priorities for 2009-10. The Agency's total actual spending for 2009-10 was $5.2 million.
The only statutory item for AHRC pertains to contributions to employee benefit plans in the amount of $390,600.
