Common menu bar links
Breadcrumb Trail
ARCHIVED - Patented Medicine Prices Review Board Canada
 This page has been archived.
This page has been archived.
Archived Content
Information identified as archived on the Web is for reference, research or recordkeeping purposes. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats on the "Contact Us" page.
SECTION IV - OTHER ITEMS OF INTEREST
Organizational Information
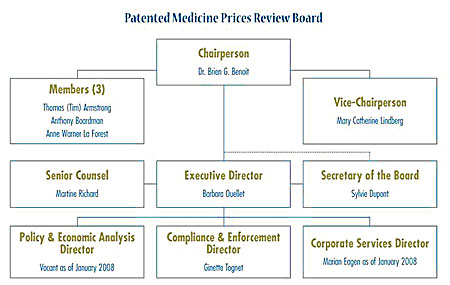
The Board consists of not more than five members who serve on a part-time basis, appointed by the Governor-in-Council, including a Chairperson and Vice-Chairperson. The Chairperson is designated under the Patent Act as the Chief Executive Officer of the PMPRB with the authority and responsibility to supervise and direct its work. The Executive Director manages the work of the staff. Senior staff consists of the Executive Director, the Director of Compliance and Enforcement, the Director of Policy and Economic Analysis, the Director of Corporate Services, the Secretary of the Board and Senior Counsel.
The Compliance and Enforcement Branch is largely responsible for the review of prices of patented medicines and the application of the Compliance and Enforcement Policy. The Policy and Economic Analysis Branch is largely responsible for undertaking policy analysis and leading the review of and consultation on the Excessive Price Guidelines, and for conducting analyses and preparing reports on price trends and other economic studies. The Secretariat supports the operations of the Board, provides overall direction to the Communications Program and coordinates the Access to Information and Privacy Programs. The Senior Counsel provides legal advice on statutory interpretation and on regulatory and policy issues. The Corporate Services Branch provides direction and administrative support with regard to information management, technology, business planning, finance and human resources.
1As a result of the June 2006 Treasury Board decision, the PMPRB received an additional $5.0M in 2007-2008, on top of its core A-base of $6.5M. Given the interim funding was sun-setting on March 31, 2008, some positions could not be staffed. As well, some hearings were settled when the patentees unexpectedly submitted Voluntary Compliance Undertakings (VCUs), while other matters that were anticipated to move to a hearing were delayed as a result of further discussions with patentees.
2 Unless otherwise indicated, reference to the number of patented drug products in this report reflects the calendar year 2007, as the price regulatory cycle for patented drug products is based on the calendar year.
3 A registration number (drug identification number) that the Health Products and Food Branch of Health Canada assigns to each prescription and non-prescription drug product marketed under the Food and Drug Regulations. The DIN is assigned using information in the following areas: manufacturer of the product; active ingredient(s); strength of active ingredient(s); pharmaceutical dosage form; brand/trade name; and route of administration.
4 The Guidelines are published in the PMPRB's Compendium of Guidelines, Policies and Procedures, which is available on the Web site: www.pmprb-cepmb.gc.ca, under Legislation, Regulations, Guidelines.
5 Details of pricing information that patentees must file are contained in Section 4 of the Regulations. The Patentees' Guide to Reporting outlines the four classes of customer: hospital, pharmacy, wholesaler and other.
6 For the purposes of the Board's price review, a new patented drug product in 2006 is defined as any patented drug product introduced in Canada, or previously marketed but first patented between December 1, 2005, and November 30, 2006. The same approach is used for all years due to the timing of the filing requirements under the Patented Medicines Regulations, 1994.
7 For the purpose of this report, existing medicines include all patented drug products that were introduced prior to December 1, 2005.
8 The full text of each VCU is available on the Web site: www.pmprb-cepmb.gc.ca, under Regulatory, Voluntary Compliance Undertakings.
9 The total number of hearings was subsequently reduced to eight as a result of the Hearing Panel's acceptance of a VCU to resolve the matter of 3M Canada and the medicine Airomir, and the matter of Janssen-Ortho and the medicine Risperdal Consta. On July 25, 2007, a further Notice of Hearing was issued in the matter of Abbott Laboratories Limited and the price of the medicine Zemplar bring the number of ongoing hearings to nine.
10 The Patented Medicines Regulations require patentees to file information according to customer classes in each province and territory; these classes, which are hospitals, pharmacies, wholesalers and other, are specified in the Patentees' Guide to Reporting developed by the Board.
11 Results for the fourth customer class, "Other", are not provided. Primarily, it is made up of healthcare institutions other than hospitals, such as clinics and nursing homes, and may include physicians, etc.
12 IMS Health's Retail Drug Monitor, 2007 (www.imshealth.com). IMS Retail Drug Monitor provides estimates of direct (i.e., from the manufacturing company) and indirect (i.e., through a wholesaler) drug purchases by pharmacies in 13 major markets: Argentina, Australia, Brazil, Canada, France, Germany, Italy, Japan, Mexico, New Zealand, Spain, the U.K. and the U.S. These figures are ex-manufacturer prices and include all prescription and certain over-the-counter data.
13 The R&D-to-sales ratios presented in Table 4 include research expenditure funded by government grants. If the government-funded component is excluded, the ratios for all patentees and for the members of Rx&D in 2007 are 8.0% and 8.6%, respectively.
