Common menu bar links
Breadcrumb Trail
ARCHIVED - Health Canada
 This page has been archived.
This page has been archived.
Archived Content
Information identified as archived on the Web is for reference, research or recordkeeping purposes. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats on the "Contact Us" page.
Section II Analysis of Performance by Strategic Outcome
Health Canada's Program Activity Architecture (PAA)
This section reports on our results in detail based on our PAA, which links budgets to expenditures and, in turn, to performance.
Planned and Actual Spending by Strategic Outcome, Program Activity and Sub-Activity
(millions of dollars)
| Program Activity | Planned Spending | Authorities | Actual Spending | Program Sub-Activities |
|---|---|---|---|---|
|
Strategic Outcome #1 |
||||
| Health Policy, Planning and Information | 288.4 | 312.6 | 290.4 | |
| 158.1 | 162.9 | 147.7 | Health Care Policy | |
| 6.6 | 6.6 | 4.8 | Intergovernmental Affairs | |
| 16.6 | 19.5 | 12.9 | Strategic Health Policy | |
| 25.0 | 38.4 | 37.9 | International Affairs | |
| 5.4 | 5.7 | 5.4 | Women's Health | |
| 34.8 | 35.4 | 32.2 | Applied Research, Dissemination and Accountability | |
| 1.0 | 0.9 | 1.9 | Nursing | |
| 40.9 | 43.2 | 47.6 |
Official Language Minority Community Development |
|
| Strategic Outcome #2 Access to Safe and Effective Health Products and Food and Information for Healthy Choices |
||||
| Health Products and Food | 262.1 | 278.2 | 262.3 | |
| 125.8 | 133.6 | 125.9 | Pre-market Regulatory Evaluation and Process Improvement | |
| 13.1 | 13.9 | 13.1 | Information, Education and Outreach on Health Products, Food and Nutrition | |
| 104.9 | 111.2 | 104.9 | Monitoring Safety and Therapeutic Effectiveness and Risk Management | |
| 18.3 | 19.5 | 18.4 | Transparency, Public Accountability and Stakeholder Relationships | |
|
Strategic Outcome #3(a) |
||||
| Healthy Environments and Consumer Safety | 289.9 | 305.3 | 294.1 | |
| 29.9 | 40.5 | 40.5 | Workplace Health and Public Safety | |
| 83.5 | 86.1 | 85.0 | Safe Environments | |
| 31.3 | 33.1 | 31.1 | Product Safety | |
| 65.7 | 66.4 | 60.8 | Tobacco Control | |
| 79.5 | 79.2 | 76.7 | Drug Strategy and Controlled Substances | |
|
Strategic Outcome #3(b) |
||||
| Pest Control Product Regulation | 51.6 | 68.0 | 62.7 | |
| 25.6 | 33.6 | 27.5 | New Pest Control Product Registration and Decision making | |
| 9.8 | 12.8 | 12.1 | Registered Pest Control Product Evaluation and | |
| 7.6 | 10.0 | 11.2 | Decision making Compliance | |
| 2.6 | 3.3 | 4.1 | Pesticide Risk Reduction | |
| 6.0 | 8.3 | 7.8 | Regulatory Improvement | |
|
Strategic Outcome #4 |
||||
| First Nations and Inuit Health | 2,119.1 | 2,126.0 | 2,088.0 | |
| 292.9 | 326.2 | 290.7 | First Nations and Inuit Community Health Programs | |
| 76.5 | 71.9 | 69.6 | First Nations and Inuit Health Protection | |
| 247.0 | 270.5 | 289.0 | First Nations and Inuit Primary Health Care | |
| 966.3 | 1,018.7 | 996.4 | Non-Insured Health Benefits (NIHB) | |
| 536.4 | 438.7 | 442.3 | Governance and Infrastructure Support to First Nations and Inuit Health System | |
Note: The DPR highlights results achieved for key initiatives and PAA sub-activities outlined in the 2006-2007 Report on Plans and Priorities.
Strategic Outcome #1 - Strengthened Knowledge Base to Address Health and Health Care Priorities
Program Activity Name: Health Policy, Planning and Information
Expected Results:
- Goals and objectives identified for specific strategies and initiatives
- Knowledge development and transfer of specific health policy issues
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
| 288.4 | 312.6 | 290.4 |
Human Resources (FTEs):
| Planned | Actual | Difference |
|---|---|---|
| 627 | 606 | 21 |
The objective of this program activity is to provide policy advice and support to the Minister in making decisions to protect and improve the health of Canadians. Health Canada develops policies and builds and maintains linkages with provinces, territories, and other partners and stakeholders to support health care system reform. We administer the Canada Health Act and facilitate access to health care services for official language minority communities. We work with international organizations, and bilaterally with key countries, to advance a global health agenda. Grants and contributions accounted for approximately 68 percent of spending under this program activity, demonstrating our commitment to achieving results in collaboration with partners in the health system.
Grants Explanation of the above financial information:
Variances between planned spending versus total authorities are mainly due to:
- funding for Federal Contaminated Sites Action Plan
- funding for the Canadian Partnership against Cancer
- contributions to the Government-Wide 2006-2007 $1 Billion Spending Restraint Exercise
- funding from Management Reserve - Litigation Management.
Actual spending is $22.2 million lower than total authorities mainly due to:
- lapse in the Health Council special purpose allotment
- contributions to the Government-Wide 2006-2007 $1 Billion Spending Restraint Exercise
- year end adjustments of Department of Justice expenditures
- other operating lapses in various programs.
In the Report on Plans and Priorities (RPP), we identified eight priorities under this program activity, as well as two areas addressing the role of science and horizontal linkages.
Partner in Health Reform
Canadians expect to have accessible, high quality health care services throughout their lives. To this end, the Department supported development of Patient Wait Times Guarantees (PWTGs), the Health Council of Canada and the Primary Health Care Transition Fund (PHCTF). The Department also continued to support initiatives in health human resources, home care, palliative care and access to health care for people living in official language minority communities.
One of the Government's five priorities was to negotiate PWTGs with provincial and territorial governments. All provinces and territories have now committed to establishing a PWTG by 2010 in one of the following priority clinical areas: cancer radiation, cataract surgery, hip and knee replacement, cardiac care, diagnostic imaging or primary health care. In addition, each province and territory has committed to undertaking at least one PWTG pilot project to test guarantees and inform their implementation. The Government of Quebec passed legislation to establish a framework to implement a guarantee of access to hip and knee replacement and cataract surgery.
Our Department also developed four PWTG pilot projects to help advance best practices and the reduction and better management of wait times. Two projects focus on diabetes and prenatal care in selected First Nations communities and are administered by Health Canada's First Nations and Inuit Health Branch. A third project addresses wait times for diabetic foot ulcer care for First Nations communities in Manitoba and is being administered by Saint Elizabeth Health Care. A fourth project in national paediatric surgical wait times is led by the Paediatric Surgical Chiefs of Canada and the Hospital for Sick Children in Toronto.
Health Canada also provided funding to the Canadian Institutes of Health Research to support research regarding the relationship between wait times and health and improving access to appropriate health services. In February 2007, the Minister of Health and his Saskatchewan counterpart co-hosted a Conference on Timely Access to Health Care that showcased provincial successes and innovations. Further, Health Canada released the report, Healthy Canadians: A Federal Report on Comparable Health Indicators 2006, which provides a snapshot of the health status of Canadians and the performance of the health care system. The report also responds to First Ministers' commitments to greater transparency and accountability in the health care system.
Following the 2004 First Ministers' agreement to provide first-dollar coverage for certain home care services, we provided policy advice within the Department and to federal partners concerning access by First Nations, Inuit and veterans to home care services at the expected levels. In March 2007, we hosted a forum with provincial and territorial stakeholders to look at opportunities for integration between home and primary health care. As well, our Department is exploring ways to work with the Canadian Home Care Association to advance home care integration models across Canada.
Health Canada continued to support the work of the Health Council of Canada, which has a mandate to monitor and report on implementation of the 2003 and 2004 Health Accords and to report annually on health status and health outcomes of Canadians.
In response to a First Ministers' 2000 commitment, the PHCTF was launched. Between 2001 and 2006, this $800 million federal investment supported the efforts of provinces, territories and stakeholders to reform the primary health care system. With the end of the PHCTF, our focus turned to applying lessons learned across Fund projects to support ongoing primary health care reform activities.
Key PHCTF dissemination activities included synthesis papers highlighting results on chronic disease management and collaborative care; a national conference in February 2007; a Best Practices Network event on responding to community needs; and fact sheets and a database on the results of each PHCTF initiative.
We supported programs and services to improve access to health care for people in official language minority communities, consistent with the Official Languages Act. Two contribution agreements were launched to provide primary health care services within English and French-speaking minority communities.
Health Canada hosted a national forum on Palliative and End-of-Life Care in March 2007. The purpose was to support exchange of best practices across Canada, as well as to celebrate five stakeholder groups for palliative and end-of-life care initiatives supported by Health Canada from 2002 to 2007. We also provided funding for development of a web-based research centre at the Canadian Virtual Hospice. This will ensure that the growing community of palliative care researchers have the necessary tools to provide the scientific basis for improving care.
- Conference on Timely Access to Health Care
- Healthy Canadians: A Federal Report on Comparable Health Indicators 2006
- Primary Health Care Transition Fund
Hepatitis C
The pre-1986/post-1990 Hepatitis C Settlement Agreement was completed in December 2006. Preparations were initiated for the compensation process, subject to a decision expected in 2007 by the courts that have been involved in this process. This should lead to the first payments beginning in 2007-2008.
Pandemic Influenza
Our Department played many roles to ensure Canada's readiness to deal with the potential emergence of pandemic influenza. These included discussions concerning innovative approaches and incentive mechanisms to stimulate research, development, and equitable global access to pandemic influenza vaccines, as well as pneumococcal vaccines. Internationally, as Chair of the Asia-Pacific Economic Cooperation (APEC) Health Task Force, Health Canada was playing a leadership role in coordinating regional response and planning for pandemic influenza and other emerging infectious diseases. We facilitated development of the APEC Action Plan on the Prevention and Response to Avian and Influenza Pandemics and followed up in 2007 with the first regional report on implementation of the plan.
Mental Health
In the RPP, we committed to work with partners to build the foundation of a national approach to mental health and mental illness. This led to the announcement of the Mental Health Commission of Canada in Budget 2007. The Commission will undertake activities in three areas: knowledge exchange, anti-stigma efforts, and a National Strategy on Mental Health and Mental Illness.
Pharmaceuticals Management
With respect to pharmaceuticals, Health Canada has a role in market approval, access, optimal prescribing and utilization, drug prices/expenditures and system cost, as well as the safety and effectiveness of drugs once on the market.
The Department's Blueprint for Renewal initiative is focused on modernizing the regulatory system for therapeutic products.
In collaboration with other departments, including Foreign Affairs and International Trade, Health Canada plays an important role in monitoring and research activities, policy development, and provision of integrated advice (reflecting health sector interests) on pharmaceuticals-related components of: international trade negotiations and treaties; transnational issues and files such as cross-border drug sales; patent policy; and cooperative research and knowledge exchange activities bilaterally and in international fora such as the Organization for Economic Cooperation and Development (OECD) and the World Health Organization (WHO).
In 2006, Health Ministers provided First Ministers with a National Pharmaceuticals Strategy Progress Report. The report highlighted achievements and set out next steps, with a focus on five priority areas: catastrophic drug coverage; expensive drugs for rare diseases; common national drug formulary; drug pricing and purchasing strategies; and real-world drug safety and effectiveness.
Health Canada is working to ensure that current work on the NPS complements pre-existing initiatives such as the Common Drug Review (CDR)7.
7The CDR is a single process for reviewing new drugs and providing listing recommendations to participating publicly-funded federal, provincial and territorial drug benefit plans. All jurisdictions are participating except Quebec. For more information, see http://www.cadth.ca/index.php/en/cdr and http://www.hc-sc.gc.ca/hcs-sss/pharma/mgmt-gest/cdr-emuc/index_e.html. Other joint initiatives that emerged prior to the NPS and which continue to progress include the Canadian Optimal Medication Prescribing and Utilization Service (COMPUS) and the National Prescription Drug Utilization System (NPDUIS).
Legislative Renewal and Regulatory Reform
Work continued to update the Food and Drugs Act, the Hazardous Products Act and the Radiation Emitting Devices Act. We focused on policy development for the collection, use and disclosure of health information and improved compliance and enforcement.
Health Canada improved its regulatory processes in response to the Smart Regulation Initiative and other government-wide policies. Triage and prioritization models were developed to ensure greater efficiencies while improving implementation of cost-benefit analysis, instrument choice and performance measurement.
Establishment of the Assisted Human Reproduction Agency of Canada and new regulations
The Assisted Human Reproduction Act (AHRA) seeks to protect and promote the health, safety, human dignity and rights of Canadians who are born of the use of reproductive technologies and to foster the application of ethical principles in relation to assisted human reproduction.
The Agency created under the Act is the Assisted Human Reproduction Agency of Canada. The Government announced the appointment of a Chairperson, President and eight additional members to its Board of Directors in December 2006. Health Canada continued research and consultations to develop the necessary regulatory framework for the AHRA. We reviewed feedback on section 8 (relating to consent) draft regulations which were subsequently published in Canada Gazette, Part II in June 2007 and will take effect on December 1, 2007. Health Canada continues to work on remaining regulations required by the AHRA.
Assisted Human Reproduction Agency of Canada
Health Human Resources
At the heart of any health care system are the people who deliver care – health human resources (HHR). Governments at the federal and provincial/territorial levels have recognized that it is critical to ensure the adequate supply, distribution and utilization of health human resources. In response to the 2003 and 2004 First Ministers' agreements on health, Health Canada worked to implement its responsibilities under the Pan-Canadian HHR Strategy which is being renewed for 2008-2013. The Strategy includes several initiatives: Pan-Canadian Health Human Resource Planning; Interprofessional Education for Collaborative Patient-Centred Practice; and Recruitment and Retention. In 2005, the Internationally Educated Health Professionals Initiative (IEHPI) was established to increase the supply of priority health care providers through assessment and integration of internationally educated health professionals. The IEHPI is a five-year $75 million initiative that is also part of the Pan-Canadian HHR Strategy. Multi-year contribution agreements are in place with most of the provinces and territories as well as innovative pan-Canadian projects covering seven health professions: medicine, nursing, pharmacy, occupational therapy, physiotherapy, medical laboratory technology and medical radiation technology.
As a result of its participation in the Federal Council's Ontario Information Technology Network, the Ontario Region was able to sign agreements with other federal departments to share infrastructure resources and to arrange interchange opportunities and assignments for staff. An interchange arrangement was helpful in quickly filling a vacancy in the Thunder Bay office.
We provided funding to key stakeholder-driven initiatives. For example, in response to the shortage of family physicians, the College of Family Physicians of Canada created Family Medicine Interest Groups that are using different tools to encourage more medical students to choose family medicine as a postgraduate specialty. We also supported the 2007 international conference of leaders in HHR research and policy to examine the medical work force as it concerns the evolution of health care delivery systems.
An innovative interprofessional program aimed at orienting IEHPs to the Canadian health care system was developed arrangement with representatives from provinces and territories as well as six health professions. A multi-media faculty development program for teachers of international medical graduates was developed and will be fully implemented in 2007-2008. Similarly, a curriculum for faculty members training internationally educated nurses was created and will be piloted in 2007-2008. Supported by Health Canada, Ontario's Access Centre for IEHPs was formally launched in December 2006, and has been providing IEHPs with referral, counselling and bridging services. As of April 2007, the Centre has supported close to 400 IEHPs to integrate into the health care system.Under Interprofessional Education for Collaborative Patient-Centred Practice, nine new learning projects were funded for $6.7 million, bringing the total to 20 projects and $20 million. The Canadian Interprofessional Health Collaborative received $775,000 to identify and share best practices in interprofessional education and collaborative practice, and to translate this knowledge so that it can be used to transform health care.
Health Human Resources Strategy
In addition to these priorities, we addressed two areas of importance across this program activity.
Role of Science
Innovative health sciences and technologies offer potential new ways to prevent, diagnose and treat thousands of conditions affecting Canadians. These include genetic technologies, stem cell research and nanotechnologies. They also present certain challenges, such as ensuring that intellectual property and patent rights concerning genetic inventions are compatible with appropriate patient access and that Canadians receive high quality genetic services.
In consultation with many partners in Canada, our Department worked closely with the OECD to develop OECD Guidelines on Quality Assurance in Molecular Genetic Testing that are to be published in 2007. The Guidelines are an important tool to promote safe, effective and appropriate use of genetic testing in Canada. During the electronic public consultation period on the Guidelines, Canada was recognized as having the highest stakeholder participation rate of any OECD member state.
Health Canada has played a key role in the Budget 2007 commitment to invest $30 million in the Rick Hansen Foundation's Spinal Cord Injury Translational Research Network. The goal is to accelerate the translation of research discoveries into practical benefits for Canadians with spinal cord injuries while generating savings in health and social services and accelerating scientific advances towards a cure.
Health Canada conducted extensive policy research on intellectual property and patents in medical genetics and stem cells. Through workshops and symposia, we have encouraged stakeholders to adopt OECD Licensing Guidelines for Genetic Inventions. These and other creative licensing strategies will help overcome patent-related barriers to research and improve patient access to innovative biotechnology products in a cost-effective manner.
Horizontal Linkages
Research activities focused on issues such as First Nations and Inuit health sustainability, public system health care comparative analysis, health innovation and healthy communities. We continued our examination of factors influencing sustainability of the health care system including the rapidly growing pharmaceutical sector and productivity in the health sector.
Consistent with a government-wide commitment, we addressed gender and diversity issues in areas such as mental health, cancer and clinical trials. We successfully negotiated for inclusion of health, gender and diversity considerations in the deliberations of the Working Group on Trafficking in Persons.
Health Care Policy
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
158.1
|
162.9
|
147.7
|
Health Canada provides policy leadership and advice on issues related to the health care system and its renewal, such as improving access to quality care, increasing the supply and improving the mix of health care professionals, and enhancing the accountability of the system to Canadians.
Policy leadership is also provided in health care delivery, particularly home care, continuing care, palliative care and primary health care, and also to issues such as the responsiveness of the health care system to aging, chronic disease management and e-health. Programs promote and facilitate effective and innovative planning, education, training, management, recruitment and retention of health human resources in Canada.
In addition to initiatives described previously under this strategic outcome, the Department provided policy leadership and coordination on other issues that are being addressed across the Health Portfolio. For example, we worked with many partners to identify how the Government of Canada could best address the needs for a coordinated approach to cancer. This supported the Government decision to create the Canadian Partnership Against Cancer (CPCC), a not-for-profit corporation. CPCC was established to implement the Canadian Strategy for Cancer Control, a five-year plan developed by more than 700 cancer survivors and experts. The CPCC has begun to take shape to serve as a clearing house for state-of-the-art information about preventing, diagnosing, and treating cancer. Its work is supported by the $260 million announced in the 2006 budget.
We also were assigned the responsibility of leading federal policy development in relation to autism spectrum disorders. This including guiding and supporting actions designed to expand understanding of autism among researchers and health professionals and to provide more information to families and others who deal with autism. Our efforts included close cooperation with partners such as the Public Health Agency of Canada and the Canadian Institutes of Health Research.
Intergovernmental
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
6.6
|
6.6
|
4.8
|
We continued to administer the Canada Health Act (CHA), which included investigations into potential cases of non-compliance and analysis of relevant emerging issues, such as patient charges for primary care in private facilities, possible extra-billing by physicians and charges for surgical services by private clinics. We continued to see the traditionally high level of provincial and territorial compliance, which we detail to Parliament and Canadians in the Canada Health Act Annual Report.
We provided strategic and tactical advice and support on the full range of intergovernmental health-related issues and activities, with emphasis on pandemic preparedness and Patient Wait Times Guarantees.
International
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
25.0
|
38.4
|
37.9
|
Health Canada continues to initiate, coordinate, and monitor departmental policies, strategies and activities that help promote Canadian priorities and values internationally. We collaborated with external health partners such as the WHO and the Pan American Health Organization (PAHO) on pandemic influenza preparedness, HIV/AIDS and global health security.
Health Canada coordinates the global engagement component of the Federal Initiative to Address HIV/AIDS in Canada, to ensure policy coherence of Canada's international HIV/AIDS activities. Notable achievements included a strong, coordinated and effective presence at the XVI International AIDS Conference in Toronto, August 13-18, 2006. Health Canada and the Public Health Agency of Canada signed a formal partnership agreement in August 2006 with the Joint United Nations Programme on HIV/AIDS (UNAIDS) to promote joint actions aimed at strengthening global response to the HIV and AIDS epidemic.
On August 15, 2006 in Toronto, the Health Ministers for Canada and France signed a Joint Declaration of Intent Regarding Cooperation Between the Department of Health of Canada and the Ministry of Health and Solidarity of the French Republic in the Field of Health for the Period 2006-2010. The Declaration is a framework arrangement that outlines mutual work the two countries plan to undertake in the next four years. Key areas are pandemic influenza preparedness, the strengthening of health care systems, HIV/AIDS, sexually transmitted infections, hepatitis B/C, tuberculosis and sexual and reproductive health. Other potential areas of cooperation could be on physical activity, cancer and mental health.
Assisted Human Reproduction Implementation Office
Health Canada provides policy analysis and advice towards the establishment of the Assisted HumanReproduction Agency of Canada as well as developing the regulatory framework required by the AHR Act.
Legislative Renewal
Legislative renewal activities concentrate on updating and strengthening health protection legislation to ensure that it is responsive to present and future social and technological realities.
Applied Research, Dissemination and Accountability
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
34.8
|
35.4
|
32.2
|
Health Canada helps to build the analytical foundation for health policy decision making, performance measurement and reporting. This includes conducting economic analysis of health policy issues, funding external policy research in priority areas, and running a policy research publications program, which includes publication of the Health Policy Research Bulletin. We develop, in collaboration with partners and stakeholders, federal policy on investments in Canada's health statistics system and coordinate departmental core data requirements with data providers.
Official Language Community Development
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
40.9
|
43.2
|
47.6
|
We provided leadership in responding to the health needs of official language minority communities by administering Health Canada's responsibilities under Section 41 of the Official Languages Act.
We supported programs and services to improve access to health care for people in official language minority communities, consistent with the Official Languages Act. Two contribution agreements were launched to provide primary health care services within English and French-speaking minority communities. A report analyzing the evolving health access concerns of Francophone minority communities was completed and presented to the Minister in February 2007 by the Consultative Committee for French-Speaking Minority Communities. Working with Statistics Canada and several other federal departments, we launched the 2006 Survey on the Vitality of Official Language Minorities to improve the Government's ability to measure their health-related challenges. The results of this Survey will be available from Statistics Canada in 2007-2008. We partnered with the Department of Canadian Heritage in launching a learning tool for development of status reports and action plans required under Part VII of the Official Languages Act and its accountability frameworks.
Strategic Outcome #2: Access to Safe and Effective Health Products and Food and Information for Healthy Choices
Program Activity Name: Health Products and Food
Expected Results:
- Access to safe and effective health products and food and information for healthy choices
| Performance Indicators | Results |
|---|---|
| Level of satisfaction of Canadians and health professionals with the information disseminated for healthy choices and informed decision making | This indicator is being revised. No current data exists. |
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
262.1
|
278.2
|
262.3
|
Human Resources (FTEs):
2,592| Planned | Actual | Difference |
|---|---|---|
|
2,592
|
2,563
|
29
|
Explanation of the above financial information:
- Variances between planned spending versus authorities are mainly due to:
- Funding from Management Reserve - Natural Health Products
- Funding from Management Reserve - Litigation Management
- Funding for Therapeutics Access Strategy.
The actual spending is $15.9 million lower than authorities mainly due to:
- Lapse of frozen allotment for Access to Medicines Regime
- Year end adjustments of Department of Justice expenditures
- Other operating lapses in various programs.
Health Products and Food is a regulatory program. Its objective is to evaluate and monitor the safety, quality and effectiveness of the thousands of drugs, vaccines, medical devices, natural health products and other therapeutic products available to Canadians, and nutritional quality of their food. We also review veterinary drugs sold in Canada as well as foods derived from animals treated with these drugs. We promote the health and well-being of Canadians by developing nutritional policies and standards such as Canada's Food Guide and providing information to the public in newsletters such as It's Your Health. The Government is committed to continually modernizing its legislative and regulatory frameworks to keep pace with advancing science and technology, existing and emerging public health challenges, consumer expectations in terms of safety, the need for transparency, faster drug approvals, international developments and other factors. Since 1953, responsibilities for health products and food safety have been primarily defined through the Food and Drugs Act.
Health Canada has identified challenges that must be met to ensure continued, timely access to safe and effective health products and a safe and nutritious food supply. These include but are not limited to: an outdated regulatory tool kit that is increasingly limited and inflexible in responding to today's environment; the regulatory system's incapacity to consider a product through its entire life cycle; and a regulatory system with insufficient resources for long-term efficiency and sustainability. The Office of the Auditor General came to similar conclusions when it reviewed this area for its November 2006 report. 8
Consequently, in October 2006, we released Blueprint for Renewal: Transforming Canada's Approach to Regulating Health Products and Food for consultation. This policy review document builds on progress made over the last few years to improve the regulatory system's efficiency, safety and transparency. The Blueprint reflects input from Canadians and commits us to improve information for decision making. It is supported by detailed action plans, setting the stage for consultations on and implementation of specific Blueprint initiatives in 2007-2008 and 2008-2009, including a new progressive licensing framework for pharmaceuticals and biologics. We will continue to report progress on these and longer-term initiatives through the Blueprint web-site and in reporting frameworks such as the DPR.
The Department addressed other areas that complement these directions as well as issues raised by the Auditor General. For example, we began work on a cost recovery framework and a comprehensive review of programs and resources. The cost recovery regime should be implemented in 2008-2009, updating a 10-year-old fee regime. This will support a long-term, stable and sustainable funding strategy for our regulatory programs. The assessment of all programs and activities will define the level of activities, performance and resources required to meet our regulatory and other responsibilities, based on the full cost of these activities.
Under this Health Products and Food program activity, are four sub-activities as defined in our Program Activity Architecture (PAA). Achievements under each of the sub-activities are outlined below.
8 OAG report: http://www.oagvg.gc.ca/domino/reports.nsf/html/20061108ce.html
Pre-market Regulatory Evaluation and Process Improvement
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
125.8
|
133.6
|
125.9
|
Improved timeliness, transparency and predictability of the regulatory process Improved timeliness, transparency
and predictability of the regulatory
process
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Improved timeliness, transparency and predictability of the regulatory process |
Percentage of overall workload in backlog and percentage of decisions issued within target for submission reviews of:
|
|
We continued to implement a long-term government commitment to improve the timeliness of the regulatory process for therapeutic products to ensure that Canadians have faster access to the safe drugs they need. This had many elements.
Improved Speed of Decision Making
Using funding received in Budget 2003 under the Therapeutics Access Strategy, a five-year, $190 million initiative, we improved our speed of decision making on new product reviews. This is demonstrated in the table above. In addition, we cleared the backlog of new biologic submissions by September 2006.
We made similar progress for medical device applications, which are assigned to classes based on their potential risk to humans. For example, bandages are Class I (low risk) devices, while pacemakers and HIV test kits are Class IV (highest risk). 11 Our processing of Class II applications was much better than the 51 percent performance in 2004. In the area of natural health products, we are making six times as many daily decisions as we did a year ago.
We continued to reduce decision times for veterinary drugs and set service standards. We reviewed 93 percent of data packages submitted prior to October 1, 2005, exceeding our target of 90 percent. We also completed several submission reviews that were not on our target list.
The ongoing challenge continues to be significant backlogs in our pre-market review of submissions for natural health products, veterinary drugs and food products. We have reduced backlogs at some stages of our work and are pursuing measures to process these applications more quickly and consistently in comparison with international performance targets.
The Blueprint for Renewal
Under the Blueprint described earlier, a progressive licensing framework for pharmaceuticals and biologics will facilitate access to drugs while continuing to monitor safety, efficacy, and quality throughout a drug's life cycle. The project achieved its early-stage objective and benefited from outreach to patient and consumer groups, industry, academia, health care professionals and provincial representatives. We expect to formulate a more detailed framework during 2007. 12
In 2001, Health Canada established a framework and regulations to oversee clinical trials that support applications for new drugs. At that time, we committed to assess the impact of the regulations and seek advice on improvements in three to five years. As part of the Blueprint initiative, we began a public e-consultation in June 2006. This led to a March 2007 workshop with stakeholders on specific possible improvements.
In addition to work under the Blueprint, we began the review of regulations and processes related to pre-market safety assessment and authorization of foods and food products. We developed a draft Guide for the Preparation of Submissions on Food Additives and an options paper for a modernized regulatory framework for food additives that is expected to be released soon for external consultation.
11 Classes of medical devices:
Class Class I| Class | Risk | Example |
|---|---|---|
| Class I | Lowest risk | Reusable surgical scalpel, bandages, culture media |
| Class II | Low risk | Contact lenses, epidural catheters, pregnancy test kits, surgical gloves |
| Class III | Moderate risk | Orthopedic implants, glucose monitors, dental implants, haemodialysis systems, diagnostic ultrasound systems |
| Class IV | High risk | HIV test kits, pacemakers, angioplasty catheters |
12 http://www.hc-sc.gc.ca/ahc-asc/branch-dirgen/hpfb-dgpsa/blueprint-plan/index_e.html
http://www.hc-sc.gc.ca/dhp-mps/homologation-licensing/develop/plan_e.html
Clinical Trials
In 2005, Health Canada received $170 million over five years to improve the safety of drugs, medical devices and other therapeutic products. As part of this initiative, we are strengthening the oversight of pharmaceutical clinical trials and investigational testing for medical devices conducted in Canada. New funding for 20062007 enabled us to add staff to handle significant increases in drug clinical trial applications, clinical trial Adverse Reaction Reports and safety reports. This has resulted in more timely responses and appropriate linkages with our pre-market review and post-market surveillance activities, although we recognize gaps that we must still address.
In addition, we increased efforts to make our internal processes as consistent and transparent as possible. As a result of posting the Clinical Trials e-manual on our website in 2006 we had fewer calls from clinical trial sponsors for information. We also discovered fewer deficiencies while screening clinical trials and investigational testing applications from clinical trial sponsors.
Regulatory Actions
We continued to develop regulatory frameworks. One seeks to minimize the potential health risks to Canadian recipients of human cells, tissues and organs through proposals for Safety of Human Cells, Tissues and Organs for Transplantation Regulations. Extensive consultations will continue in 2007-2008. We are at an earlier stage of consultations to guide a renewed regulatory framework for whole blood and blood components, typically used in transfusions, under the Food and Drugs Act.
Health Canada continued to lead a federal stewardship approach to manage risks and benefits of emerging biotechnologies and nanotechnologies products and services. This included work on horizontal policies, identification of barriers and implementation of strategies for supportive environments. An interdepartmental Biotechnology Regulatory ADM Steering Committee considered common biotechnology regulatory challenges among departments.
We released revised guidelines on the safety assessment of novel foods to help improve the transparency of this regulatory process. We held training sessions and a pilot workshop with academia, government and industry to further a tiered, risk-based approach.
As promised in the RPP, we began to develop a new regulatory approach for radiopharmaceuticals used for diagnosis and radiation therapy. We are moving forward on two regulatory initiatives. The first will address basic clinical research involving radiopharmaceuticals or positron-emitting radiopharmaceuticals. The second will require drug identification numbers (DINs) on radiopharmaceuticals.
The Department also started work, in collaboration with Agriculture and Agri-Food Canada, on a new regulatory framework for health claims for foods, including the use of logos and symbols. This will modernize the current system and support informed consumer choice with appropriately substantiated claims.
The Community of Federal Regulators is a partnership of all federal departments and agencies with regulatory roles. In order to learn from each other and accelerate innovations consistent with the Cabinet Directive on Streamlining Regulation, 12 departments and agencies signed a Memorandum of Understanding, approved a business plan, developed a learning strategy and exchanged information and best practices.
Information, Education and Outreach on Health Products, Food and Nutrition
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
13.1
|
13.9
|
13.1
|
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Improved adoption in making safe and healthy choices for health products, food and nutrition | Percentage of target populations using information in their decision making |
Health Canada continued to provide useful information about risks and benefits related to health products and food. For example, we conducted an educational campaign to increase consumer awareness of food safety related to meat and poultry. We completed a consultation on labeling information to reduce risks from unpasteurized juice and cider. Given concerns about health issues related to sprouted seeds and beans, we developed a policy and educational material including a page on our website 15 . A review of the risks of mercury to human health was conducted and standards and consumption advice updated on limiting exposure to mercury from certain predatory fish. We also completed a survey of benzene levels in non-alcoholic beverages and followed up with industry to ensure that levels found in beverages aimed at children were reduced through product reformulation. We released over 100 health advisories for health professionals and consumers and also developed a strategy for consumer information.
The Department provided ongoing objective information on emerging technologies and their applications. This included a list of potential nanotechnology-based products and/or delivery systems that fall under our authority. We also developed and implemented a pilot project - a high school Biotechnology Teachers' Kit to introduce students to biotechnology, to stimulate discussion on benefits and risks associated with biotechnology products and to learn how these products are regulated by Health Canada.
In February 2007, we launched the revised Canada's Food Guide after extensive consultations with stakeholders and the public. We disseminated 6.1 million copies and 300,000 resource guides for educators and communicators. Other resources include a web component with interactive material. From the launch of the Guide until the end of March, we recorded 6,768,674 visits to the Guide website and demand for copies has exceeded supply.
We began work with the Public Health Agency of Canada (PHAC) and the World Health Organization (WHO) on an international framework to promote and support healthy eating and physical activity through schools. This framework is to be one of the tools provided by WHO to implement the global strategy on diet, physical activity and health. In partnership with the U.S. Institute of Medicine, Health Canada contributed to developing and disseminating Dietary Reference Intakes: The Essential Guide to Nutrient Requirements, a summary report on Dietary Reference Intakes (DRIs), available in both official languages. The DRIs underpin all nutrition programs and policies in Canada and are used in risk assessment, standard-setting and policy development. A web-based Interactive Nutrition Label and Quiz helps Canadians make informed food choices and draws continued attention to nutrition labeling.
Health Canada continued to distribute the Canadian Adverse Reaction Newsletter and publish it in the Canadian Medical Association Journal. There was also a 19 percent increase in MedEffect Canada e-Notice subscribers.
15 http://www.hc-sc.gc.ca/iyh-vsv/food-aliment/sprouts-germes_e.html
http://www.hc-sc.gc.ca/fn-an/securit/facts-faits/rawmilk-laitcru_e.html
http://www.hc-sc.gc.ca/iyh-vsv/food-aliment/juice-jus_e.html
http://www.hc-sc.gc.ca/fn-an/label-etiquet/meat-viande/index_e.html
http://www.hc-sc.gc.ca/fn-an/label-etiquet/allergen/index_e.html
http://www.hc-sc.gc.ca/fn-an/label-etiquet/nutrition/index_e.html
Monitoring safety and therapeutic effectiveness and risk management
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
104.9
|
111.2
|
104.9
|
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Strengthened vigilance over safety and therapeutic effectiveness for health products and food on the market | Overall rating of Health Canada's post-market safety and therapeutic effectiveness activities | We are working on developing a new indicator |
We followed through on our RPP commitments under this sub-activity. For example, we carried out nutrition-related surveillance activities such as posting a guide to accessing and interpreting Canadian Community Health Survey data on our website, as well as printing and distributing the guide to stakeholders.
The Department received $190 million and $170 million over five years through the 2003 and 2005 budgets, for the Therapeutics Access Strategy and the Therapeutic Product Safety Initiative, respectively. These funds have enabled us to enhance post-market surveillance of drugs and other therapeutic products. For example, the Therapeutics Access Strategy has facilitated the reduction in backlog reports while the Therapeutic Product Safety Initiative has increased our scientific/medical capacity to monitor and evaluate safety information and act decisively to protect Canadians.
In 2006, we awarded a contract to develop a more sophisticated Adverse Reaction Reporting System that will collect and analyze adverse reaction information more effectively. In 2007-2008, the system will focus on post-market adverse reactions to pharmaceuticals, biologics and natural health products. It will later be expanded to include pre-market Adverse Reaction Reports from clinical trials.
We consulted on possible Environmental Assessment Regulations for substances regulated under the Food and Drugs Act to minimize the effects of therapeutic products on the environment, beginning with pharmaceuticals, veterinary drugs, radiopharmaceuticals, medical devices and cosmetics.
Health Canada's Inspectorate Quality Management team, managed within the Quebec Region, conducted four evalua-tions of international mutual recognition agreements covering the Good Manufacturing Practices of drug/medici-nal products with the Czech Republic and Hungary. The regulatory authorities in these two countries are now seen as equivalent relating to the mutual recogni-tion agreements pertaining to drugs and other medicinal products.
Health Canada conducted 51 clinical trial inspections, although our target was 60. This represents 1.2 percent of clinical trials, versus the targeted 1.5 percent. Our goal is to achieve the international level of 2 percent in future years, as advocated by the Standing Committee on Health in 2004. The challenge has been the availability of trained inspectors to perform Good Clinical Practices (GCP) inspections, especially given the time required to train new inspectors and competing priorities. Of the 51 clinical trials, 49 were rated compliant (96.1 percent), indicating that subjects in these trials are not exposed to undue risks and that trials are generally compliant with Canadian regulations.
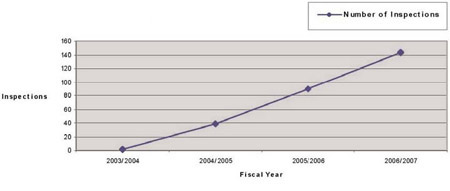
We conducted 144 inspections of medical device establishments representing a 62 percent increase from the previous year. This fell below our target of 170 for two major reasons: more resources were required for inspection follow-up and to deal with medical device incidents that arose during the year. Nevertheless, we still made a significant improvement in the number of medical device establishments inspected, as illustrated in the figure below. 19
Medical Device Establishment Inspections Conducted by Fiscal Year
In 2006, the Health Portfolio received funding over five years to improve preparedness for avian and pandemic influenza. Health Canada has used this funding to develop a guidance document that should accelerate access to available pandemic drugs.
We received 15,295 domestic Adverse Reaction Reports (compared to 15,107 in 2004-2005 and 14,868 in 2005-2006). Also received were 10,686 domestic reports of suspected adverse reactions to pharmaceuticals, biologics, natural health products and radiopharmaceuticals. An initial report and all subsequent information received as follow-up reports are combined and considered to be one case. Most domestic cases were reported by health professionals.
19 http://www.hc-sc.gc.ca/dhp-mps/compli-conform/index_e.html
Transparency, Public Accountability and Stakeholder Relationships
Financial Resources (millions of dollars):
|
Planned Spending
|
Authorities |
Actual Spending
|
|---|---|---|
|
18.3
|
19.5
|
18.4
|
|
Expected Results
|
Performance Indicators
|
Results
|
|
Improved public confidence and trust in the safety of health products, food and the regulatory system Level of public confidence in safety of health products, food and nutrition |
Level of public confidence in safety of health products, food and nutrition Percentage of stakeholders who hold a positive view on HPFB's transparency and openness regarding regulation of health products and food |
We are working on developing a new indicator We are working on developing a new indicator |
In recent years, Canadians have sought increased access to information and decision making about the processes through which food and therapeutic products are regulated. Accordingly, we developed and launched a policy on public input in the review of our regulated products. The policy sets out when Health Canada is expected to seek public input, what information will be disclosed to allow for informed participation, and how public input will be incorporated in a decision about a product. We expect this process to improve the credibility and quality of decisions.
In summer 2006, the Department held consultations on the Blueprint for Renewal initiatives described earlier. We posted a discussion document and electronic workbook on our Blueprint website. More than 300 stakeholders provided views on the vision and objectives. We also held regional meetings as well as consultations on Blueprint themes and initiatives such as the proposed progressive licensing framework, a policy on public input in the review of regulated products, a renewed external charging framework and a regulatory modernization strategy for food and nutrition.
There is no requirement in Canada for sponsors of clinical trials to disclose publicly the existence or results of those trials. Because disclosure of clinical trial information in a publicly available registry would increase access to that information, we convened the first meeting of an External Working Group to develop preliminary options for registration in Canada. This built on international and domestic initiatives as well as results of public consultations that we held in 2005. We developed an online workbook to get public input on policy options, later posting the results on our website. Also posted were the External Working Group's final report and recommendations on how to proceed with registration and disclosure of clinical trial information in Canada.
Strategic Outcome #3(a): Reduced Health and Environmental Risks from Products and Substances, and Safer Living and Working Environments
Program Activity Name: Healthy Environments and Consumer Safety
In 2005 we opened our Office of Paediatric Initiatives as a focal point for an integrated approach to health and safety issues affecting children, from food and nutrition to drugs and vaccines and other therapeutic and diagnostic health products. We have invited stakeholders to identify nominees for the soon-to-be established Paediatric Expert Advisory Committee on Health Products and Food.
Expected Results:
- Reduced risks to health and safety, and improved protection against harm associated with workplace and environmental hazards and consumer products (including cosmetics)
|
Performance Indicators
|
Results
|
|
Percentage of federal public employees remaining at work through and following an injury, illness or traumatic incident |
84% of federal workers who had to leave work for more than 13 weeks returned to work. |
| Treasury Board Secretariat Statistics on leave, accommodation and injury in the workplace | Received 4,747 requests for ergonomic assessments, completed 96%; received 2,376 requests for Fitness to Work health evaluations, completed 90%. |
| Level of client satisfaction with occupational health and contingency planning services | Employee Assistance Program scored above 80% on surveys and interviews, including follow-up surveys that take place three months after service. |
| Client satisfaction surveys | 97.2% of respondents are satisfied with the Employee Assistance Program |
| Percentage of Canadians who are aware that their health can be affected by environmental factors | Development of baseline data taking place. |
|
Reported incidents of product-related deaths and injuries associated with: consumer products; cosmetics; workplace chemicals; new chemical substances; products of biotechnology; radiation-emitting devices; environmental noise; solar UV radiation Reduced health and safety risks associated with tobacco consumption and the abuse of drugs, alcohol and other substances |
Development of baseline data taking place. |
| Prevalence of drug and substance abuse in Canada. Canadian Alcohol and Drug Use Monitoring Survey | Enhanced capacity to assess prevalence of substance use and abuse is being developed. |
| Smoking prevalence in Canada.Reduction in smoking prevalence from 25% to 20% Canadian Tobacco Use Monitoring Survey | 19% of the population, aged 15 years and older smoke. |
Financial Resources (millions of dollars):
| Planned Spending |
Authorities
|
Actual Spending
|
|---|---|---|
|
289.9
|
305.3
|
294.1
|
Human Resources (FTEs):
| Planned |
Planned
|
Difference |
|---|---|---|
|
1,956
|
1,950
|
6
|
Healthy Environments and Consumer Safety activities are founded in legislation that includes the Food and Drugs Act, Controlled Drugs and Substances Restraint Exercise Act, the Hazardous Products Act, the Radiation Emitting Devices Act, the Canadian Environmental Protection Act and the Tobacco Act. These Acts encompass diverse elements such as drinking water quality, air quality, radiation exposure, products of biotechnology and of other new and emerging technologies (including nanotechnology), substance use and abuse (including alcohol), consumer product safety, tobacco and second-hand smoke, workplace health, and chemicals in the environment and the workplace. We are also engaged in the Government's public safety and antiterrorism initiatives, inspection of food and potable water for the travelling public and health contingency planning for visiting foreign dignitaries.
Explanation of the above financial information:
- Variances between planned spending versus total authorities are mainly due to:
- Contributions to the government-wide 2006-2007 $1 billion Spending Restraint Exercise
- Funding from Management Reserve - Public Service Health Program
- Funding from Management Reserve - Litigation Management.
Actual spending is $11.2 million lower than total authorities mainly due to:
- Year end adjustments of Department of Justice expenditures
- Other operating lapses in various programs.
We seek to achieve our priorities through: enhanced compliance with regulations, standards and guidelines; increased awareness of regulated health products; action related to healthy and safe living, working and recreational environments; enhanced involvement of stakeholders; and improved scientific knowledge and capacity to support decision making. We collaborated extensively with partners and stakeholders inside and outside Canada, had an active presence in every region and used risk management processes to identify priorities for action. The Department fulfilled its responsibilities in accordance with sustainable development prin-ciples in order to achieve economic, social, cultural and environmental objectives.
We had success in fulfilling our plans and priorities as set out in the RPP and in addressing new Government priorities that emerged such as the Clean Air Agenda and Environmental Agenda.
In response to concerns raised by the Office of the Auditor General, Health Canada has implemented policies and procedures to improve accountability and stewardship. We have developed short-term and longer-term strategies and priorities for reducing health and environmental risks to Canadians.
Our achievements and challenges in the five sub-activities under this program activity are described below.
Safe Environments Program
Financial Resources (millions of dollars):
|
Planned Spending
|
Authorities
|
Actual Spending
|
|---|---|---|
|
83.5
|
86.1
|
85.0
|
| Expected Results |
Performance Indicators
|
Results
|
|---|---|---|
| Availability and Canada-wide adoption of measures to control the risks to human health posed by environmental contaminants |
Percentage of completion of legislated |
Baselines being established |
| Increased knowledge, understanding and involvement by Canadians in environmental health issues |
Percentage of Canadians who are aware that their health can be affected by environmental factors | |
| Science-based decision making within Canada regarding health risks of environmental contaminants | ||
| Improved scientific knowledge and capacity within the Canadian scientific community and international collaboration on environmental health issues to ensure that Canadians have increased confidence in environmental health information and protection mechanisms |
We continued to identify and manage health risks posed by environmental factors in living, working and recreational environments. The scope of activities included: drinking water quality, climate change, clean air programming, contaminated sites, toxicology, and regulatory activities. The Department was also involved in risk assessment and management of chemical substances, microbiological pathogens, environmental noise, environmental electromagnetic frequencies, solar ultraviolet radiation, as well as preparedness for nuclear and environmental disasters. Research related to these activities and to exposure assessment, hazard identification, mechanistic and population studies supported our science-based decision making.
We were involved in developing elements of the Government's Clean Air Agenda and Environmental Agenda. Collaboration with Environment Canada led to a comprehensive strategy to address air emissions from indoor and outdoor sources. Included is a regulatory approach to emissions of greenhouse gases and air pollutants caused by fuels and consumer and commercial products and those generated by industrial sectors such as electricity generation, upstream oil and gas, pulp and paper, and chemicals.
We carried out our responsibilities under the Canadian Environmental Protection Act, including completing identification and prioritization (“categorization”) of the 23,000 substances on the Domestic Substances List (DSL). About 4,000 were identified as requiring further action. Of these, 1,200 were identified as low priority, 2,600 as medium priority, and 200 as high priority.
The Alberta Region, in collaboration with the Alberta Heritage Foundation for Medical Research, the Alberta Centre for Child, Family and Community Research and key Alberta part- ners and stakeholders, hosted a 2006 Child, Health and the Environment symposium. The subsequent work of the Alberta Child Health and Environment Advisory Group led to recommendations for leaders and champions in Alberta on needed research in this area. As well, the group offered advice to the CanadianCMP Partnership on Child Health and the Environment's third workshop - Research Informing Policy.
Based on these categorization results, Health Canada developed, in conjunction with Environment Canada, the Chemicals Management Plan (CMP), a key component of the Government's Environmental Agenda. The CMP will build on Canada's position as a global leader in safe management of chemical substances and products, with a focus on timely response to environmental and health threats. The CMP enables integration and coordination of the Canadian Environmental Protection Act (CEPA) with other federal legislative tools, such as the Food and Drugs Act, Hazardous Products Act and the Pest Control Products Act. In addition to increasing health and environmental Childresearch, monitoring and tracking, the CMP provides an opportunity for industry and other stakeholders to inform the decision-making process. The CMP sets recomtimelines between now and 2020 for addressing the 4,000 substances that met categorization criteria. The CanadianCMP places the onus on industry to provide the Government with information about how they are safely managing 200 high priority chemical substances. The Government is separating those into batches of 15 to 30 substances and is publishing a batch in the Canada Gazette every three months. The first batch was published in February 2007.
The Department worked with provincial and territorial governments on drinking water quality, including finalization of four Drinking Water Quality Guidelines. In partnership with the Public Health Agency of Canada, we began development of a drinking water advisory system to help track Boil Water Advisories and their sources. We also developed a protocol for a coordinated and systematic federal approach to dealing with outbreaks of water-borne illness and contamination of drinking water.
Health Canada continued its involvement in developing an Air Health Indicator (AHI) for 2008 that will evaluate trends in air quality and associated health impacts over time. We also rolled out the pilot version of the Air Quality Benefits Assessment Tool (AQBAT). It was used to estimate health impacts for the Climate Change and Health Vulnerability Assessment, the benefits of gasoline blends with 10 percent ethanol, and various scenarios under potential new regulations to address air pollution. The Border Air Quality Strategy (BAQS) was completed and included an Air Quality Health Index and two regional pilot studies. One looked at impacts of air pollution on the health of children and other vulnerable populations.
Departmental nuclear emergency response capabilities were used to deal with three events. Canada's networks provided the only international confirmation of the underground nuclear bomb test by North Korea. We conducted analysis of radioactively contaminated mushrooms destined for the U.S. and imported via Canada from Eastern Europe. Our laboratories were involved in assessment and monitoring of over 40 Canadians deemed to be at risk of contamination from a Polonium-210 incident in London, England. Our Department also continued support for development of the Accident Reporting and Guidance Operational System (ARGOS) and the Federal Nuclear Emergency Plan electronic mapping systems as well as work to achieve more rapid processing of emergency samples. We also undertook preliminary analysis and evaluation of the effect of radon in Canadian households to guide the expected Radon Reduction Strategy.
Departmental contributions ensured proper scope of the human health component of the Canadian International Polar Year (IPY) program (2007-2009). Outcomes will include a better understanding of the current health disparities in the North, of the impact of climate change on health, and of community resilience to change. The Department participated in the development of new methods to assess impacts of environmental change on Arctic populations and ensured that high priority health issues will be addressed during IPY
Product Safety Program
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
31.3
|
33.1
|
31.1
|
| Expected Results |
Performance Indicators
|
Results
|
|---|---|---|
| Reduced risk of death and injury from exposure to hazardous products and substances associated with: consumer products; cosmetics; workplace chemicals; new chemical substances; products of biotechnology; radiation emitting devices; environmental noise; solar UV radiation |
Reported incidents of product-related deaths and injuries Level of exposure to hazardous products and substances associated with: consumer products; cosmetics; workplace chemicals; new chemical substances; products of biotechnology; radiation emitting devices; environmental noise; solar UV radiation Working on development of baseline data through performance measurement and the Product Safety Program Capacity Assessment Project. |
Working on development of baseline data through performance measurement and the Product Safety Program Capacity Assessment Project. |
To reduce the number of unsafe consumer products on the market in Canada, we conducted ongoing regulatory, monitoring and compliance activities. We also provided information, education and guidance to the public and industry, such as hazard and technical information to importers and manufacturers to encourage safer product design. In addition we identified unsafe products and product-related risks through research, laboratory testing and investigation, surveillance and monitoring activities and carried out regulatory and policy development. More than 12,000 inspections comprising market surveillance in 10 consumer product areas and over 4,000 customs referrals led to removal of approximately 1,600 product lines from the market. Approximately 27 contracts involving over 2,400 consumer products were tested as part of pre-market assessment to reduce the likelihood of hazardous products available on the market.
For example, we worked on child safety issues including strangulation by the cords of window coverings such as curtains and blinds. We carried out a multimedia information campaign and drafted regulations mandating performance requirements. No incidents related to this issue were reported during the year. We continued development of regulations for children's products, such as crayons, crib toys, clothing, and strollers, under the Lead Risk Reduction Strategy (LRRS). As well, final publication of revised Glazed Ceramics and Glassware Regulations achieved harmonization between American and Canadian standards.
To minimize future threats of skin cancer among today's children, the Department continued with the Sun Awareness Project, a school-based program that educates children about sun exposure. More than 100,000 students have been taught sun safety in elementary and secondary school classrooms since 2005.
Health Canada collaborated with partners in Canada and internationally to implement the Globally Harmonized System of Classification and Labelling of Chemicals (GHS), which, in the Canadian context, deals with consumer chemical products and workplace chemicals.
The Department continued to assess the potential health risks of new chemicals such as fabric dyes and fuel additives, as well as biotechnology products. Approximately 500 new chemical notifications were received.
We completed about 850 assessment reports, significantly reducing our backlog. Risk management actions were renewed for four new perfluorinated chemical substances (commonly used as water and grease repellants for materials such as paper, textiles, carpet and leather, or in the manufacturing process for non-stick coatings on items such as pots and pans), and additional information-gathering conditions were imposed on 12 other new substances.
Weblinks: Product Safety Program
 Product Safety Program
Product Safety Program- Lead Risk Reduction Strategy
- CEPA (Canadian Environmental Protection Act)
- Consumer Product Safety
Workplace Health and Public Safety
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
29.9
|
40.5
|
40.5
|
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Reduced risks to health and safety and improved protection against harm associated with workplace and environmental hazards and consumer products |
Level of client satisfaction with occupational health and contingency
|
|
| Level of reported incidents of deaths and injuries associated with: workplace chemicals; new chemical substances; radiation emitting devices; and environmental noise |
|
|
| Statistics on leave, accommodation, and injury in the workplace |
|
|
| Healthy Public Service |
Percentage of federal public |
|
| Client satisfaction with occupational health and contingency planning services |
|
|
| Improved public health for the travelling public | Percentage occurrence of incidents of gastrointestinal diseases on cruiseships with a target of less than 2% of passengers and crew |
|
The Workplace Health and Safety Program (WHPSP) provides occupational health and safety services to approximately 200,000 federal government employees, protects the health and safety of dignitaries visiting Canada, and promotes health and safety in workplaces. WHPSP also inspects and assesses cargo and passenger conveyances to protect travelling Canadians.
The Department contributed to the health and safety of federal workers by responding to:
- requests for occupational health and safety services;
- ergonomic service requests;
- Employee Assistance Program (EAP) consultations; and
- workplace investigations and consultations.
Survey respondents expressed a high level of satisfaction with the Employee Assistance Program.
To improve the health of travelling Canadians, Health Canada is collaborating with the Public Health Agency of Canada (PHAC) to prepare for and manage events that may require quarantines. Educational material on virus prevention in tourism is a joint effort between the Department and municipal health authorities. We are additionally responsible for International Health Regulations under Canada's obligations to the World Health Organization (WHO). Health Canada continued to inspect ships, aircraft and passenger trains and ancillary services, achieving the results in the table above. In collaboration with the U.S. Centers for Disease Control and the cruise industry, we are combatting the spread of norovirus among travelers through inspections and the reporting of illness in advance of arriving at Canadian ports.
The Department continued to fulfill its obligations for protecting the health and well-being of foreign dignitaries and federal public servants. We organized 114 health plans for visiting dignitaries, including the Prime Ministers of Australia, Japan and senior U.S. government officials. Work on future requirements, including the Francophonie Summit and the Olympic Games in 2010, has already been initiated.
Emergency preparedness activities involving federal, provincial and municipal authorities and agencies will improve preparedness in the event of terrorist acts of a chemical, biological, radiological or nuclear nature. We received additional funding to cover our operational deficit for these activities, stabilize the remaining fiscal year, train staff and upgrade critical technical equipment.
Workplace Health and Public Safety Program
Tobacco Control Program
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
65.7
|
66.4
|
60.8
|
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Reduce smoking prevalence among the Canadian population to 20% |
Smoking prevalence rates
|
19% of the population, aged 15 years and older, smoke. |
| Reduce number of cigarettes sold in Canada by 30% |
Consumption rates - number of cigarettes sold in Canada
|
Cigarette sales declined from nearly 42 billion in 2001 to 30 billion in 2006, a decrease of close to 29%. |
Health Canada continued to lead the Federal Tobacco Control Strategy (FTCS), and worked to reduce health and safety risks associated with tobacco consumption. We developed and administered programs, and partnered with provinces, territories and stakeholder groups to reach individuals with appropriate tools, information and resources. We developed, implemented and enforced regulations pursuant to the Tobacco Act. Our research, monitoring, surveillance and reporting activities added to our knowledge base.
Manitoba and Saskatchewan Region staff in Healthy Environments and Consumer Safety pro-gram and the First Nations and Inuit Health programs collaborated with First Nations organiza-tions to adapt the Tobacco Control Program Retailer Tool kit to ensure it would have cultural significance. First Nations facilitators were trained and several Retailer Tool kit sessions were delivered to urban reserve retailers in Saskatchewan. As well, an Aboriginal tobacco project officer was employed in the region to facilitate regulatory education prior to the gradual intro-duction of enforcement activity in urban reserves. This encouraged voluntary compliance by urban First Nations retailers.
The 10-year objectives of the FTCS, which began in 2001, are to:
- reduce smoking prevalence to 20 percent, from 25 percent in 1999;
- reduce the number of cigarettes sold by 30 percent (from 45 billion to 32 billion);
- increase retailer compliance regarding youth access to sales from 69 percent to 80 percent;
- reduce the number of people exposed to environmental tobacco smoke in enclosed public spaces; and
- explore how to mandate changes to tobacco products to reduce hazards to health.
In addition to the results in the chart above, Canadian Tobacco Use Monitoring Survey (CTUMS) data collected between February and June 2006 identified a continued downward trend in smoking among Canadians aged 15 years and older. Maintaining or improving smoking levels in the over-15 population will require sustained attention. Adults aged 20-24 are an important sub-population, since young adult males report the highest prevalence for smoking (29 percent), with 20 percent smoking daily. The 2006 survey also found the rate of retailers refusing to sell tobacco products to youth was 81.7 percent. By comparison, the rate was 47.9 percent in 1995, when this was first measured.
An evaluation of the first five years of the FTCS found that the Strategy is: making progress in meeting the five objectives: cost-effective, provides good value for money; very relevant to Canadians and stakeholders alike; and has demonstrated success in several program interventions. That evaluation predicted that the first five years of the FTCS should result in health benefits and net economic benefits, such as:
- 25,500 fewer cancer, circulatory and chronic obstructive pulmonary disease cases;
- 6,300 fewer disease-related deaths;
- 153,000 fewer disabilities-adjusted life-years lost;
- $933 million increase in tobacco taxation revenue; and
- $1.46 billion decrease in direct health costs.
The FTCS evaluation found that regulatory interventions and taxation (affecting price) were the most effective policies. It had difficulty in quantifying the impacts of grants and contributions and national mass media campaigns on changing attitudes around tobacco or smoking behaviour.
We used these evaluation results to inform our directions for the subsequent four years of the Strategy. Those directions include a Framework for Action on Tobacco Control for Youth and Young Adults in conjunction with provincial and territorial partners. We also carried out the necessary research to support preparation of regulations that would ban terms such as “light” and “mild” in relation to tobacco products.
Drug Strategy and Controlled Substances
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
79.5
|
79.2
|
76.7
|
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Reduced health and safety risks associated with the abuse of drugs, alcohol and other controlled substances by managing the Controlled Drugs and Substances Act and its Regulations, and providing national leadership for Canada's Drug Strategy |
|
|
Health Canada reduces the harmful consequences associated with substance use/abuse through prevention, enforcement and treatment activities. By providing leadership for Canada's Drug Strategy (CDS), which is transitioning into the National Anti-Drug Strategy, we develop prevention strategies as well as monitor and report current and emerging drug trends to Canadians. We administer the Controlled Drugs and Substances Act, develop new or amended regulations as required, and provide analysis, scientific advice and identification services of seized controlled substances to law enforcement agencies.
According to the most recent Canadian Addictions Survey (CAS) in 2004, alcohol consumption and illegal use of drugs pose serious health threats and have an economic impact estimated at $39.8 billion or $1,267 per Canadian. Alcohol accounted for about $14.6 billion or 36.6 percent of the costs and illegal drugs accounted for approximately $8.2 billion or 20.7 percent. Risky drinking practices and the use of marijuana, cocaine/crack, LSD, Speed and heroin increased between 1994 and 2004. Prescription drug abuse involving opioids, sedatives/hypnotics and stimulants are another concern. Health Canada began to update CAS as an early step in establishing a new Canadian Alcohol and Drug Use Monitoring Survey (CADMUS). This involved work on the core questionnaire and testing of new elements concerning abuse of psychoactive pharmaceutical products.
The Interim Year 2 evaluation report on CDS was tabled and we began to address its recommendations to: streamline indicators, enhance capacity to evaluate the Drug Strategy Community Initiatives Fund (DSCIF), described below, and other programs and develop collaborative models.
The Alcohol and Drug Treatment and Rehabilitation (ADTR) Program continued to fund improved treatment for women and youth. A review of the ADTR concluded that systemic change was needed to move substance abuse treatment systems toward more evidence-informed practices. In response, we met with provincial health representatives to review the program and discuss its reorientation. Three drug treatment courts were fully implemented in Ottawa, Winnipeg, and Regina for a total of six such courts co-funded with Justice Canada. These specialized courts offer treatment as an alternative to incarceration for non-violent offenders who are addicted to cocaine or opiates.
Our drug analysis laboratories worked with law enforcement agencies to identify and analyze approximately 105,000 samples, an increase of six percent over the previous year. Our expert advice and aid led to the dismantling of 37 clandestine labs and destruction of 118,006 seized substances.
Health Canada funded 16 new projects under DSCIF, for a total of 175 projects since its inception. The DSCIF was established under the CDS for initiatives at the national, regional, provincial/territorial and local levels to facilitate community based-solutions to substance abuse problems and promote public awareness. Our $2,912,599 in contribution funding generated almost $2 million in funding from other sources. The DSCIF will be transitioning to reflect new priorities under the National Anti-Drug Strategy, through performance measurement and evaluation work already initiated.
Weblinks: Drugs and Controlled Substances

- Canada's Drug Strategy
- Medical Use of Marihuana
- Controlled Substances and Precursor Chemicals
- Health Canada's Marihuana Supply
- Drug Analysis Service
- National Framework for Action to Reduce the Harms Associated with Alcohol and Other Drugs and Substances in Canada
- Treatment and Rehabilitation
- Drug Strategy Community Initiatives Fund
- Be Drug Wise
- Canada's Drug Strategy Publications
- Overview of Alcohol and Other Drug Use in Canada
Strategic Outcome #3(b): Reduced Health and Environmental Risks from Products and Substances, and Safer Living and Working Environments
Program Activity Name: Pest Control Product Regulation
Expected Results:
- Protected health and environment
- Increased use of reduced-risk pest management practices and products
- Increased public and stakeholder confidence in pesticide regulation
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Access to safer pesticides | Number of new reduced-risk active ingredients available for use in Canada | 5 |
| Percentage of reduced-risk chemicals and percentage of biopesticide active ingredients registered/pending registration in the U.S. that are registered/pending registration in Canada | 71.7% reduced-risk chemicals and 32.5% biopesticides | |
| Number of new active ingredients registered through the PMRA/U.S. EPA Joint Review or work share program | 4 | |
| Number of active ingredients addressed through reevaluation | 10 (244 cumulatively) | |
| Strengthened compliance with the Pest Control Products Act (PCPA) and Regulations | Feedback from public and stakeholders | positive |
| Users informed of reduced risk practices | Number of proposed and final regulatory decisions posted on the website | 38 |
| Transparency of pesticide regulation | Implementation of reading rooms and adverse effects reporting | Implementation complete, no adverse effects requests/reports recieved |
| Improved regulatory efficiencies and cost effectiveness | Efficiency gains achieved through electronic processes and harmonization permit the integration of new science policies and methodologies | Reduction of paper volume, centralized information repository supports more efficient delivery of transparency requirements, automation of Confidential Business Information (CBI) and privacy identification and segregation. |
| Informed public and stakeholders | Feedback from public and stakeholders | positive |
| Number of web hits | 749, 566 | |
| Number of responses provided to the public through the Pest Management Information Service | 6,000 |
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
51.6
|
68.0
|
62.7
|
Human Resources (FTEs):
| Planned | Actual | Difference |
|---|---|---|
|
652
|
574
|
78
|
A significant achievement was the coming into force of the Pest Control
Products Act (PCPA) which strengthened the legal framework for pesticide regulation. Highlights include additional authorities to protect human health and the environment, increased transparency and greater post-market controls.
Explanation of the above financial information:
Variances between planned spending versus total authorities are mainly due to:
- Funding from Management Reserve - Pesticides Regulatory
- Funding for Collective Agreement
The actual spending is $5.3 million lower than total authorities mainly due to:
- Additional collection of revenue well above the historical trend
- Year end adjustments of Department of Justice expenditures.
Many of the targets set in the RPP centred on supporting implementation of the new PCPA, in particular to ensure that stakeholders were well-informed about the features and requirements. Where possible, we incorporated stakeholder feedback to ensure our services would meet their needs. Consultation with our Advisory Council also guided our actions.
International regulatory cooperation was another priority. We worked with member countries of the Organization for Economic Cooperation and Development (OECD), including the United States and Mexico, to increase availability of reduced-risk pesticides for Canadian growers without compromising human health and the environment. We also consulted with and shared scientific knowledge and best practices with other departments, provincial and territorial governments and regulators in other countries.
The Department continued to recruit and retain employees through the Pest Management Regulatory Agency Science Development Program. We reviewed this program, which is aimed at biologists and chemists, and began improvements so that we will have the specialized staff we need to deliver results.
The pesticide regulation program activity encompasses five sub-activities. Achievements under each of the sub-activities are outlined below.
Regulatory Improvement
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
6.0
|
8.3
|
7.8
|
The PCPA which came into force in June 2006 increased the transparency of our activities, enabled greater public participation, expedited registration of lower risk products and developed a new administrative Maximum Residue Limit setting process designed to protect human health and the environment.
We have streamlined the evaluation needed to support registration of lower risk products. To increase the efficiency of our process, we conducted 38 consultations with companies before they sought formal approval of new registrations for lower risk products. We also collaborated with counterparts internationally to develop a regulatory approach for these lower risk pesticides. As well, we finalized a new formulants policy that encourages the use of less toxic ingredients. In our work on the Government's Chemicals Management Plan, we participate in risk assessment and risk management of high priority chemical substances used in pesticide products.
To support transparency requirements of the PCPA, our new electronic Public Registry gave the public access to information about new applications to register or amend pest control products, evaluation reports and conditions of registrations of newly registered or reevaluated pesticides. The PCPA incorporates into law our practice of providing the public the opportunity to comment on proposed major registration decisions.
Mandatory Sales Information Reporting Regulations as well as mandatory Incident Reporting Regulations were published. These provide information to the public about registered pesticides in the Canadian marketplace.
Enhancements to our online services included a Pesticide Products Label database that is updated in real time, and a new Incident Reporting function. Effective electronic submission tools have resulted in over 80 percent of applications being submitted electronically.
New Pest Control Product Registration and Decision Making
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
25.6
|
33.6
|
27.5
|
Before new pesticide products can be registered for use in Canada, Health Canada conducts extensive pre-market assessment to ensure their use poses no unacceptable risks. Using modern scientific methods and international best practices, we conducted human health, safety and environmental risk assessments, as well as value assessments.
We continued to work closely with our counterparts internationally on a joint review program. We commenced two joint reviews with other OECD member countries. The NAFTA joint review program has resulted in 21 joint reviews and seven work shares, facilitating access to 11 conventional and newer reduced-risk chemicals. The first NAFTA pesticide label was approved simultaneously by Canada and the U.S., allowing for free movement of that pesticide between the two countries. The expansion of products with NAFTA labels strengthens competitiveness of North American growers without compromising Canada's high standards for human health and the environment.
Registered Pest Control Products Evaluation and Decision Making
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
9.8
|
12.8
|
12.1
|
Health Canada has a responsibility to reevaluate older pesticides on the market to determine if their use is acceptable in consideration of modern information requirements and scientific assessment methods. We made 17 final reevaluation decisions, 12 proposed decisions and four interim decisions. This brought our cumulative total to 244 pesticides addressed since 2001, or 61 percent of the pesticides in the reevaluation program. At the request of stakeholders, we published a reevaluation status table for the 401 pesticide active ingredients, providing them with regular updates.
Compliance
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
7.6
|
10.0
|
11.2
|
Health Canada has the responsibility to promote, maintain and enforce compliance with the PCPA. We conducted 437 investigations and delivered 13 compliance programs, assessing levels of compliance in blueberry, grape and head lettuce growers; lawn care applicators; and importers of an own-use product. We worked with provincial and other federal regulators and built on our efforts to develop performance indicators for compliance.
Pesticide Risk Reduction in Agriculture
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
2.6
|
3.3
|
4.1
|
In partnership with Agriculture and Agri-Food Canada (AAFC), we developed and implemented commodity-based risk reduction approaches that included two new crops (blueberry and raspberry). Health Canada and AAFC also coordinated pre-submission consultation meetings with U.S. pesticide registrants who own microbial and low risk alternatives. We worked with several industrial sectors to integrate sustainable pest control into their respective strategies for issues such as the Mountain Pine Beetle in the forestry sector and the Richardson's ground squirrel population in the Prairies. As well, we promoted sustainable pest management to municipal associations and at homeowner trade shows.
 Weblinks:
Weblinks:
Strategic Outcome #4: Better Health Outcomes and Reduction of Health Inequalities between First Nations and Inuit and Other Canadians
Program Activity Name: First Nations and Inuit Health
Expected Results:
The objective of the First Nations and Inuit health program activity is to improve health outcomes, by ensuring the availability of, and access to, quality health services, and supporting greater control of the health system by First Nations and Inuit.
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
2,119.1
|
2,126.0
|
2,088.0
|
Human Resources (FTEs):
| Planned | Actual | Difference |
|---|---|---|
|
2,884
|
2,993
|
109
|
Explanation of the above financial information:
Variances between planned spending versus authorities are mainly due to:
- Funding for Federal Contaminated Sites Action Plan
- Contributions to the Government-Wide 2006-2007 $1 Billion Spending Restraint Exercise
- Funding for Non-Insured Health Benefits Program
- Funding for Indian Residential Schools Program.
The actual spending is $38 million lower than authorities mainly due to:
- Delays in Indian Residential Schools Program
- First Ministers and Aboriginal Leader Meeting reprofiles
- Contributions to the Government-Wide 2006-2007 $1 Billion Spending Restraint Exercise
- Operating Budget carryforward in NIHB
- Transfers for capital projects to PACR.
Since development of the RPP, Health Canada has continued to work with First Nations and Inuit and other health partners in delivering programs
and services under the four key priorities.
Continued health-related programs and services
In partnership with First Nations and Inuit, we provided primary health care services in approximately 200 remote communities, and home and community care services in about 600 communities. We directly employed 670 nurses in First Nations communities. Including nurses funded by Health Canada but employed by First Nations, the nursing work force is approximately 1,100. These nurses deliver health services through nursing stations, community health centres, and other health facilities.
Through our Regional Offices and in partnership with First Nations and Inuit, we delivered community programs that focused on children and youth, mental health and addictions, environmental health, and commu-nicable and chronic disease prevention and management. These supplemented health services that provincial, territorial and regional health authorities provided. Through the Non-Insured Health Benefits (NIHB) Program, we provided additional benefits to eligible First Nations and Inuit regardless of their place of residence. These include coverage for drugs, dental care, vision care, medical supplies and equipment, short-term crisis intervention mental health services, and medical transportation.
The Department faces the same challenges as other health care providers such as increasing costs, demand for new health technologies, human resource shortages and an aging population. The First Nations and Inuit health system has additional challenges due to rapidly growing populations, higher than average rates of disease and injuries, and populations living in small communities, often in rural and remote areas.
Improving First Nations and Inuit health outcomes also requires action on broader determinants of health, such as economic development, education, housing and culture, so that communities can become sustainable, culturally strong and economically viable.
| Expected Results | Performance Indicators | Results |
|---|---|---|
|
Strengthened community programs |
Life expectancy (at birth, on and off reserve) |
While still behind the Canadian average, life expectancy for First Nations has increased. In 1980, First Nations males’ life expectancy was 60.9 years, and females’ was68.0 years. By 2001, life expectancy was estimated at 70 years for males and 76 for females. |
| Better health protection | Infant Mortality Rates | Infant mortality rates for First Nations have exhibited a downward trend. The Department is working with stakeholders on improved methodology for tracking this data. |
| Improved primary health care | Birth weight | Statistics for 2000 indicate that 4.7% of First Nations births are classified as low birth weight compared with 5.6% in Canada overall. The high birth weight rate for the First Nations is 21%, almost double the Canadian rate of approximately 13%. Health Canada continues to work in maternal and prenatal health to improve outcomes. |
| Access to Non-Insured Health Benefits.(NIHB) |
NIHB client utilization rates |
NIHB utilization rates represent those clients who receive at least one pharmacy benefit paid through the Health Information and Claims Processing Services system, as a proportion of the total number of clients in the fiscal year. In 2005-2006, the national utilization rate for the pharmacy benefit was 65%. Regional rates ranged from 76% in Saskatchewan to 47% in the N.W.T. and Nunavut. |
Improving quality of and access to health-related programs and services
Health Canada worked towards seamless integration of services; increasing the number of Aboriginal health professionals; supporting First Nations accreditation; improving community dental care capacity; and capital improvements and investments.
Through the community health component of the Labrador Innu Healing Strategy, Atlantic Region staff collaborated with the
Mushuau Innu First Nation in completing a functional and capital plan for a healing lodge
and wellness facilities, planned to
open by June 2007.
Labrador-based staff collaborated with the
Sheshatshiu Innu First Nation to survey approximately 400 community members. The information gathered will be used to plan health prevention and intervention delivery in line with the community's needs. The two groups also collaborated on a training plan for addictions treatment staff to enhance skills and reduce turnover.
We implemented the Aboriginal Health Transition Fund (AHTF). The Fund is designed to improve access to and quality of health services for Aboriginal peoples through better adaptation of provincial and territorial health services and their integration with federal health programs. Examples of achievements include agreements for integration projects in the provinces and territories, and communication activities to build First Nations' awareness of health services. AHTF projects support joint community health planning; coordination and co-location of health services; improved health information management, including better immunization databases and patient charting; increased access to clinical knowledge; and collaborative work on suicide awareness and response. Two advisory groups have been created, with representation from all five National Aboriginal Organizations, provinces and territories, and the federal Health Portfolio.
The Health Integration Initiative, which ended in March 2006, funded eight pilot projects. The national evaluation report, which is expected to be submitted for approval in fall 2007, found that this Initiative led to sustainable partnerships. Projects under AHTF will build on the achievements and lessons learned.
We initiated work to increase Aboriginal students' awareness of health careers; partnered with other stakeholders to improve retention of health care workers in Aboriginal communities; and increased support programs available to Aboriginal health care students. Achievements include: dissemination of career awareness materials to isolated communities using video conferencing and telehealth technologies; facilitating changes to curricula of schools of medicine and nursing; and establishment of First Nations and Inuit capacity for health human resource planning in all First Nations and Inuit provincial, territorial and land claim organizations. Funding totalling $2 million was provided to the National Aboriginal Achievement Foundation to provide bursaries and scholarships for Aboriginal health career students, and to promote health careers in First Nations and Inuit schools. An agreement negotiated with the M�tis National Council will provide $10 million in bursary and scholarship funding for M�tis health career students over the next four years.
Our nurses were supported with professional training and educational resource materials. Training materials include a Primary Care Clinical Practice Guidelines manual, a self-study guide, and an education program for Emergency Labour Competency. These prepare nurses to transition more readily to using an Electronic Health Record system in First Nations communities. National Nursing Security and Awareness Training sessions were also conducted and training materials developed.
The Department launched the Nursing Portal in June 2006 at the Canadian Nurses Association Biennial Conference. This bilingual electronic gateway provides nurses access to an array of resources and services to support nursing practice. Planning continued on enabling Health Canada, as an employer of nurses working in First Nations communities, to post specific employer information, provide education courses and customize clinical resources.
First Nations Community Health Services, National Native Alcohol and Drug Abuse Program Treatment Centres, and Youth Solvent Abuse Treatment Centres have been pursuing accreditation with the Canadian Council on Health Services Accreditation for a number of years. There are 49 First Nations health organizations in the process of being accredited or already accredited. This ensures that First Nations health facilities offer service comparable to accredited provincial/territorial health services.
The Children's Oral Health Initiative was implemented in 140 First Nations communities and services were provided to 8,000 children.
To improve the working environment of clients and staff, and to enhance the quality of health care services, we constructed 14 health facilities, expanded six health facilities, and completed four major recapitalization initiatives.
Given the importance of environmental management in the Departmental Sustainable Development Strategy, we spent approximately $2 million to: conduct pilot Environmental Compliance Audits and Environmental Site Assessments; assess a waste water system to determine whether a new system is needed; upgrade a clinic crawlspace at Norway House Hospital in response to mould; conduct comprehensive microbial investigations at three health facilities to determine the extent and severity of mould contamination; monitor health facilities with previous mould issues; and perform a water well seal integrity study at four wells in Alberta as pilot projects for a larger project.
Promoting healthy living and disease prevention
Health Canada continued to focus on enhancing and strengthening maternal and child health, mental wellness, suicide prevention, prevention of chronic disease, communicable disease readiness and safe drinking water.
The Department funded 40 Maternal and Child Health programs in First Nations communities and supported other communities to provide the program in subsequent years. Activities included: hiring and training family visitors; “train the trainer” postpartum home visit training for 40 nurses and development of a screening catalogue and case management tool kit. In February 2007, Health Canada launched a Healthy Pregnancy campaign to provide women with information to make healthy lifestyle choices before and during pregnancy. Two distinct Healthy Pregnancy campaigns were developed to meet the needs of First Nations and Inuit communities.
Approximately 9,000 children participated in the Aboriginal Head Start On-Reserve (AHSOR) Program. We provided training for outreach and home visit workers in smaller communities and for asset mapping, family support and nutrition. The Department also enhanced the AHSOR capital infrastructure by spending $7.6 million to support capital projects. Seventeen Early Childhood Development single-window service delivery demonstration projects in First Nations communities tested the impact of streamlined funding, program reporting and community development.
A Mental Wellness Strategic Action Plan was developed, and the stakeholder approval process is under way. In addition, 60 community-based Aboriginal Youth Suicide Prevention projects were funded and implemened.
Through the Health Integration Initiativeproject in Nunavut, Health Canada's Northern Region, the Government of Nunavut and Nunavut Tunngavik Incorporated collaborated on identifying actions to better integrate federal and territorial programs and services in maternal and child health, addictions and mental healthand oral health. This was the first tripartite health project to be managed and coordinated by an Inuit organization in Nunavut, enabling it to be delivered from an Inuit perspective.
In November 2006, the Department received approval to provide professional counselling, emotional and and cultural supports to eligible former students of Indian Residential Schools under the new Indian Residential Schools Settlement Agreement.
Activities to advance a strategic and comprehensive approach to other chronic diseases in addition to diabetes were also undertaken. For example, an analysis of effective interventions to prevent chronic disease in indigenous populations was finalized and community-based models for integrated chronic disease prevention and management were developed and supported.
Pandemic influenza plans were drawn up to support First Nations communities in local preparedness and ensure better regional communicable disease emergency coordination with provinces, territories and stakeholders. A stockpile of personal protective equipment was replenished for use by front-line workers in First Nations communities in communicable disease emergencies. Health Canada provided support to the Assembly of First Nations to pilot and test a culturally-appropriate approach to pandemic planning in three First Nations communities.
By March 2007, over 40 percent of drinking water distribution systems, cisterns and community wells were routinely monitored for bacteriological contaminants as recommended in the Guidelines for Canadian Drinking Water Quality. A total of 153,604 water samples were analyzed in First Nations communities, an increase of 29 percent from last year. Health Canada is working with First Nations leadership to help communities improve their understanding of and response to Drinking Water Advisories. Six out of our seven regions now have water databases in place to monitor sample results. No instances of water-borne disease outbreaks were identified.
The British Columbia Region continues working toward accurate and timely reporting from all communities who are engaged in the Drinking Water Safety Program, while developing other communities. There are 136 community water technicians trained and supported to provide sampling programs for community water systems, in order to meet Canadian drinking water guidelines. Health Canada and the water technicians are also preparing for spring flood conditions.
Health Canada developed a school kit to raise awareness and instill an appreciation for the importance of clean, safe and reliable water
among First Nations children. We also developed communication products to focus on preventive activities related to Drinking Water Advisories.
Improving accountability and performance measurement
The Department undertook initiatives to support and enhance health surveillance, information analysis, research, and data collection and analysis. An Infant Mortality Working Group was created, with participation from various levels of government, National Aboriginal Organizations and academics, to address deficits in infant mortality data for First Nations, Inuit and M�tis. The Group is developing several pilot projects, to be funded by Health Canada and the Public Health Agency of Canada, that will improve data quality and coverage as well as local data capacity.An integrated Aboriginal health surveillance strategy has been initiated to advance the Integrated Public Health Surveillance and Information plan. Pilot projects for enhanced health information management have been developed in the Alberta and Atlantic regions. These will improve surveillance systems in those regions while providing direction for other regional development in the long term.
A key challenge in reporting health information for First Nations and Inuit is to identify Aboriginal people from within administrative databases. Health Canada has funded and participated in an innovative Statistics Canada project to develop life expectancy estimates in Inuit-inhabited regions. This has led to new data on northern life expectancy. Health Canada is also supporting regional data infrastructure through the Health Data Technical Working Group, with representation from the National Aboriginal Organizations. This Group provides technical expertise on data and epidemiological matters and produces in-house statistics for the Department. Their major product is the Statistical Profile on the Health of First Nations in Canada.
The First Nations Regional Longitudinal Health Survey (RHS) is a “survey of First Nations developed by First Nations,” and is funded by Health Canada through contribution agreements. Results of the 2002-2003 RHS, released just prior to last fiscal year, have been shared with federal and provincial governments and First Nations to support evidence-based planning and evaluation. Discussions are ongoing on the next cycle of the RHS.
Health Canada finalized a Performance Measurement Strategy Reporting Template to support First Nations recipients in data collection at the community level beginning in 2008-2009.
In the RPP, the Department identified programs and services that were the organizational base for our initiatives across all four priorities just described. The charts below set out details regarding our use of resources, expected results, performance indicators and results achieved for each program area.
First Nations and Inuit Community Health Programs
Child and Youth programs
Maternal, infant and child health; increasing children's knowledge of language and culture; and their readiness for school are the main priorities of child and youth programs. The programs are: Aboriginal Head Start On-Reserve; Canada Prenatal Nutrition; Fetal Alcohol Spectrum Disorder; and Maternal and Child Health.
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Improved continuum of programs and supports in First Nations and Inuit communities | Number and percentage of communities with programs | 40 Maternal and Child Health programs have been established. Other communities have been supported to provide the program in subsequent years. |
| Increased participation of Aboriginal individuals, families, and communities in programs and supports | Number and type of participants in programs by program type | Approximately 9000 children participated in Aboriginal Head Start On Reserve programs. |
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
| 108.3 | 110.4 | 97.9 |
Mental Health and Addictions Programs
These programs provide culturally appropriate counselling, addiction prevention and mental wellness services that are largely delivered by Aboriginal people. They include: Building Healthy Communities; Brighter Futures; National Native Alcohol and Drug Abuse Program - Residential Treatment; National Native Alcohol and Drug Abuse Program - Community-based; Youth Solvent Abuse Program; First Nations and Inuit Tobacco Control Strategy 21 ; National Aboriginal Youth Suicide Prevention Strategy; Labrador Innu Comprehensive Healing Strategy; and Indian Residential Schools Resolution Health Support Program (formerly known as Indian Residential Schools - Mental Health Support Program).
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Improved continuum of programs and services in First Nations and Inuit communities | Number and percentage of communities with programs | All First Nations communities have access to National Native Alcohol and Drug Abuse Program, Youth Solvent Abuse Program and mental health programs. 60 community-based suicide prevention projects were delivered in . |
| Increased participation of First Nations and Inuit individuals, families and communities in programs and services | Number and type of participants in programs by program type | All former students of Residential Schools and their families are now able to access health supports via the Indian Residential Schools Resolution Health Support Program. 585 clients accessed professional mental health services. |
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
147.7
|
191.0
|
166.4
|
21 As part of the federal expenditure review, $2.5 million in 2006-2007, $8.3 million in 2007-2008, and $10.8 million in ongoing funding for the First Nations and Inuit Tobacco Control Strategy was targeted for reduction. Funding for 2007-2008 will be directed at completing initiatives and working with partners to develop a new policy approach.
Chronic Disease and Injury Prevention Programs
Over the long term, these programs will contribute to prevention of chronic disease and injuries within First Nations and Inuit communities. They include: Aboriginal Diabetes Initiative; Nutrition and Physical Activity Promotion; and Injury Prevention.
| Expected Results | Performance Indicators |
Results |
|---|---|---|
| Improved continuum of programs and supports in First Nations and Inuit communities | Number and percentage of communities with programs | More than 600 communities have access to health promotion and diabetes prevention services. Examples include: school policies emphasizing healthy snacks, children’s camps that focus on preventing diabetes through healthy lifestyles, and walking clubs to help those at risk. |
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
37.0
|
24.6
|
26.4
|
First Nations and Inuit Health Protection and Public Health
Communicable Disease Control Programs
These programs protect First Nations and Inuit communities from communicable diseases through measures to manage, contain and control risks of outbreaks. They include: Tuberculosis; Immunization; HIV/AIDS; and Communicable Disease Emergencies.
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Improved access to communicable disease prevention and control programs for First Nations and Inuit individuals, families and communities |
Number and percentage of communities with programs |
All First Nations communities on-reserve are supported Inuit communities are supported in communicable disease prevention and control through contribution agreement funding or through National Aboriginal |
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
27.6
|
26.2
|
30.6
|
Environmental Health and Research Programs
These programs create and maintain healthy and safe community environments through: investigation of potential environmental health-related outbreaks; raising awareness of water-borne, food-borne and vector-borne illnesses including health problems from poor indoor air quality, such as mould. They provide for pest control and build community human resource capacity to adapt to environmental conditions, to maintain safe environments and to deal safely with hazards. These programs include: First Nations Water Management Strategy; West Nile Virus; Contaminated Sites; Transportation of Dangerous Goods; Food Safety; Facilities Health Inspections; housing; and research.
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Improved environmental health risk management | Number of communities with environmental health officers Number of communities equipped with water testing/sampling kits | 478 communities have dedicated environmental health officers. 546 communities are equipped with water testing/sampling kits. |
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
48.9
|
46.3
|
39.0
|
First Nations and Inuit Primary Health Care
Comprehensive health care services are provided to remote First Nations and Inuit settlements to supplement primary care provided by provincial, territorial and/or regional health authorities. These include emergency and acute care. Health Canada ensures links to other health care providers and/or institutions as required by the client's condition. The continuum of care includes illness and injury prevention and health promotion such as the Home and Community Care Program and the Oral Health Strategy.
| Expected Results | Performance Indicators | Results |
|---|---|---|
| Improved access to primary health care programs and services for First Nations and Inuit individuals, families and communities |
Number and percentage of communities with programs |
605 of 645 (94%) First Nations communities have access to home care services, represents 97% of eligible population. 55 (100%) Inuit communities have access to home care services, represents 100% of eligible population. |
| Number of treatment centres by type, in the communities | 54 Alcohol and Drug Abuse and 8 Youth Solvent Abuse treatment centres are in operation. | |
| Eligible client utilization rates of NIHB - Dental Benefits | In 2005-2006, the national utilization rate for the dental benefit was 37%. Regional rates ranged from 46% in Quebec to 30% in Manitoba. |
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
247.0
|
270.5
|
289.0
|
Non-Insured Health Benefits
The NIHB program provides 765,000 registered First Nations and recognized Inuit with a limited range of medically necessary goods and services not provided through private or provincial and territorial health insurance plans. The benefits include: dental care, vision care, pharmacy benefits (prescription drugs and some over-the-counter medication), medical supplies and equipment, crisis intervention, mental health counseling, transportation to medically required health services and payment of health premiums for individuals in Alberta and British Columbia.
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
966.3
|
1,018.7
|
996.4
|
Governance and Infrastructure Support
Health Governance and Infrastructure Support aims to increase First Nations and Inuit control over health programs, establish an adequate First Nations and Inuit infrastructure and services and improve capacity to generate and use health information. These activities include: health planning; capacity building; integration and coordination of health services; stewardship and health research; knowledge and information management. In November 2006, the Government signed a Memorandum of Understanding with the Province of British Columbia and the British Columbia First Nations Leadership Council. This MOU committed the parties to building a tripartite relationship for improving the health of B.C. First Nations, and led to the signing of the Tripartite First Nations Health Plan for British Columbia in June 2007.
Financial Resources (millions of dollars):
| Planned Spending | Authorities | Actual Spending |
|---|---|---|
|
536.4
|
438.7
|
442.3
|
Weblinks:

- Non-Insured Health Benefits 2005-06 Annual Report
- Non-Insured Health Benefits Program
- Aboriginal Head Start On-Reserve
- Fetal Alcohol Spectrum Disorder
- Aboriginal Diabetes Initiative
- Injury Prevention
- Indian Residential Schools Resolution Health Support Program
- National Native Alcohol and Drug Abuse Program
- Drinking Water Quality
- Immunization Schedule for Infants and Children
- Targeted Immunization Program
- e-Health
- Aboriginal Health Human Resources Initiative
