Common menu bar links
Breadcrumb Trail
ARCHIVED - Hazardous Materials Information Review Commission Canada
 This page has been archived.
This page has been archived.
Archived Content
Information identified as archived on the Web is for reference, research or recordkeeping purposes. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats on the "Contact Us" page.
Section II Analysis of Performance by Strategic Outcome
Strategic Outcome
Trade secret exemptions within WHMIS that balance the right of industry to withhold bona fide confidential business information with the right of employers and workers to be provided with complete and accurate information on the health and safety hazards posed by workplace chemicals.
Program activity: Claims Exemption Process
Financial Resources ($ thousands)
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
| 3,506 | 3,828 | 3,621 |
Human Resources (FTEs)
| Planned | Actual | Difference |
|---|---|---|
| 35 | 32 | 3 |
Under this activity, HMIRC registers claims for exemption received from a supplier or employer who wishes to withhold critical proprietary information, decides on the validity of the claim, adjudicates and issues decisions on the compliance of MSDS or label to which the claim relates, and administers an appeal process to these decisions.
Performance Analysis
Claims Exemption Process
| Expected Results | Indicators |
|---|---|
|
|
|
|
|
|
Key Program: Claims Processing
Under this sub-activity, HMIRC registers claims that enable companies to sell and/or distribute their products while the claim is being processed. Then the validity of the claim for exemption is determined, based on the Hazardous Materials Information Review Regulations criteria, and the MSDS is evaluated to ensure compliance with WHMIS requirements. Decisions are issued and published in the Canada Gazette.
The expected results for Claims Processing focus on the balance sought by the Commission – workers right to accurate information versus industry's right to protect trade secrets. Through the registration of claims, the Commission seeks to ensure a speedy process that both minimizes disruption of business and ensures the protection of confidential business information. Through the review of the claims and associated MSDSs and labels, the Commission aims to ensure a speedy, fair and impartial review of the information provided to workers on the health and safety hazards of those products associated with the claims for exemption.
| Expected Results | Performance Indicators |
|---|---|
| Manufactures can import, distribute and sell products | Number of claims registered Elapsed time between receipt of claim and registration Number of complaints from claimants about delays |
| MSDSs comply with legislation | Number of violations found on MSDSs by type Average number of violations per claims processed Extent to which claimants have complied with orders within the 75 calendar days allowed |
Claims Registration
A total of 332 claims were filed in 2007-08. Of these, 97% (322) were verified and registered within the seven-day turn around time specified in the Commission's service standard. The remaining 3% of applicants were required to provide additional information before verification and registration. The Commission has received no complaints relating to delays in registering claims.
Figure 1: Percentages of new and refiled claims registered, 2005–06 to 2007–08
Approximately 70% of the claims registered in 2007-08 (i.e. 233) were original filings and the remaining 30% (i.e. 99) were refilings, after three years, of previously approved claims, as required by law.
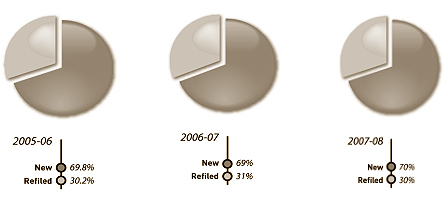
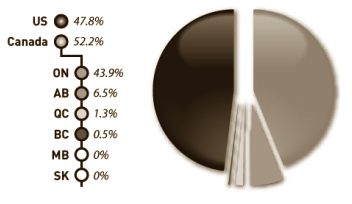
Figure 2: Origin of Claims, 2007-08
Consistent with previous years, nearly 48% of claims were from U.S. suppliers, while the remaining 52% were from Canadian suppliers - the majority of which were from Ontario a trend that has emerged over the last five years.
Claims Processing
In 2007-08, backlog reduction was a top priority for the Commission. With the efficiencies and process refinements introduced, and with a plan for an expanded evaluation team, the Commission was able to begin to make strides. During the year, a total of 376 claims for exemption were processed to the stage where the MSDS was reviewed and an advice document (AD) prepared. This represents an apparent significant increase (69%) over 2006-07 a year where there were a particularly high number of complex claims, recruitment challenges, and a limited number of trained staff.
Of the 376 claims mentioned above, 284 were processed to the point of publishing a decision and an order in the Canada Gazette. Claimants are required to comply with the Commission's statutory orders within 75 calendar days of publication. As notices of decisions and orders are published in the quarter after they were rendered, HMIRC can only report on claimant's compliance for the first three-quarters of the fiscal year. During this time period, the Commission issued 149 decisions that required orders to correct MSDSs; of these, 112 decisions were complied with within the allotted timeframe – 37 were still outstanding at year end. The Commission continues to pursue delinquent claimants until the all orders are complied with.
Once again, the Commission gave priority attention to claims for products considered to pose higher risks to workers. Of the 284 decisions in 2007-08, 55% were classified as high-hazard – those that pose a major risk to workers' health if not properly managed – were reviewed without significant delay. This practice allows corrected MSDSs for high-hazard products to reach the workplace sooner, reducing the risk of exposure or injury to workers and contributing to the health of Canadians.
MSDS Violations
An MSDS is required to be fully compliant with the Hazardous Products Act, the Canada Labour Code, the Controlled Products Regulations and the provincial and territorial occupational health and safety legislation when a claim is submitted to the Commission. To ensure this is the case, the Commission reviews the MSDSs for all claims. As in previous years, in 2007-08, only about 6% of the MSDSs (17/284) submitted were found to be compliant, and on average, there were 8 violations per claim. As shown in Table 1, on page 18, of the MSDSs that were non-compliant, approximately 58.5% of the violations were related to toxicological properties, hazardous ingredients and first aid measures. Accurate information on these three components is a significant contributor to the protection of worker health and safety. The volume of violations related to elements with potential long-term health implications–mutagenic, developmental, reproductive and carcinogenic–was low, from zero to 2.2%, although still presenting some cause for concern for workers, in particular women of childbearing age.
Table 1: MSDS violations, 2005–06 to 2007–08
| Violation Category | Number of Violations by Year | ||||
|---|---|---|---|---|---|
| 2005–06 | 2006–07 | 2007–08 | Total | % | |
| Toxicological properties | 850 | 372 | 733 | 1955 | 30.1 |
| Hazardous Ingredients | 333 | 257 | 448 | 1038 | 16.0 |
| First Aid measures | 370 | 249 | 184 | 803 | 12.4 |
| Preparation Information | 232 | 237 | 251 | 720 | 11.0 |
| Registry number/date of filing | 263 | 59 | 58 | 380 | 5.8 |
| Physical Data | 95 | 92 | 85 | 272 | 4.2 |
| Reactivity Data | 117 | 33 | 68 | 218 | 3.4 |
| Hazard Classification | 76 | 53 | 598 | 188 | 2.8 |
| Format/Wording | 57 | 82 | 104 | 243 | 3.7 |
| Fire or Explosion Hazard | 58 | 52 | 109 | 219 | 3.4 |
| Headings | 52 | 41 | 54 | 147 | 2.3 |
| Generic Chemical Identity | 43 | 53 | 106 | 202 | 3.1 |
| Product Information | 55 | 15 | 17 | 87 | 1.3 |
| Preventive Measures | 14 | 6 | 12 | 32 | 0.5 |
| Total | 2615 | 1601 | 2288 | 6504 | 100 |
| Number of claims/controlled products | 298 | 192 | 284 | 774 | |
| Average number of violation occurrences per claim | 8.7 | 8.3 | 8.0 | 8.4 | |
Backlog Reduction Plan Estimates
An ongoing challenge for the Commission has been forecasting the number of claims for exemption that chemical manufacturers, importers and suppliers will file for review and decision. Table 2 illustrates the methodology the Commission uses to estimate the number and type of claims for the next three years and the progress the Commission plans to make in reducing its claims backlog over the next three fiscal years.
The Commission established fiscal years 2005-06 and 2006-07 as a baseline for forecasting the workload for future years. Using this baseline, the Commission forecasts that it will receive 370 claims for fiscal years 2008-09 to 2010-11 – see Subtotal (1) in Table 2 on page 21.
Legislation requires that claims for exemption be refiled every three years. As in previous years, the estimated number of refiled claims for the next three fiscal years is based on a rate of 30% of the projected number of claims received in that fiscal year – Subtotal (1). The estimated number of claims that will be received in 2008-09 to 2010-11 is 111 claims – 30% of the 370 estimated annual claims received. Consequently, the number of new claims is forecasted at 259 claims (370 -111) – see New Claims and Refilings, Table 2 on page 21.
In any given year, claims are withdrawn for various reasons. For example, when a company undergoes a management change, the new management may decide to declare the ingredient it previously sought to protect; or, the company may no longer sell the product. In most cases, the Commission staff will have already spent a significant amount of time reviewing these claims before they are withdrawn.
The Commission estimates that 15% of all claims brought forward from a previous year will be withdrawn. The result is an estimated 106 claims will be withdrawn in 2008-09, 87 claims in 2009-10 and 64 claims in 2010-11.
The number of claims processed in 2008-09 is expected to increase significantly based on efficiencies and increased resources. As a result, HMIRC is forecasting it will process 500 claims in 2008-09 and 525 claims in 2009-10 and 2010-11 – see Subtotal (2), Table 2 on page 21. As shown in Table 2, it is estimated the backlog balance will be 268 claims at the end of fiscal year 2010-11; leaving a reasonable level of claims to be processed; the backlog will be virtually eliminated in 2011-12.
Table 2: Actual claims for 2006–07 to 2007–08 and Estimated claims for 2008–09 to 2010–11
| Actual number of claims | Estimated number of claims | ||||
|---|---|---|---|---|---|
| 2006–07 | 2007–08 | 2008–09 | 2009–10 | 2010–2011 | |
| Brought forward | 708 | 813 | 708 | 578 | 423 |
| PLUS | |||||
| New claims | 267 | 233 | 259 | 259 | 259 |
| Refilings | 120 | 99 | 111 | 111 | 111 |
| Subtotal (1) | 387 | 332 | 370 | 370 | 370 |
| MINUS | |||||
| Withdrawls | 90 | 153 | 106 | 87 | 64 |
| Claims processed | 192 | 284 | 394 | 483 | 461 |
| Subtotal (2) | 282 | 437 | 500 | 525 | 525 |
| EQUALS | |||||
| Backlog Balance | 813 | 708 | 578 | 423 | 268 |
* All forecasted numbers are adjusted annually to ensure that all projections remain meaningful.
Other Program: Dispute Prevention/Appeals
Under this sub-activity, HMIRC administers an appeal process. Claimants have 45 days to launch an appeal once the decision on a claim exemption is published in the Canada Gazette. An independent tripartite board is then convened to hear the appeal and render a decision. The Commission is also responsible for a dispute prevention process that works in conjunction with the appeals process by identifying and resolving problems and complaints, where possible, before an appeal becomes necessary.
Performance Measurement Strategy
The expected results for dispute prevention/appeals focus on the two desired characteristics of the Commission's dispute resolution process. First, that the handling of disputes or appeals is achieved in a fair and effective manner which will provide claimants some flexibility in the approach they choose to resolve issues. Second, that decisions resulting from the dispute resolution process be impartial and evidence-based.
The expected results of the dispute prevention/appeals sub-activity and proposed performance indicators to measure achievement of those results are detailed in table on the next page.
| Expected Results | Performance Indicators |
|---|---|
| Resolution of issues raised during the information exchange phase of claims processing (i.e., dispute prevention) | Number of issues raised and resolved |
| Appeal decisions | Number of appeals/decisions Number of screening officer's decisions varied or rescinded by appeal board's decisions |
As part of its mission, the Commission supports fair dispute resolution processes. In 2007-08, the Commission adopted a dispute prevention philosophy. Through dialogue and information sharing, HMIRC was able to clarify its information requirements to claimants, which has resulted in the exchange of information needed for screening officers to render fair and impartial decisions. The dialogue has helped prevent significant disputes from arising.
In 2007-08, claimants were contacted 285 times for clarification or to address potential issues prior to a dispute arising. Of these, only two instances resulted in a dispute; at year-end these issues were still unresolved.
For additional information on Dispute Prevention/Appeals, please refer to the HMIRC Web site: www.hmirc-ccrmd.gc.ca/english/institutional/aboutus.shtml#dispute
Departmental Priorities
An important part of the Commission's mandate is to ensure, through its scientific review process, the completeness and accuracy of the health and safety information supplied to employers and workers using hazardous products via the MSDSs supplied by suppliers claiming trade secret protection. For this reason, HMIRC's Departmental Priorities are aimed at improving the Claims Exemption Process to ensure that employers and workers in Canada have the timely information they need to use hazardous materials without risk of injury and without short or long-term health threats. The Commission's work supports improved occupational health and safety for Canadian workers, a key element to achieving a healthy Canadian population.
Improve services to clients and stakeholders
Throughout the year, Commission staff remained committed to providing service excellence to prospective claimants seeking information about the claim process and the review of MSDSs. The Commission responded promptly to queries directed to its area of expertise. The Commission was also expedient in responding to queries received from the general public and professionals alike, involving our partners in occupational health and safety when necessary.
Extensive work was completed in 2007-08 to create the conditions for eliminating the Commission's claim backlog. In addition to recruitment of new evaluators, the evaluator-training program was revamped and a career-development program was created to build capacity for future years.
Following the Operations Branch's two-day consultation workshop with its staff, HMIRC undertook a full review of its MSDS evaluator's handbook and converted it from a reference guide to a user manual–introducing step-by step instructions to help staff become familiar with the procedures and requirements associated with MSDS evaluations more efficiently. Operations managers created a three-day course to orient staff to the details of the claim assessment process, and began development of a template for advice documents that will act as a complement to the user manual.
In 2007-08, the Commission also strengthened and piloted its buddy system, pairing new staff with seasoned evaluators for one to two months of on-the-job coaching. The system is intended to significantly shorten the training period for scientific evaluators. The coaching program was very successful. Those that participated in the program were able to complete their first Advice Document (AD) within one month, as compared to the almost six months needed in the past. This program will be extended to new evaluators expected to join the Commission in 2008-09.
To improve retention, managers defined a four-staged career-progression path for toxicologists and other scientists, from entry-level evaluators to senior biologists, and made a commitment to this process.
Development of an electronic Application for a Claim for Exemption form was a pilot project undertaken in 2007-08. The form features interactive elements that simplify the application process for claimants and allows applicants to customize the forms to suit their unique requirements. Feedback was gathered through the pilot phase; refinements will be applied in the coming year.
In an effort to ensure use of the most up-to-date scientific information on hazardous ingredients, the Commission updated its scientific reference database to include new publications on 708 ingredients contained in products submitted for claim exemptions.
The Commission also upgraded its claim management and time utilization databases to include indicators for improved performance measurement. These updates will allow for easier mining of the data on the performance indicators.
As an organization with a diverse stakeholder base and a very specific mandate, HMIRC is aware of the importance of communicating its role to its occupational safety and health partners and those in related fields.
The Commission's Web site continues to be the primary outreach and communications tool for both claimants and stakeholders. In 2007-08 the Commission continued the work required in preparation for legislative changes and a comprehensive Web site review was undertaken. A virtual Web site was developed to prepare for the implementation of the new legislative amendments and general improvements were instituted to simply claimants' access to information on the site. The Commission also prepared the Web site to meet the Common Look and Feel Standards for the Internet (CLF 2.0) requirements. All changes to the CFL 2.0 Web site will be completed prior to the deadline of December 31, 2008.
As in past years, the Commission participated in several industry trade shows, including the Society of Toxicology's 47th Annual ToxExpo held in Seattle, Washington, and the Eleventh International Congress of Toxicology, celebrating its 30th anniversary in Montreal, Quebec. The ToxExpo registered 700 toxicologists and 360 exhibitors, specializing in such areas as consumer product safety, safety assessment, data management, MSDSs management and regulatory services, to name a few. HMIRC also presented its findings on the trends related to MSDS violations resulting from its review of supplier claims to the Canadian Association of Manufacturers and Exporters. Events like these are key opportunities for the Commission to educate and inform claimants and potential claimants of its mandate, role and activities.
The Commission continued to reinforce its links with several organizations that have WHMIS-related mandates, including the Canadian Centre for Occupational Health and Safety (CCOHS) and the National Office of WHMIS (NOW) of Health Canada.
Monitor and implement changes resulting from Canada's response to the Globally Harmonized System of Classification and Labelling of Chemicals
The Commission continued to monitor and participate in technical discussions on Canada's implementation of the Globally Harmonized System for the Classification and Labelling of Chemicals (GHS). This is an international initiative lead by the UN to develop a single, globally harmonized system to address classification of chemicals according to their hazards and communicate the related information through labels and safety data sheets. The objective is to enhance the protection of human health and the environment by providing an internationally comprehensible system for hazard communication. HMIRC provides unique expertise and knowledge given its position as Canada's only federal/provincial/territorial agency that manages trade secret protection within WHMIS.
The Commission is involved in several aspects of the GHS as a member of: the tripartite WHMIS Current Issues Committee, which ensures a common understanding of the GHS as it evolves and establishes a Canadian position on these developments across all WHMIS stakeholders; the technical tripartite working group associated with the Current Issues Committee, which is developing consensus approaches for the implementation of the GHS; and the Intergovernmental WHMIS Coordinating Committee, which establishes consensus among the multiple government jurisdictions responsible for WHMIS on the implications for governments arising from the GHS. As progress is made on implementing the GHS in Canada, the Commission can contribute its expertise and experience in MSDS compliance for the benefit of all WHMIS stakeholders.
The Commission has also begun to monitor changes in the trade secret protection mechanisms in other countries and to determine how related GHS provisions are being implemented. By keeping abreast of the evolution of trade secret protection mechanisms globally, the Commission will be able to share best practices from the Canadian experience. It will also be able to ensure that, consistent with the Canadian approach, international efforts to harmonize trade secret protection mechanisms maintain a balance between the protection of worker health and safety and the need for suppliers to protect their trade secrets.
Operationalize the legislative amendments to the Hazardous Materials Information Review Act
On March 29, 2007, Bill S-2 amending the Hazardous Materials Information Review Act received Royal Assent and became Chapter 7 of the Statutes of Canada 2007. These legislative amendments received the support and unanimous endorsement of the Council of Governors, which represents the workers, employers and suppliers, and the federal, provincial and territorial governments.
In summary, the implementation of these amendments will reduce the time required to review claims for exemption from disclosure of confidential information, speed up correction of the information that workers need to handle hazardous materials safely, and expedite the appeal process. For more details on effects of the legislative amendments see the Improve Services to Our Clients and Stakeholders section of the HMIRC Performance Report for the period ending March 31, 2007, on the Treasury Board Web site: http://www.tbs-sct.gc.ca/dpr-rmr/2006-2007/inst/mhi/mhi00-eng.asp
Following Royal Assent of the legislative amendments, HMIRC worked in close consultation with its Council of Governors and its stakeholders to develop regulatory amendments to the Hazardous Materials Information Review Regulations and Hazardous Materials Information Review Act Appeal Board Procedures Regulations.
The regulatory amendments will:
- permit the electronic filing of claims once the Commission has the necessary facilities for accepting the secure transmission of claims electronically;
- bring the regulations into compliance with the Modernization of Benefits and Obligations Act by removing the terms 'related persons' and 'related group' in identifying potential conflicts of interest and instead, requiring counsel and experts to satisfy appeal boards that they have no personal interest in the proceedings;
- ensure that Commission staff have access to appeal board decisions for the purposes of policy-making and training;
- contribute to streamlining, improving and expediting the appeal process; and
- update wording in the Regulations.
The regulatory amendments were published in the Canada Gazette, Part I on April 12, 2008 with a comment period of 75 days. HMIRC will consider all comments before submitting the regulatory amendments to the Treasury Board for final approval.
In preparation for the amendments, the Commission undertook certain pilot initiatives in 2007-08. A voluntary compliance program was tested, giving claimants the ability to voluntarily correct common errors and omissions in their MSDSs before formal submission to the Commission. At the time of a claim's registration, HMIRC reviews each MSDS against a list of common errors and omissions and communicates the results to the claimant; the claimant is able to then promptly make the necessary revisions and resubmit the corrected MSDS. The objectives of this initiative were to reduce the number of published orders resulting from simple errors on a MSDS, to expedite processing of the claims by the Commission, and to accelerate the availability of accurate MSDSs in the workplace. Since the inception of this initiative, its use by suppliers has been exceptional; more than two-thirds have used the program. As a result, in 2008-09, the program will be fully implemented and claimants will be strongly encouraged to participate.Enhance management excellence
As a small organization with a limited budget, the Commission is highly focused on service delivery. The Commission works collaboratively with it Health Portfolio partners, particularly Health Canada on portfolio and government issues. The Commission continues to use interdepartmental resources and expertise through Memoranda of Understanding between itself and Health Canada.
In 2007-08, the Commission undertook a thorough operational review based on Management Accountability Framework principles, and solicited input from staff on areas for improvement. The results of these efforts have been reflected in a revised organizational structure and action plans for both the Operations and Corporate Services and Adjudication Branches. With those plans in place and the renewed organizational framework, the Commission has embarked on a comprehensive optimization of operational procedures and processes.
Recruiting and retaining qualified personnel is an ongoing challenge for HMIRC, given the highly specialized nature of its scientific work and a shortage of qualified talent. After collaborating with other organizations to gather best practices, the Commission's management team created a comprehensive Human Resources (HR) strategy that defined multiple channels for recruitment. A comprehensive HR plan was developed and refined in order to address the Commission's corporate needs, so that it can continue to provide claimants and stakeholders with the best service possible.
Over the past year, the Commission produced a comprehensive business case which identified a shortfall in resources required to maintain and increase the Commission's operational and administrative capacity. Near the end of the fiscal year, the Commission secured funding for the first year of a three-year backlog elimination plan.
