Common menu bar links
Breadcrumb Trail
ARCHIVED - Canadian Food Inspection Agency
 This page has been archived.
This page has been archived.
Archived Content
Information identified as archived on the Web is for reference, research or recordkeeping purposes. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats on the "Contact Us" page.
2. Analysis of Program Activities by Strategic Outcome
2.1 How the Agency Plans and Reports
The planning requirements of the Canadian Food Inspection Agency (CFIA) are set out in the Canadian Food Inspection Agency Act and in Treasury Board policies and guidelines. The CFIA is required to produce an annual Report on Plans and Priorities (RPP) and an annual Performance Report (PR).
In accordance with Treasury Board requirements on the Management of Resources and Results Structure, the CFIA’s planning and reporting framework is based on Strategic Outcomes (SO). These SOs are outlined in its Corporate Business Plan 2003–08 and are elaborated in detail in the CFIA’s Reports on Plans and Priorities and the Performance Reports. Financial information in these reports is also aligned in this manner.
Under each Strategic Outcome, the CFIA reports on ongoing activities under each sub-activity as outlined in the Report on Plans and Priorities. In accordance with the CFIA’s 2006–07 Report on Plans and Priorities, the Performance Report is structured around Strategic Outcomes, program sub-activities and expected results. Progress on Special
Initiatives is reported in Section 3.1 where risk mitigation strategies are also identified by this symbol![]() .
.
Refer to Section 4.2 — Notes on Reporting Against the Report on Plans and Priorities for more information.
Reporting Performance
In Section 2.3, performance information and expected results for each Strategic Outcome are, where possible, described and measured against targets using compliance and other relevant performance indicators. Targets measure the performance of industry and the CFIA against expected results set by the CFIA.
The 2005–06 Performance Report marked the first time that the CFIA reported against specific targets. The CFIA’s 2006–07 Performance Report builds upon this work, and it reports on a twoyear trend for performance indicators that are based on outcomes and have established targets, adding substantially to the CFIA performance story. Future Performance Reports will include longer trending periods and a broader set of performance indicators.
In 2006–07, a review was completed to perform a preliminary assessment of the systems controls (both manual and automated). In order to carry out the review, the CFIA identified sources of data; identified the controls in place through interviews and; made a preliminary assessment of those controls based upon manual and system components and the impact of downstream manipulation and management review. The assessment of the system controls, was the first step in a continuum of reviews, which will serve to ensure that CFIA is able to provide accurate data for performance reporting.
Building on the work of the review, which identified that action taken in management review and control weaknesses would be beneficial, the CFIA also implemented additional controls on all performance information gathered and included it in the Performance Report. These controls included: implementing rigor in the collection, review and substantiation of targeted data; building further management accountability into the process; and supplementing control processes by identifying and verifying the name of data sources, the time period covered, calculations and calculation methodology.
The assessment of quality of data found in this Performance Report is based upon the preliminary assessment of the system controls, as well as the additional management controls implemented for the preparation of the report.
Table 2.1.1 provides a summary of ratings for data quality for the targeted data reviewed, including the additional management controls implemented for the preparation of the Performance Report. The Agency will continue to review certain data systems and improve upon management controls and present those findings in future performance reports.
Key findings of this review include that 70% of the targeted data have controls rated as “good”. The balance of targeted data reviewed was rated as “reasonable” and has no more or less significance or impact on overall data quality than those rated as “good”. While two thirds of the targeted data reviewed have adequate controls, it has been concluded that targeted data should be tested, with controls imposed and documented in an information management system.
Performance Targets
In 2005–06, the CFIA set up a working group to begin to establish and formalize performance targets in all critical program areas. The targets set during 2005–06 are used in the 2006–07 Performance Report and are based on historical averages of actual performance or on expected results of effective programming (e.g., compliance rate for industry conformity to regulatory standards, control of entry and spread of animal and plant diseases). Industry compliance targets of less than 100% are representative of the CFIA risk-based inspection approach, which targets areas of high risk and past non-compliance.
When interpreting performance information we must consider that the CFIA carries out monitoring activities on an industry-wide basis and also conducts targeted monitoring of problem areas along the agri-food continuum (farm to plate) and within specific food sectors. When a program specifically targets areas of past non-compliance or responds to complaints, the compliance levels identified cannot be fairly considered against the industry-wide compliance. Hence, while clearly indicated in the report, these targets are not reported in the same manner as results of broader monitoring programs. The CFIA strives to promote improved industry compliance on a year-to-year basis and has an enforcement and compliance strategy to address all instances of non-compliance. Results achieved against targets, as well as non-targeted performance information are reported in the following section of this report.
The 2006–07 Performance Report is the second report in which the CFIA reported results against targets that were established in 2005–06. To more comprehensively report on its core performance in the future, the CFIA plans to expand and further refine these targets. As a first step, the CFIA refined its Strategic Outcomes and Program Activities — approved in May 2007 by Treasury Board. Subsequent to this, the CFIA began reviewing its Performance Measurement Framework consistent with the Treasury Board Management Resources and Results Structure Policy implementation with the intention of reviewing and building upon the targets and indicators set in 2005–06.
|
Table 2.1.1 — Rating Summary of Data Systems and Process Controls |
||
|
Data Systems and Controls Rating |
Definition |
Number of Data Systems and Controls |
|
Good |
Has clearly defined policies and procedures in place |
31 |
|
Reasonable |
Has compensating controls in place to make up for lack of defined |
12 |
|
Weak |
Has no defined policies/procedures or compensating controls in place |
1 |
Assessment of ComplianceAs a regulatory agency, the principal means by which the CFIA carries out its mandate is by measuring rates of compliance with Canadian food, animal, and plant regulatory requirements. The CFIA promotes compliance by conducting inspections, audits, product sampling and verifications. The CFIA also carries out education and awareness activities to increase regulated parties’ understanding of statutory requirements and standards. Compliance rates are an indicator of the extent to which industry has adhered to federal acts and regulations. The CFIA takes a number of approaches to assessing compliance. These include:
The compliance result of a targeted program is qualitatively different from that of a monitoring program in terms of its implications for food safety, animal health or plant protection. The compliance tool chosen by the CFIA is thus based on risk. Where compliance rates appear in this report, the approach used to assess compliance is noted. The complexity of the agri-food sector and the inherent variability of the biological and production systems underpinning it are such that some degree of non-compliance is inevitable. A compliance rate of less than 100% means that some proportion of the facilities or products inspected by the CFIA has failed to meet certain requirements or standards as defined by the regulations. Major variances have the potential to pose a significant risk to human, animal or plant health and/or other program objectives. These are always met with vigorous enforcement actions to assure protection of Canadians and the plant and animal resource base. Some deficiencies represent “minor” variances and do not pose a significant risk to human, animal or plant health. |
2.2 Office of the Auditor General Reports
|
The Auditor General’s assessment of CFIA’s performance information is presented in section 2.2.2 of this report. This information, which is presented in section 2.3, has not been audited; the assessment is done only at a review level of assurance. The Auditor General’s audit opinion on the CFIA financial statements is presented in section 3.5.1 of this Performance Report. These audited statements are presented in section 3.5. The Auditor General has not reported on other sections of this report. |
2.2.1 Management Representation Statement
The Canadian Food Inspection Agency's (CFIA) 2006–07 Performance Report for the year ending March 31st, 2007, was prepared under the direction of the president and the Executive Management Committee of the CFIA and approved by the Minister of Agriculture and Agri-Food Canada. In accordance with the Canadian Food Inspection Agency Act, the report also includes an assessment of the fairness and reliability of the performance information conducted by the Auditor General of Canada.
I submit for tabling in Parliament, the 2006–2007 Performance Report for the Canadian Food Inspection Agency.
This document has been prepared based on the reporting principles contained in the Guide for the Preparation of Part III of the 2007–08 Estimates: Reports on Plans and Priorities and Departmental Performance Reports:
- It adheres to the specific reporting requirements outlined in the Treasury Board Secretariat guidance;
- It is based on the Agency’s Strategic Outcome(s) and Program Activity Architecture that were approved by the Treasury Board;
- It presents consistent, comprehensive, balanced and reliable information;
- It provides a basis of accountability for the results achieved with the resources and authorities entrusted to it; and
- It reports finances based on approved numbers for the Estimates and the Public Accounts of Canada.
Carole Swan
President
2.2.2 Auditor General’s Assessment of Performance Information
2.3 Performance by Strategic Outcome
The CFIA presents its performance information based on the Program Activity Architecture, in line with the presentation of planning information in the Report on Plans and Priorities. For each Strategic Outcome, the CFIA sets the results context in terms of outcomes for Canadians and elaborates the performance story at the Program Activity level. The focus is on program sub-activities, as the CFIA has identified expected results at this level. Balanced performance information is provided for each expected result in support of demonstrating progress towards achieving the overarching Strategic Outcome.
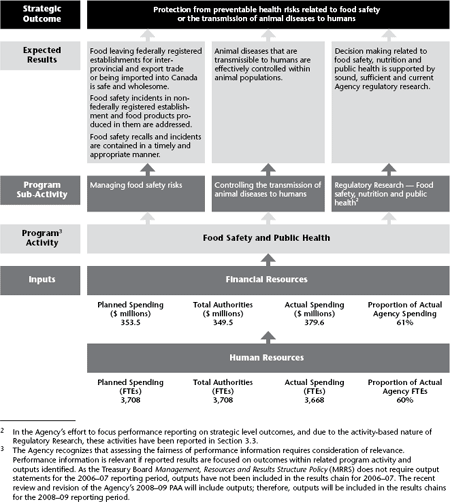
2.3.1 Strategic Outcome: Protection from preventable health risks related to food safety or the transmission of animal diseases to humans
|
|
The CFIA, along with many federal, provincial, territorial and municipal organizations, is working to protect the health of Canadians. The CFIA’s primary contribution is helping to ensure that food is safe, that consumers have appropriate information on which to base healthy food choices and that the risk of contracting animal diseases (e.g., avian influenza (AI)) is minimized.
To achieve this outcome, the CFIA works in collaboration with a number of partners and stakeholders, including Health Canada (HC), the Public Health Agency of Canada (PHAC), Agriculture and Agri-Food Canada (AAFC), and provincial and territorial governments.
In 2006–07, the CFIA spent approximately 61% of its budget to achieve this Strategic Outcome.
2.3.1a Program Sub-Activity: Managing food safety risks
In managing food safety risks at the federal level, the CFIA is responsible for food inspection and compliance activities, and Health Canada is responsible for the development of food safety policies, standards and regulations, which the Agency enforces.
Of the $379.6 million the Agency spent to achieve this Strategic Outcome in 2006–07, approximately $284.0 million was devoted to managing food safety risks.
|
Expected Result: Food leaving federally registered establishments for inter-provincial and export trade, or being imported, is safe and wholesome |
Inspection is a critical element in ensuring that domestic and imported food products do not pose a significant threat to the health of Canadians. The CFIA inspects federally registered food establishments and food products to verify that food traded inter-provincially and internationally, or food imported into Canada, is safe and wholesome. The CFIA identifies and focuses its inspection activities on high-risk sectors and commodities as part of its proactive riskmanagement approach.
Establishment inspections
In order to ship certain products to other provinces and countries, food processing plants must be federally registered. Generally each establishment is subject to an initial and an annual registration process to confirm that critical systems and controls are in place. The CFIA inspects these plants regularly to ensure that they comply with federal regulations. The level of inspection depends on the spectrum of risks managed by the Agency, with higher-risk products or manufacturing processes receiving more attention. While most facilities are inspected at least once each year, some are inspected every day.
The CFIA works toward having industry achieve full compliance with legislative requirements. However, with the complexity and inherent variability of the agriculture and food processing and distribution sectors, some degree of non-compliance is inevitable. The CFIA, therefore, focuses its inspection work on systems, processes and facilities that have the most direct effect on the safety of the product. The CFIA’s working assumption is that as industry improves compliance, food safety risks will diminish.
When CFIA inspectors detect non-compliance, the processing establishment is required to correct the related deficiencies. Serious deficiencies are corrected on a priority basis, and in some cases, production is stopped and products are recalled from the marketplace. Non-compliant facilities are subject to re-inspection to confirm that they have taken corrective steps identified by inspectors.
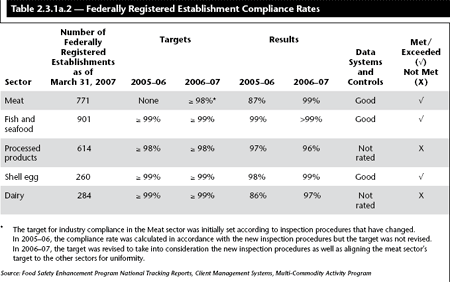
![]() The target compliance rates for various commodities requiring federally registered establishments are listed in Table 2.3.1a.2.
The target compliance rates for various commodities requiring federally registered establishments are listed in Table 2.3.1a.2.
In 2006–07, CFIA monitoring inspections indicated a high level of compliance in the registered sector. These compliance rates provide assurance that the risks to food safety in the federally registered sector are well managed and, as a result, that the food it produces is safe.
![]() The target for federally registered establishment compliance was met in the meat, fish and seafood, and the shell egg programs. It was not met for processed foods or dairy, although the dairy sector showed signs of improvement from 2005–06.
The target for federally registered establishment compliance was met in the meat, fish and seafood, and the shell egg programs. It was not met for processed foods or dairy, although the dairy sector showed signs of improvement from 2005–06.
As of December 2005, the meat slaughter and processing industry moved to a new food safety control system (Hazard Analysis Critical Control Point system or HACCP). Under the HACCP inspection system, industry identifies specific hazards and measures for their control to ensure the safety of food. CFIA inspectors evaluate industry compliance to regulatory requirements through audits, inspections and sampling. The change to the more demanding HACCP system was a significant one for the industry and a concerted effort involving CFIA inspection staff and industry management helped to improve the rate of compliance during the transitional phase. As of the end of 2006–07, almost all federally registered meat establishments were in compliance with the HACCP system, with compliance rates rising from 87% in 2005–06 to 99% in 2006–07. The CFIA will undertake proceedings to cancel the licence of establishments unable to demonstrate satisfactory compliance.
For the second year in a row, compliance rates in fish and seafood establishments remained at 99% or greater. The compliance rate for fish and seafood is “post-corrective action” meaning establishments that have undertaken corrective actions before the end of the reporting period, resulting in compliance, are included. The high rate of compliance (post-corrective action) in the monitoring of domestic fish and seafood establishments demonstrates the ability of non-compliant facilities to collaborate with the Agency to implement acceptable corrective actions, which result in domestic products that meet health and safety standards. In addition to the public health benefits, this high compliance rate also results in economic benefits for Canadians, in terms of market access for these products.
The decrease in compliance this past year by 1%, from 97% (2005–06) to 96% (2006–07), for processed products establishment inspection is a direct result of the implementation of a consistency project that has highlighted a number of additional elements which must be noted as deficiencies by inspectors. This improved process provides a higher quality of compliance overall, however, it has resulted in an increased number of deficiencies being noted in the short term. The CFIA is in the process of updating the Establishment Inspection Manual to improve clarity and to ensure continued consistency in the application of regulations.
For the shell egg sector, CFIA has established a target of 99%. The CFIA fully met its expected target for this sector. This is an improvement of 1% over last year’s results, reflecting CFIA’s continuing endeavours to improve compliance and ensure the health and safety of Canadians.
Compliance for the dairy program has increased from 86% to 97% over the past year but is not yet meeting its 99% target. In the year 2005–06, the dairy program introduced new control standards and inspection approaches that resulted in an overall reduction of compliance due to the more comprehensive controls. As the industry has adjusted to the new control and inspection approaches, the compliance rate for federally registered dairy establishments has climbed to just below the target during the 2006–07 reporting period.
By ensuring maximum compliance in federally inspected establishments, the CFIA helps to protect Canadians from preventable health risks related to food safety.
HACCP Recognition4
In order for a meat establishment to be granted the mandatory HACCP recognition, the establishment must be proven to meet a variety of conditions outlined by regulation. As of the end of the 2006–07 reporting period, all of the 742 federally-registered meat and poultry establishments had been evaluated by the CFIA to ensure compliance with the requirement to adopt the HACCP approach.
The number of federally registered establishments with recognized, voluntary HACCP systems in place also increased this year. As of March 31, 2007, the following ratios indicate the number of voluntary HACCP systems put in place, by commodity: 525 processed product establishments out of a possible 614; 18 egg establishments out of a possible 281; 66 dairy establishments out of a possible 282; and six honey establishments out of a possible 206. The CFIA will continue to promote further adoption of the HACCP approach in the remaining voluntary establishments and to assist processors in identifying all critical stages that may affect the safety and quality of food products.
Product testing
In addition to inspecting food processing establishments, the CFIA tests regulated commodities to confirm that they comply with applicable laws and regulations. This testing assists in verifying that domestic and imported food products do not pose a significant risk to the health of Canadians.
Health Canada establishes regulations under the Food and Drugs Act and policies related to chemical residues in foods. These include maximum levels for pesticide residues, veterinary drug residues and environmental contaminants in food.
The CFIA’s program for monitoring chemical residues has monitoring, surveillance and compliance components. In the monitoring phase, an unbiased selection of samples is taken from the normal food supply and is tested for chemical residues. The CFIA uses the monitoring data to prevent potential health hazards caused by chronic exposure to contaminants. This is done by monitoring areas of concern, examining trends of prevalence and developing effective action plans to deal with health risks. Health Canada conducts re-evaluations for pesticides and other contaminants in the food supply, to verify that standards remain appropriate or to modify standards where necessary.
Every finding of chemical residues in food products is evaluated to determine if there is a violation of Canadian standards and if the violation poses a potential health risk to consumers. Where maximum levels have not yet been established by Health Canada for specific chemical residues in particular foods, any residue found is considered to be a violation. In many cases, such violations may not pose an unacceptable health risk, however, the CFIA investigates all violations to promote compliance.
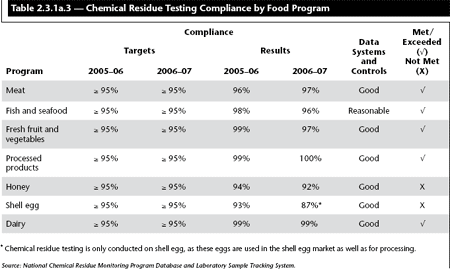
Table 2.3.1a.3 illustrates the proportion of domestic samples with compliant maximum residue levels.
![]() The 2006–07 target for product testing (chemical residue) was set at greater or equal to 95% for all programs.
The 2006–07 target for product testing (chemical residue) was set at greater or equal to 95% for all programs.
The target was met or exceeded in five of seven programs: meat (97% compliance), fish and seafood (96% compliance), fresh fruit and vegetables (97% compliance), processed products (100% compliance) and dairy (99% compliance). These results are consistent with the 2005/06 results. Slight declines in compliance (2%) for fish and seafood and fresh fruit and vegetables are likely attributable to the sample size and are not considered significant as the targets were exceeded.
The 95% target was not met in two programs: honey and shell egg. Honey had a compliance rate of 92%, down from 94% in 2005/06. Shell egg had a compliance rate of 87%, down from 93% in 2005/06. Health Canada has yet to establish maximum residue levels for both commodities and the results reflect the detection of extremely low levels of chemical residues. Accordingly, despite not meeting the target, there is no significant risk to consumers.
CFIA, in partnership with Health Canada, continues to work collaboratively to take precautionary measures such as setting working residue levels for chemical contaminants that do not yet have established maximum residue levels. Through this work the CFIA continues to safeguard the food supply for Canadians.
Enforcement
Once inspections have determined that regulated parties do not meet their legislative requirements, the CFIA responds to this noncompliance. Specific responses can be directed at the product and/or the regulated party.
In 2006–07, CFIA investigated 372 instances of non-compliance as compared with 318 in 2005–06 to the Canada Agricultural Products Act, the Fish Inspection Act, the Food and Drugs Act, and the Meat Inspection Act. Combined with investigations carried over from previous reporting periods, these investigations resulted in 48 convictions and $221,750 in fines. In 2005–06, there were 41 convictions and $95,705 in fines. The number of enforcement actions is only a partial indicator for measuring the effectiveness of CFIA’s continuum of enforcement work such as the issuance of detentions, seizures and warning letters.
|
Expected Result: Food safety incidents in nonfederally registered facilities and food products in them are addressed |
The “non-federally registered” food sector covers a wide range of products, including infant foods, alcoholic beverages, bakery products and cereal products. The establishments that produce these products are not federally registered. Jurisdiction over the non-registered sector is shared between the federal, provincial and territorial governments. The CFIA enforces the food safety provisions of the Food and Drugs Act and regulations for foods in this sector. The CFIA monitors non-federally registered food sector commodities using a risk-based management model, prioritizing compliance activities in areas of high risk, enforcement actions in areas of low compliance, and gathering intelligence related to contraventions.
Central to the monitoring of non-registered products and facilities in Canada are scientific committees. These consist of food safety experts from the CFIA, Health Canada and other government departments and agencies. These committees evaluate potential risks to food safety, and strategies to assess those risks are developed on a project-by-project basis. These assessments take into account complaints from the public or industry, information relating to recalls or foodborne illness, and review of the scientific literature. The committees then identify and prioritize these risks in terms of their potential implications for food safety and develop strategies for managing them effectively.
In 2006–07, the CFIA, in collaboration with food safety experts and science committees, developed inspection projects targeting areas of high risk and prioritizing enforcement actions in areas of past low compliance. These projects were intended to improve the compliance of imported and domestic foods leaving nonfederally registered establishments with health and safety standards set by Health Canada.
![]() The target to measure activities related to the safety of food leaving non-federally registered establishments was to establish inspection projects to address ≥90% of the major health risks identified by the science committee. The CFIA exceeded this target in 2006–07 by completing projects covering 94%
of the identified major health risks. The Food Safety Program developed 13 projects that addressed 16 of the 17 identified major health risks by the Food Safety Science Committee. A work specification was also developed for 2007–08 for the remaining project on
Ochra-toxin in foods. Trending data are not available for this expected result as no data were collected or reported in
2005–06. Data reporting and trending for this target will be available next year.
The target to measure activities related to the safety of food leaving non-federally registered establishments was to establish inspection projects to address ≥90% of the major health risks identified by the science committee. The CFIA exceeded this target in 2006–07 by completing projects covering 94%
of the identified major health risks. The Food Safety Program developed 13 projects that addressed 16 of the 17 identified major health risks by the Food Safety Science Committee. A work specification was also developed for 2007–08 for the remaining project on
Ochra-toxin in foods. Trending data are not available for this expected result as no data were collected or reported in
2005–06. Data reporting and trending for this target will be available next year.
|
Expected Result: Food safety recalls and incidents are contained in a timely and appropriate manner |
The CFIA works in partnership with Health Canada, Public Health Agency of Canada, provincial public health and food/agriculture inspection agencies as well as the food industry to operate a food safety and emergency response system. This response system can be triggered by a consumer complaint, information from industry or trading partners, or the results of inspection and monitoring activities of the CFIA or provincial food inspection agencies. Potential hazards, in the form of undeclared allergens, microbiological or chemical contamination, or extraneous material are investigated and appropriate risk management actions are taken to protect consumers.
Investigations and Recall Incidents
![]() One of the key measures that the CFIA uses to assess its performance in managing food safety risks is the time it takes to respond to situations requiring a Class 1 recall. A Class 1 recall is carried out when there is a reasonable probability that the use of, or exposure to, a food product in violation of
standards will cause serious adverse health consequences or death. To determine this, the CFIA’s regulatory partners, in consultation with CFIA technical experts, provide the CFIA with a risk assessment. The CFIA uses the assessment as a basis in developing a risk management strategy, of which one option could be a Class 1 recall. Once an assessment has been received that
indicates there is a risk to the public and Class 1 recall is warranted, the CFIA issues a public warning within 24 hours of the recall decision — the CFIA target for timeliness.In 2006–07, as in 2005–06, the Agency met the target 100% of the time.
One of the key measures that the CFIA uses to assess its performance in managing food safety risks is the time it takes to respond to situations requiring a Class 1 recall. A Class 1 recall is carried out when there is a reasonable probability that the use of, or exposure to, a food product in violation of
standards will cause serious adverse health consequences or death. To determine this, the CFIA’s regulatory partners, in consultation with CFIA technical experts, provide the CFIA with a risk assessment. The CFIA uses the assessment as a basis in developing a risk management strategy, of which one option could be a Class 1 recall. Once an assessment has been received that
indicates there is a risk to the public and Class 1 recall is warranted, the CFIA issues a public warning within 24 hours of the recall decision — the CFIA target for timeliness.In 2006–07, as in 2005–06, the Agency met the target 100% of the time.
|
|
2005–06 |
2006–07 |
|
Food Safety Investigations |
2,675 |
2,915 |
|
Food Recall Incidents |
259 |
246 |
In 2006–07, there were 2,915 food safety investigations conducted with 246 incidents resulting in recalls, a decrease from the 259 recalls issued in 2005–06.6 The lower number of recalls is attributable to the fact that most investigations determined that the vast majority of food safety incidents had not put the public at risk.
An initial recall situation (primary recall) may lead to additional recalls (secondary recall) related to the original issue. Common factors between primary and secondary recalls may include product safety concerns or process deviations. For example, an out-of-country manufacturer recalls a product that was shipped to multiple importers. The spinach recall in the fall of 2006 is an example that involved one packer in the U.S. that shipped its products to 58 importers in Canada.
There were several high-profile recall incidents in 2006–07. They were:
- Clostridium botulinum in imported carrot juice
- E. Coli 0157:H7 in fresh spinach (U.S. outbreak) Tampering with infant formula, processed meat products, oranges, fresh turkey and cookies
- Salmonella in chocolate products
- Salmonella on imported cantaloupes, spinach and peanut butter
- Staphylococcus toxin in prepared cakes (Quebec outbreak)
Additional information on food recalls can be found on the CFIA’s website. This information is updated regularly. The CFIA also has a Food Recall List Service that advises our subscribers from the health industry and private sectors on food recall incidents.
The CFIA is involved in a number of public awareness initiatives to promote food safety. For example, the CFIA’s on-line subscription service sent updates to 22,417 subscribers on a variety of key food allergy concerns in 2006–07. There were 2,143,940 viewings of the food recall and allergy alert pages on the CFIA website. More than 252,500 CFIA-produced food safety information publications were distributed to the public through Service Canada sites and at more than 50 public events across the country.
Public opinion research conducted in October 2006 and in March 2007 suggests that the CFIA is meeting its objective of ensuring that the public is aware of food safety risks: 82% of Canadians said that they had heard about a food recall in the last year. The research also indicates a positive link between Canadians’ awareness of food recalls and their confidence in the food supply.
2.3.1b Program Sub-Activity: Controlling the transmission of animal diseases to humans
|
Table 2.3.1b.1 — Financial Resources: Controlling the transmission of animal diseases to humans* |
|||
|
Planned Spending |
Authorities |
Actual Spending |
Proportion of Actual Agency Spending |
|
70.3 |
77.1 |
95.6 |
15% |
|
* The Avian Influenza TB submission had a reference to include those resources as an item under the general Public Security and Anti-Terrorism Framework. Hence, the Planned Spending and Authorities figures for Preparing for Emergencies reflect the increased funding ($31 million and $28.8 million, respectively) related to Avian and Pandemic Preparedness. The Actual Spending figures were coded by program managers to “Protecting Canada’s Livestock” and “Controlling the transmission of animal diseases to humans”. In 2007–08 and future years, the allocation of the budget will be realigned accordingly. |
|||
Recent emergencies have brought to the forefront the relationship between animal and human health. The CFIA, in co-operation with its partners, carries out several programs and activities to help ensure zoonotic diseases (i.e. animal diseases), that are transmissible to humans either through contact or via the food chain, are controlled in animal populations. These programs focus on early detection, rapid response, and strong domestic and international co-ordination. The key strategies relating to this sub-activity include disease surveillance, testing activities, and control measures to mitigate the risk to animal, and indirectly to human health.
Of the $379.6 million the CFIA spent to achieve this Strategic Outcome, approximately $95.6 million was devoted to control the transmission of animal diseases to humans.
|
Expected Result: Animal diseases that are transmissible to humans are effectively controlled within animal populations |
To protect the health of Canadians, it is critical that the CFIA carry out timely and effective surveillance, detection and control activities for zoonotic diseases. When the presence of a reportable disease is confirmed in Canada, the CFIA minimizes the spread of infection by implementing disease-specific biosecurity measures, including quarantines and movement controls. When eradication activities are necessary, the CFIA ensures humane destruction and appropriate disposal of affected animals, thereby minimizing the risk that susceptible livestock are exposed to potential sources of infection.
National identification tagging program: This program provides individual animal identification and herd-of-origin trace-back for cattle and bison. Every bovine animal must be identified with an official tag before leaving the herd of origin or co-mingling with cattle of other owners. The program is managed by the Canadian Cattle Identification Agency (CCIA) and mandated under the Health of Animals Regulations. The CCIA and CFIA work together to achieve effective control of animal disease risks associated with foodborne illnesses.
![]() In 2006–07, the CFIA conducted inspections at feedlots, slaughterhouses and auctions to confirm compliance with the tagging regulations.7 Compliance has remained high, with the estimated compliance rate for individual animals at all site types at
99.1%. This exceeds the target of 97% and is consistent with the 2005–06 compliance rate of 99%.
In 2006–07, the CFIA conducted inspections at feedlots, slaughterhouses and auctions to confirm compliance with the tagging regulations.7 Compliance has remained high, with the estimated compliance rate for individual animals at all site types at
99.1%. This exceeds the target of 97% and is consistent with the 2005–06 compliance rate of 99%.
Highlight on AIAvian Influenza Since 2004, Canada has faced two outbreaks of AI in domestic poultry flocks, though neither of these outbreaks was of the highly pathogenic Asian h4N1 virus that has been linked to illness and death in humans in Asia, Africa, and Europe. The CFIA continues to develop effective response strategies should it confirm that AI, regardless of the strain, is present in domestic poultry. In February 2006, the CFIA established a dedicated working group to guide and oversee the five components of the Agency’s AI strategy: prevention and early detection; preparedness; partnerships; emergency response; and communications. Significant progress was made during 2006–07 in all five areas. Prevention and early detection: The CFIA announced stronger controls over imports of live birds to Canada and continued to promote industry adoption of best practices in farm biosecurity that are recognized as a key preventative measure in reducing the introduction and spread of infectious agents into animal production. The CFIA also worked with the poultry industry and provinces to design an AI surveillance program and contributed to Canada’s Wild Bird Avian Influenza Survey. Preparedness: The CFIA strengthened its surge capacity to respond to outbreaks by creating staffing reserves, emergency response equipment stockpiles, and enhancing its information systems. The CFIA reviewed emergency response activation protocols, developing a consistent national approach, and fully implementing Level 5 protocols as of December 2006. In particular, the following achievements were made: the CFIA National Emergency Response Plan was instituted; the National Emergency Response Team was formalized; and a number of Avian Influenza Emergency Preparedness exercises were delivered. In January 2007, the Canadian Veterinary Reserve was established to identify available private sector veterinarians to help respond to animal health emergencies. The CFIA also partnered with provincial and university laboratories to improve domestic surveillance capability and early detection of AI by establishing a National Avian Influenza Laboratory Network for rapid testing and reporting. Partnerships: The CFIA worked with provincial governments to update the joint Foreign Animal Disease Emergency Support (FADES) agreements and led the development of the Zoonotic Illness Outbreak Response Protocol to ensure a co-ordinated federal government response in the event of zoonotic disease outbreaks. The CFIA also worked with the World Organisation for Animal Health to develop new international biosecurity guidelines for raising, handling and transporting influenza-susceptible animals. Emergency response: In 2006–07, the CFIA developed the National Emergency Response Plan and other emergency response plans to address an outbreak of highly pathogenic AI in domestic poultry. An AI National Emergency Response Team was pre-identified, consisting of key personnel familiar with Incident Command Systems. The Agency also worked with federally registered food processing establishments to enhance preparedness of this sector, developing food safety guidelines and movement procedures for poultry products in the event of an outbreak. Several additional key plans were developed for responding to foreign animal disease outbreaks such as the Animal Health Functional Plan and the Notifiable Avian Influenza Hazard Specific Plan. Emergency procedural documents relating to humane destruction of birds, disposal, cleaning and decontamination were also prepared. In addition, the CFIA initiated procurement of 10 million doses of poultry vaccine to be stored at key locations in Canada, should traditional disease control measures be overwhelmed. Communications: Regular AI updates and a notification service were introduced on the CFIA’s website. A travellers’ campaign and a biosecurity campaign were launched to address two of the most likely pathways for the introduction of highly pathogenic AI into Canada. Activities for the Travellers’ Biosecurity Campaign included advertising and distributing information materials through travel agents, airlines and at international points of entry. Information materials, including a calendar, brochures and posters were developed for the biosecurity campaign and distributed to small flock owners through feed producers and hatcheries. |
Highlight on BSE8Bovine Spongiform Encephalopathy Controlling BSE disease is critical — for animal and public health, domestic and international confidence in the integrity of Canada’s food safety programs, and for the economy. BSE became a reportable disease in 1990 and an active surveillance program for the disease was implemented in 1992. The provinces, industry, universities and private-sector veterinarians have collaborated with the CFIA in BSE surveillance and testing work. In 2006–07, the CFIA carried out a number of activities to strengthen Canada’s scientific and policy response to BSE in order to improve detection, evaluate the effectiveness of measures in place, and provide the foundation for maintaining consumer and international confidence in Canadian animals and animal products. The CFIA also devoted much effort to developing appropriate indicators to track performance, and to building systems needed to collect performance information and to report on the results of its BSE programs. The CFIA’s enhanced BSE programs are grouped into four program areas: surveillance and testing; enhanced tracking and tracing; removing specified risk material from the food chain; and re-opening international markets. Surveillance and testing:9 Since its inception and implementation in 1992, the design of the national BSE surveillance program has been based on internationally recognized risk factors and delivered through the collaborative efforts of federal and provincial governments, universities and private veterinary practitioners. The program has been developed in accordance with international standards and reflects the demographics of the Canadian cattle population. Historically, the level of BSE testing has exceeded the international guidelines recommended by the Office International des Épizooties (OIE),10 the World Organisation for Animal Health, and was appropriate to a country with no cases of BSE. In this context, the level of testing conducted was designed to satisfy a single objective: to determine whether BSE was present in Canada. In 2003, the national BSE surveillance program confirmed BSE in a cow indigenous to Canada. This sentinel event signalled unknown prevalence of the disease and precipitated a significant increase in surveillance testing to determine the level of BSE within the national cattle population. On January 9, 2004, the government announced that BSE surveillance testing would be increased to include as many high-risk animals as possible, targeting a minimum of 8,000 samples during 2004, and a minimum of 30,000 samples in subsequent years. The level and design of this enhanced program continue to be in full accordance with the guidelines recommended by the OIE. Under the program, the high-risk cattle population that is targeted for evaluation reflects the demographics and distribution of the entire cattle population in Canada. From January 1, 2006, to December 31, 2006, 55,420 samples were evaluated by a network of federal, provincial and university laboratories. Since the enhanced BSE surveillance program was implemented in January 2004, over 136,700 samples have been evaluated for this disease. This illustrates the CFIA’s success in scaling up its surveillance testing program as well as the high degree of support the national BSE surveillance program receives from all levels of government and from producers, private veterinarians and industry stakeholders. The result of the national BSE surveillance program during 2006–07 was the detection of five cases of BSE as compared with one case in 2005–06. In all instances, the CFIA conducted a comprehensive animal and feed investigation which, in accordance with international science-based guidelines, resulted in the identification and removal of animals of equivalent risk to the BSEaffected cattle. The detection of these cases did not change any of Canada’s BSE risk parameters and was wholly consistent with the experience of other BSE-affected countries. The locations and ages of the animals involved were consistent with previous cases detected in Canada and in this context did not contribute any new information with respect to the broader international understanding of this disease. The BSE surveillance results continue to reflect an extremely low level of BSE in Canada. Based on these results for 2006–07, the annual incidence rate of BSE was determined to be 0.795 cases per million animals over two years of age as compared to 0.0145 for 2005–06. However, changes to international guidelines, implemented in 2006, have diminished the significance of this calculation. In May 2006, the OIE member countries, of which there are 168, adopted revisions to existing guidelines. The revisions removed reference to disease incidence and established a process to recognize countries as meeting the criteria of one of three possible categories with respect to BSE risk. These changes correctly emphasize a country’s response to BSE as more important than the incidence of disease, and in this context, that safe trade between BSE-affected countries can be realized based on effective risk mitigation. In May 2007, Canada received confirmation that it has been categorized by the OIE as a controlled BSE-risk country, reflecting the effectiveness of Canada’s surveillance, mitigation and eradication measures, and the efforts of all stakeholders. Enhanced Tracking and Tracing Program: The CFIA is an active participant in the development of the National Agriculture and Food Traceability System (NAFTS) starting with livestock (prioritized species are cattle, sheep and hogs) and poultry. Tracking the movement of cattle is an essential step in the control and eradication of animal disease, and in preventing the transmission of animal disease to humans. Since 2004, there has been a mandatory identification system for cattle in Canada under the Health of Animals regulation. The CFIA monitors and enforces compliance to the criteria as defined in the regulation at the point of entry into the food chain. Removing “Specified Risk Material” (SRM) from the food chain: Material from particular tissues, such as the brain, spinal cord or small intestine can harbour the BSE agent. The most effective food safety measure to protect humans from BSE exposure is to remove this material when animals are slaughtered. The CFIA monitors inspections of all federally registered establishments to confirm the removal of Specified Risk Material during slaughtering.11 Failure to comply can lead to the suspension and/or cancellation of the facility’s licence to operate.
Re-opening international markets: Demonstration of the overall integrity of Canada’s inspection controls is the foundation for trading partners to provide market access to Canadian animals and animal products. Since the identification of the first case of BSE in 2003, 24 markets that had imposed restrictions on Canadian beef exports were subsequently lifted either in full or in part by March 31, 2007. As of March 31, 2007, the CFIA had made significant progress in implementing the AIand BSE-related recommendations. For further information on this progress, please refer to the relevant discussions under Strategy: Disease surveillance and eradication activities. Further recommendations will be implemented in 2007–08 and reported upon in the 2007–08 Performance Report. |
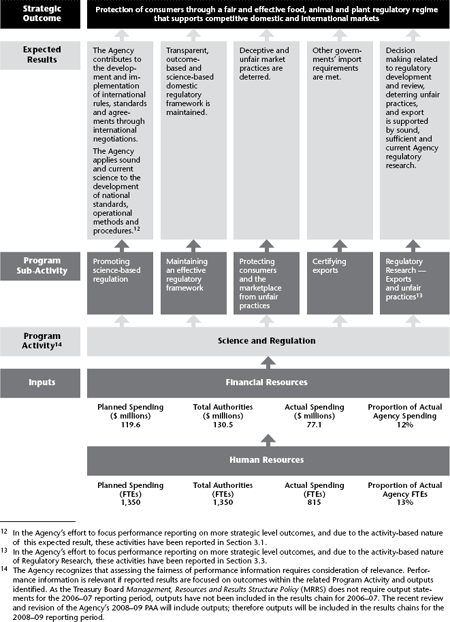
2.3.2 Strategic Outcome: Protection of consumers through a fair and effective food, animal and plant regulatory regime that supports competitive domestic and international markets
|
|
A fair and effective regulatory regime for food safety, animal health and plant protection is critical to consumer confidence and to Canada’s economy. It contributes to a competitive marketplace and protects consumers from unfair practices. It also helps to facilitate the access of Canadian products to foreign markets, thereby maintaining or expanding growth in international trade. As the primary federal regulator of food, animals, plants and related products, the CFIA is committed to promoting a regulatory regime that is fair and effective.
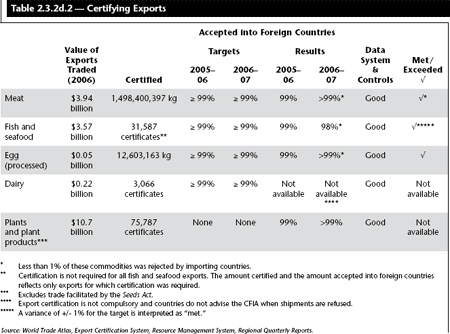
Prior to export, international convention requires the certification of quality, safety and other related standards by a national-level, competent public authority for many commodities including fish, meat, animals, and plant products. The CFIA is Canada’s regulatory authority for providing such certification. The CFIA works collaboratively with Health Canada and Foreign Affairs and International Trade Canada, among others, to fulfill this responsibility.
The activities related to achieving this Strategic Outcome work to enable all of the strategic outcomes under CFIA’s responsibility. The activities also contribute to strong international, science-based regulations, and are designed to mitigate the risks associated with failing to maintain and update the domestic legislative framework in Canada.
In 2006–07, the CFIA spent approximately 12% of its budget to achieve this Strategic Outcome.
2.3.2a Program Sub-Activity: Promoting science-based regulation
|
Table 2.3.2a.1 — Financial Resources: Promoting science-based regulation |
|||
|
Planned Spending |
Authorities |
Actual Spending |
Proportion of Actual Agency Spending |
|
51.0 |
55.8 |
16.8 |
3% |
The activities related to this sub-activity are drivers for the development of national and international science-based standards, operational methods and procedures. These activities are critical to expanding Canada’s access to global markets because they influence the development of international standards related to food safety and consumer protection, animal health and plant protection. These activities are also directly linked to the government’s priorities for public health, economic growth, environmental protection and public security.
Of the $77.1 million the CFIA spent to achieve this Strategic Outcome, approximately $16.8 million was devoted to promoting science-based regulation.
|
Expected Result: The Agency contributes to the development and implementation of international rules, standards and agreements through international negotiations15 |
Canadians benefit from safe food, a secure plant and animal resource base, and a protected environment. Science-based rules that are applied in a predictable, transparent and non-discriminatory manner on both domestic and international levels help to achieve these benefits for Canadians. The CFIA works bilaterally and multilaterally with a number of international partners to remain at the forefront of scientific developments and to advance sound, science-based decisions and policies at the international level.
In 2006–07, the Agency made significant contributions to the development of international rules and standards and advanced a number of bilateral issues in the following areas:
World Trade Organization — Sanitary and Phytosanitary Measures
The World Trade Organization (WTO) deals with the rules of trade between nations at a near-global level. Within the WTO there is an agreement on Sanitary and Phytosanitary or SPS Measures, which outlines how governments can apply food safety and animal health and plant health (SPS) measures on trade. The CFIA is the Government of Canada lead for the World Trade Organization Agreement on the Application of Sanitary and Phytosanitary Measures (SPS Agreement). An SPS Measure is any measure applied:
|
To protect animal or plant life or health, within the territory of the member, for risks arising from entry, establishment or spread of pests, diseases, disease-carrying organisms or disease-causing organisms; To protect human or animal life or health, within the territory of the member, from risks arising for additives, contaminants, toxins or disease-causing organisms in foods, beverages or feedstuffs; To protect human life or health within the territory of the member from risks arising from diseases carried by animals, plants or products thereof, or from the entry, establishment or spread of pests; or To prevent or limit other damage within the territory of the member, from the entry, establishment or spread of pests. |
The CFIA co-ordinates the implementation of SPS Measures to ensure they are consistent with Canada’s obligations under the WTO, including implementation by other federal departments and agencies, provincial, and regional bodies. In 2006–07, the CFIA led Canada’s participation at the three WTO SPS Committee meetings. The Committee is the key forum for discussing implementation of the WTO SPS Agreement and for raising SPS-related trade concerns in a multilateral environment.
World Organisation for Animal Health (OIE)
The OIE is an organization whose main objectives are to ensure transparency in the global status of animal disease and zoonotics and, through a number of activities, to safeguard world trade by publishing health standards for international trade in animals and animal products.
As a member country, Canada is a key player in OIE standard-setting processes and has access to early notification of animal disease outbreaks that may affect trade.
Canadian experts, as members of OIE ad hoc groups, have helped set standards in areas such as epidemiology. Twice a year, through the Office of the Chief Veterinary Officer, the CFIA undertakes an inclusive consultation process on new and/or revised standards developed by the OIE. The consultative process involves several federal departments and agencies, provincial jurisdictions, national industry associations and non-governmental organizations that prepare and submit comments on the standards.
Canada also works internationally with a number of OIE-member countries to build consensus for the adoption of standards at the annual General Session of the OIE. The CFIA leads Canada’s participation at the annual General Session of the International Committee of the OIE every May. The General Session provides for the adoption of new or revised standards published in the Terrestrial and Aquatic Animal Health Codes, and as such, concludes the annual cycle of development of international science-based standards for animal, public and eco-system health and for the safe trade of animals and animal products in international commerce. Canada currently serves as an elected member of the OIE Administrative Commission for 2006–09.
Codex Alimentarius Commission
Codex is an international standard-setting organization created by the Food & Agriculture Organization (FAO) of the United Nations and the World Health Organization (WHO), whose mandate is to develop food standards to protect the health of consumers and to facilitate fair practices in international food trade. Codex standards, codes of practice and guidelines serve as the WTO–SPS Agreement reference point for food safety. Through its participation in Codex, the CFIA influences the development of international standards and related texts to reflect Canadian objectives for safe food and fair trade practices, and to ensure adopted standards are based on sound science and result in a fair and effective international regulatory framework for food. The CFIA serves as the Chair of the Codex Committee on Food Labelling and, with other departments, supports hosting committee meetings in Canada.
International Plant Protection Convention (IPPC)
The IPPC is an international treaty to secure action to prevent the introduction and spread of pests of plants products and to promote appropriate measures for their control. Through proactive involvement in this international treaty, the CFIA has contributed to the development of several international standards for Phytosanitary Measures. These standards facilitate exports of Canadian plants and plant products, while improving risk management related to imports to Canada. Canada currently holds the vice chair position of the Bureau to the Commission of Phytosanitary Measures, which governs the International Plant Protection Convention.
North American Plant Protection Organization (NAPPO)
NAPPO provides a continental approach to plant protection by affording a means of sharing information and furthering common goals in plant health activities. The CFIA is actively engaged in the development of North American regional standards, such as standards for the certification of commercial arthropod biological control agents, and for the import and confined release of transgenic arthropods. The CFIA is also leading the development of guidelines for the screening of potential invasive plants prior to import.
Organisation for Economic Co-operation and Development (OECD) Seed Schemes
For the past two years, the CFIA has chaired the OECD seed schemes, which certify seed varietals being traded internationally to ensure consistently high-quality seed. The CFIA has also contributed to the development of international standards and programs for seeds, such as standards for canola.
2.3.2b Program Sub-Activity: Maintaining an effective regulatory framework
The key strategies related to this sub-activity include the development of regulations to maintain an effective regulatory framework that supports consumer protection and competitive domestic and international markets.
Of the $77.1 million the Agency spent to achieve this Strategic Outcome, $23.8 million was devoted to maintain an effective regulatory framework.
|
Expected Result: Transparent, rules-based and science-based domestic regulatory framework is maintained16 |
The CFIA made progress toward modernizing and strengthening its internal processes for developing policy and legislation specific to mandated activities in food safety and plant and animal health. These improvements are expected to result in more fair, efficient and responsive regulatory activities. More specifically, the CFIA will continue to implement and refine a new issue identification and streaming process which will include a thorough assessment of instrument options and early and ongoing consultation with stakeholders and partners. The process consists of several stages of committee review and affords various opportunities for challenge, at increasing levels of responsibility. The process will be assessed and improved on an ongoing basis, as opportunities for strengthening and/or streamlining are identified.
In addition, a new regulatory plan was developed and approved in the Fall of 2006. This plan positions the regulatory priorities in the context of policy priorities and resulted in adjusting the priority of some regulatory packages. Furthermore, in the interest of addressing the need for thorough consultation, openness and transparency, a number of regulatory amendments were subjected to additional consultation, including: Medicated Feeds, Humane Transport of Animals, Fresh Fruit and Vegetables — Licensing and Arbitration, and Seed Streaming and Variety Registration. In addition, there was a need to address a number of new priorities such as Golden Nematode, Sudden Oak Death, and Cheese Compositional Standards. It is expected that the outstanding priorities from 2006–07 will be moved forward to the Canada Gazette, Part I during 2007–08.
Legislative initiatives
Although there were no CFIA-specific legislative initiatives during 2006–07, work continued on the elaboration of options for a legislative strategy. CFIA officials worked closely with officials from Health Canada on the Health Canada Discussion Document entitled: Towards a Regulatory Modernization Strategy for Food and Nutrition. CFIA officials also worked closely with officials from other government departments in managing a number of Private Members’ Bills with implications for the CFIA, notably: S-213 (An Act to Amend the Criminal Code — cruelty to animals) led by the Department of Justice; S-205 (An Act to Amend the Food and Drugs Act — safe drinking water), led by Health Canada; and, S-283 (An Act to Amend the Food and Drugs Act [food labelling]) led by Health Canada.
Regulatory initiatives
In 2006–07, the CFIA had more than 60 proposed regulatory packages under development, spanning all Agency programs. In addition, the following CFIA regulations were published in the Canada Gazette, Part I and/or II, during this time frame:
- Regulations Amending the Plant Protection Regulations (Electronic Documentation)
- Regulations Amending the Health of Animals Regulations and the Regulations Amending Certain Regulations Administered and Enforced by the Canadian Food Inspection Agency (import requirements)
- Regulations Amending the Fish Inspection Regulations (Salted fish and other provisions)
- Organic Products Regulations
- Golden Nematode Compensation Regulations
- Regulations Amending Certain Regulations Administered and Enforced by the CFIA (Miscellaneous Program — Standing Joint Committee for the Scrutiny of Regulations amendments)
- Regulations Amending the Egg Regulations (HACCP and other requirements)
- Regulations Amending the Licensing and Arbitration Regulations (Exemption provisions)
- Certain Ruminants and Their Products Importation Prohibition, No. 2 (extension)
- Regulations Amending Certain Regulations Administered and Enforced by the Canadian Food Inspection Agency (Feedban extension)
- Phytophthora Ramorum Compensation Regulations (Sudden Oak Death)
- Regulations Amending the Introduced Forest Pest Compensation Regulations
- Regulations Amending the Livestock and Poultry Carcass Grading Regulations (Bison Grading)
- Regulations Amending the Compensation for Destroyed Animals Regulations
- Regulations Amending the Seeds Regulations (Seed Standards)
- Anthrax Compensation Terms and Conditions, No. 2.
CFIA will continue to ensure that regulatory infrastructures are developed to support consumer protection as well as competitive, domestic and international markets. Key regulatory initiatives undertaken during 2006–07 of particular importance to Canadians are discussed below.

Organic Products Regulations
The Organic Products Regulations established a system by which the CFIA, as competent authority, regulates the use of the “Canada Organic” agricultural product legend. The CFIA’s partner, Agriculture and Agri-Food Canada, establishes organic standards that provide the basis for organic regulations and also plays an important role in negotiating organic equivalency standards with the European Commission.
These regulations are built on the existing system of domestic accreditation and certification bodies to provide credibility and a basis for evaluation. The introduced regulations govern the use of a new Canada Organic logo, including certification requirements, for organic agricultural products. The regulatory framework includes a mandatory organic production standard, a certification and inspection regime as well as import requirements. The Organic Production Systems General Principles and Management Standards and Organic Production Systems Permitted Substances Lists developed by the Canadian General Standards Board form the basis of the regime.
Compensation for Destroyed Animals Regulations
Compensation for Destroyed Animals Regulations were published in 2000, replacing the Maximum Amounts for Destroyed Animals Regulations, 1992. The maximum amounts for each animal that are listed in the schedule of the Regulations have not been substantively amended since 2000 and, as a result, the list of animals and their maximum amounts no longer reflect the Canadian animal industry profile or the current animal market values. These amendments update the list of regulated animals and adjust the maximum amounts.
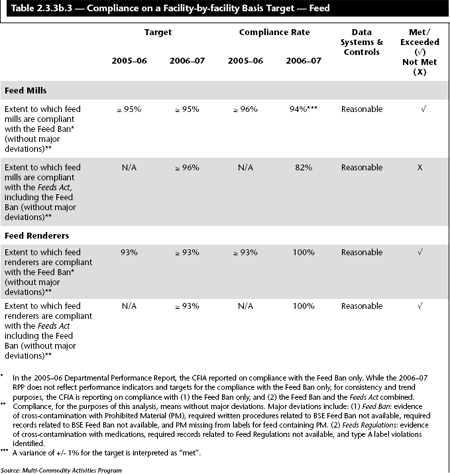
Enhancing the 1997 Feed Ban
In 2006–07, the CFIA published regulatory amendments to enhance the 1997 Feed Ban through the introduction of new requirements for the removal of Specified Risk Material from all animal feeds and fertilizers. These regulations prohibit rendering, feed production, and distribution sectors from feeding most mammalian proteins to ruminant animals, such as cattle, sheep and goats. The regulations also require these sectors to follow and document production and feeding procedures. As the regulations entered into force in July 2007, the first reporting on compliance with these new requirements will occur in the 2007–08 Performance Report.
Meat Inspection Reform
Canada’s Meat Inspection Reform is about modernizing Canada’s meat inspection system. The key objectives include: maintaining the safety and suitability of Canadian meat and meat products; sustaining consumer confidence; expanding market access; providing a sound regulatory base for industry, contributing to competitiveness; and, delivering meat inspection programs in the most efficient and effective manner possible. Meat Inspection Reform consists of changes to existing federal inspection programs and the development of a Canadian Meat Hygiene Standard.
The CFIA’s efforts to reform inspection activities in federally registered establishments include streamlining poultry inspection under the Modernized Poultry Inspection Program and the Poultry Rejection Project; red meat inspection under the HACCP-based Inspection Program; meat processing inspection under the Compliance Verification System; the opening (registration and licensing) of new establishments; the approval process of labels for meat products; and the issuance of electronic documents for import and export.
The Modernized Poultry Inspection Program (MPIP) was the CFIA’s first inspection-reform initiative. It incorporates HACCP-based principles and objective outcome measures into inspection methodology. Three outcome based performance standards were developed to assess compliance to MPIP. The standards are based on science and designed to identify and control potential food safety hazards throughout the process of preliminary poultry production. In 2006–07, the status of the program implementation in identified targeted establishments was 82.5% (33/40 federally registered poultry establishments). The Poultry Rejection Project builds on the HACCP foundation of the MPIP and enhances veterinary oversight of animal welfare, control of food safety hazards, and early detection of foreign animal diseases. The Compliance Verification System integrates HACCP Audit approaches with pre-existing inspection tools to provide an improved and seamless interface between the CFIA and its regulated parties.
The Canadian Meat Hygiene Standard and accompanying Technical Guidance Documents set out the legal and technical requirements to ensure the production of safe and suitable meat and meat products. They could be used as a foundation for provincial governments when amending their own meat inspections systems and will serve as a benchmark for the food safety requirements in federal meat inspection.
2.3.2c Program Sub-Activity: Protecting consumers and the marketplace from unfair practices
Under this sub-activity, the CFIA carries out various strategies that are intended to deter deceptive and unfair market practices. These include enforcing standards relating to labelling composition, net quantity and advertising, and how information is presented on the labels of food products. CFIA’s strategies under this subactivity also extend to promoting compliance with the Seeds Act, granting plant breeders’ rights, administering licensing and providing non-biased inspections for buyers and sellers of fresh fruits and vegetables.
Of the $77.1 million the Agency spent to achieve this Strategic Outcome, approximately $16.7 million was devoted to protecting consumers and the marketplace from unfair practices.
|
Expected Result: Deceptive and unfair market practices are deterred |
Fair labelling practices17
The Fair Labelling Practices Program protects consumers from deceptive practices (i.e. unfairly and inaccurately presenting net weight and contents through labelling) and facilitates fair competition for industry by verifying compliance with the net quantity, composition, labelling and advertising provisions of the Food and Drug Regulations and the Consumer Packaging and Labelling Regulations for both domestically produced and imported food products. This regulatory program complements similar programs in the registered sectors (i.e. meat, dairy, fish and seafood) by protecting Canadians from unfair market practices in the non-federally registered sector.
Through its technical committee process, the CFIA identifies and prioritizes potential deceptive labelling practices in the marketplace, and develops strategies to direct inspection and laboratory resources towards products and establishments that are determined as posing the greatest non-compliance risk to consumers.
The CFIA promotes compliance by conducting trader education regarding regulatory requirements, by investigating consumer and trade complaints, by inspecting and analyzing food products at the manufacturing, retail and import levels of trade, and by taking effective compliance action.
During 2006–07, 2,646 inspections of food products resulted in the identification of 12,386 violations or an average of five violations per inspection. The average of five violations per inspection represents a full spectrum of deceptive and unfair market practice situations such as net quantity, composition, adulteration, absence of mandatory label information, nutrition labelling, bilingualism, or misleading claims. This is consistent with the previous four year average of 12,150 violations. All violations resulted in appropriate enforcement action up to, and including, prosecution. Examples are detailed below.
|
In May 2006, a company was fined $14,000 for failing to include whey powder in the list of ingredients on the label of a food product. The failure to identify whey powder can be a significant omission as milk-based foods are a significant food allergen for a number of Canadians. The Food and Drug Regulations require that, with certain exceptions, prepackaged multi-ingredient foods carry a list of ingredients and their components, if any, in descending order of proportion by weight of the product or, alternatively, as a percentage of the product. In November 2006, a company was fined $4,000 for selling pre-packaged food products whose labels failed to declare the net quantity as required by the Consumer Packaging and Labelling Act. |
The Fair Labelling Practices Program is also responsible for designing programs and tools to facilitate compliance. For example, during 2006–07, activities to implement Canada’s amendments to the Food and Drug Regulations concerning nutrition labelling, nutrient content claims and diet-related health claims included: further development of inspection tools and tools to assist the industry in implementing these new regulations; training of industry and the CFIA’s staff; and responding to many stakeholder inquiries. These activities will continue during 2007–08.
In addition to its routine compliance and enforcement efforts, the CFIA carried out a number of risk-based targeted projects (for example a retail survey) that were designed to focus inspection attention on specific commodities and compliance issues on a cyclical or sector-by-sector basis. Because targeted sampling, by definition, seeks out problem areas, the resulting compliance rates are not indicative of marketplace compliance in general, but do indicate that there is a segment of the industry not fully complying with the regulations. Examples are detailed below.
|
Following this sector-by-sector approach, inspection attention during 2006–07 was focused on both domestically produced and imported bakery and cereal products. Inspections of 209 companies resulted in the identification of 1,109 violations of the Food and Drug Regulations and the Consumer Packaging and Labelling Act and Regulations. Identified violations included: misrepresentation due to inaccurate nutrition declarations (e.g., trans fatty acid, saturated fatty acid, sodium); the presence of common food allergens not declared in the list of ingredients; and misleading highlighted ingredient and flavour claims. The results of these inspections are currently undergoing in-depth analysis. Strategies and recommendations to promote compliance across the sector will then be developed and shared with the sector. Based on a risk-based targeted inspection approach, another project focused on olive oil. CFIA testing detected an increase in the adulteration of olive oil over the previous fiscal years. The rate of compliance in 2006–07 was 67%, compared to 83% last year and an average of 94% for the three preceding years (see chart below). While the year’s compliance rate is not indicative of marketplace compliance in general because the sampling was targeted at suspected problems, it does indicate that a significant volume of olive oil had been adulterated with cheaper oils such as sunflower or canola oil, or with oils derived from olive seeds rather than that from the flesh of the olive. In 2006–07, the CFIA took enforcement actions up to and including the prosecution of companies found to be in violation of regulatory standards. This is a fraudulent activity that is not welcomed by consumers and results in the properties and flavours of the oils being different than expected. One enforcement action involved fining a company $4,000 for selling extra virgin olive oil in a manner that was false or misleading. |
|
Year |
# Brands Sampled |
# Brands Showing Adulteration |
% Compliance* |
|
2006–07 |
45 |
15 |
66.7 |
|
2005–06 |
42 |
7 |
83.3 |
|
2004–05 |
64 |
5 |
92.0 |
|
2003–04 |
53 |
3 |
94.3 |
|
2002–03 |
49 |
2 |
95.9 |
|
* Since sampling is directed toward suspected problems, the above data are not indicative of marketplace compliance in general. |
|||
The CFIA has legislated criminal law authorities to conduct investigations and enforcement activities under the Consumer Packaging and Labelling Act and the Food and Drugs Act. In 2006–07, the Agency investigated six instances of major non-compliance. Combined with investigations carried over from previous reporting periods, investigations resulted in 18 convictions and violators were fined a total of $12,000, which conveys the message that the CFIA is committed to ensuring consumer protection.
Seed
Under the Seeds Act, the CFIA regulates imported and domestic seed, certifies seed exports and registers seed varieties and seed establishments. The CFIA operates two seed laboratories that provide scientific advice and test for seed germination, varietal and mechanical purity, and seedborne diseases. The CFIA also works with the Canadian Seed Institute and the Canadian Seed Growers’ Association to maintain systems for managing seed quality in Canada. These systems focus on ensuring that seeds have not been contaminated by weeds or other plants, and that what ultimately grows corresponds with what is in the bag or bulk shipment and on the label.
Other partners also help throughout the process. The Canadian Seed Institute assesses seed establishments to ensure quality standards are maintained. The Canadian Seed Growers’ Association monitors and certifies pedigreed seed for all agricultural crops, except seed potatoes. Based on the CFIA seed crop inspection reports, the Association also issues crop certificates, which indicate compliance with varietal purity standards and pedigreed seed-crop inspection procedures.
In 2006–07, the Canadian Seed Institute carried out 306 quality assessments of seed facilities from a total of 1,240 establishments, including registered seed establishments, authorized importers and private labs to confirm that they were meeting the Institute’s quality standards. This number of assessments represents a significant increase over the 205 assessments conducted in 2005–06. Results indicate that 99% of the assessed facilities had no critical deficiencies, compared with 72% in 2005–06.
Inspectors for the CFIA also conducted marketplace surveillance for both pedigreed and nonpedigreed seed, and targeted establishments with poor compliance records and those subject to complaints from seed buyers. During the fiscal year, the CFIA laboratories conducted 8,666 such tests on 6,420 samples.
![]() The target compliance rate for domestic pedigreed seed is 95% while the target compliance rate for domestic non-pedigreed seed is 85%.
The target compliance rate for domestic pedigreed seed is 95% while the target compliance rate for domestic non-pedigreed seed is 85%.
Testing carried out in 2006–07 marketplace monitoring indicated compliance rates of 93% for pedigreed seed18 and 88% for nonpedigreed seed.19 Though the target for domestic pedigreed seed was still not met in 2006–07, both compliance rates are up slightly from their respective compliance rates of 92% and 86% for testing completed in 2005–06. The 2006–07 testing also indicated a compliance rate of 96% for imported seed,20 down from 99% for testing completed in 2005–06.
The CFIA has begun discussions with the seed industry to address compliance shortfalls.
![]() During the 2006–07 reporting period, 4,076 seed growers produced more than 2,167 varieties of pedigreed seed. CFIA inspectors determined that 98%21 of these met the purity product and process standards of the Canadian Seed Growers’
Association, which confirms the maintenance of high-quality Canadian pedigreed seed. In 2005–06, the compliance rate was 99%.
During the 2006–07 reporting period, 4,076 seed growers produced more than 2,167 varieties of pedigreed seed. CFIA inspectors determined that 98%21 of these met the purity product and process standards of the Canadian Seed Growers’
Association, which confirms the maintenance of high-quality Canadian pedigreed seed. In 2005–06, the compliance rate was 99%.
In addition to the Canadian Seed Institute’s audit and verification activities, the CFIA took 308 actions in response to marketplace incidents of non-compliance or complaints. This included issuing 25 education/warning letters, 163 detentions (“stop sale” orders) and 79 refusals of entry into Canada. The CFIA also conducted 31 complaint inspections and 10 investigations. There were no referrals for prosecution in 2006–07 in response to instances of non-compliance.
Grant plant breeders rights
The CFIA grants exclusive rights to Canadian breeders for their new varieties, and pursuant to Section 78 of the Act, reports on the administration of the Plant Breeders’ Rights Act. The intent of this legislation is to: stimulate plant breeding in Canada through the protection of intellectual property rights; provide Canadian producers better access to foreign varieties of seed; and facilitate the protection of Canadian varieties in other countries.
Data are collected by calendar year under the Plant Breeders’ Rights Act. In 2006, the CFIA received 498 applications for plant breeders’ rights,22 and rights were granted to 304 plant varieties. The CFIA also renewed 1,262 varieties previously approved for grant of rights. The CFIA received $1,074,850 in revenue for its registration services.
2.3.2d Program Sub-Activity: Certifying exports
|
Planned Spending |
Authorities |
Actual Spending |
Proportion of Actual Agency Spending |
|
47.6 |
51.9 |
19.8 |
3% |
The key strategies relating to this sub-activity include maintaining good relations with foreign governments, associations and domestic industries based on science and supported by standards, as well as certifying that certain Canadian exports of food and food products as well as plants and animals and their related products meet the requirements of importing countries.
Of the $77.1 million the Agency spent to achieve this Strategic Outcome, approximately $19.8 million was devoted to certifying exports.
|
Expected Result: Other governments’ import requirements are met |
The CFIA inspects and certifies regulated commodities destined for international markets, confirming the sanitary and phytosanitary status and quality of the product exported. The proportion of certified products accepted into foreign countries is utilized as a measure of success and can be considered a measure of confidence in the Canadian food supply and the CFIA’s activities in food safety, plant and animal health (see Table 2.3.2d.2). Certification plays a crucial role in Canada’s ability to trade in international markets, as CFIA-regulated exports of food, plants, animals, and associated products were valued at $42.6 billion in 2006.
![]() The 2006–07 performance target for certifying exports is 99% or greater.
The 2006–07 performance target for certifying exports is 99% or greater.
![]() The CFIA met its established target for the meat and processed egg programs (see Table 2.3.2d.2). Factors other than health and safety violations were often to blame for the rejection of Canadian products by importing countries. For example, of the total products rejected, 4.1%23 of meat was rejected due to a labelling error, 14.5% due to contamination (e.g., extraneous materials), 15.7% due to “miscellaneous” reasons (e.g., damaged packaging), and the remaining 65.7% of rejections were due to administrative processing errors and incorrect shipping markings.
The CFIA met its established target for the meat and processed egg programs (see Table 2.3.2d.2). Factors other than health and safety violations were often to blame for the rejection of Canadian products by importing countries. For example, of the total products rejected, 4.1%23 of meat was rejected due to a labelling error, 14.5% due to contamination (e.g., extraneous materials), 15.7% due to “miscellaneous” reasons (e.g., damaged packaging), and the remaining 65.7% of rejections were due to administrative processing errors and incorrect shipping markings.
Certification of exports in the fresh fruit and vegetable and processed products programs is not mandatory. Any certification conducted by the Agency is done as a service to the industry and is rendered on a cost-recovery basis. Rejections for these commodities are not currently tracked, as foreign governments are not required to notify the Canadian government when products are rejected at their borders.
While rejection rates are only available for some CFIA-regulated commodities at this time, the CFIA is making progress on collecting more performance information for this activity and will expand reporting as the data become available.
Certification
The CFIA continues to meet increasing export demands by moving towards information technology-based systems certification programs while maintaining science-based standards and principles. In 2006–07, the Agency implemented two such certification programs: the Canadian Wood Packaging Certification Program and the Canadian Heat Treated Wood Products Certification Program. Both of these programs are system-based, and inspections are conducted by third parties and audited by the CFIA. Currently 435 facilities are registered under the Canadian Wood Packaging Certification Program and 485 under the Canadian Heat Treated Wood Products Certification Program. During this fiscal year, only one facility in each of these programs was found to be non-compliant with export requirements.
Negotiating market access
The CFIA also plays an important role in negotiating market access for Canadian agricultural products based on sound scientific information. For example, in August 2006, following detection of Golden Nematode in Quebec, extraordinary measures were undertaken by the CFIA to maintain access to markets for potatoes and horticultural products produced in Quebec and other provinces. Immediate delimitation of the infested areas through the collection and analysis of 35,564 soil samples, in addition to the declaration of the Golden Nematode Infested Places Order, allowed the Agency to effectively negotiate with the U.S. and other countries and allowed normalization of trade within eight weeks. In order to maintain market access, the CFIA also implemented a certification program for all Canadian seed potato exports to the U.S.
2.3.3 Strategic Outcome: A Sustainable plant and animal resource base
|
|
Canada’s social and economic well-being is closely linked to the health of its environment, which includes plants and animals. The promotion of a sustainable plant and animal resource base is the CFIA’s contribution to the protection of the environment, as well as the sustainability of the Canadian food supply. This entails protecting Canada’s crops, forests and livestock from regulated pests and diseases. It also includes preventing the introduction of substances into animal and plant production systems by way of animal feeds, seeds, fertilizers and supplements, or other pathways that could adversely affect human health or the environment.
To fulfill this Strategic Outcome, the CFIA works co-operatively with Agriculture and Agri-Food Canada, Natural Resources Canada (including the Canadian Forest Service), Environment Canada (including the Canadian Wildlife Service), the Canada Border Services Agency, as well as with provincial, territorial and municipal partners and stakeholders.
In 2006–07, the CFIA spent approximately 23% of its budget to achieve this Strategic Outcome.
2.3.3a Program Sub-Activity: Protecting Canada’s crops and forests
Of the $140.1 million the CFIA spent to achieve this Strategic Outcome, approximately $65.2 million was devoted to protecting Canada’s crops and forests.
The CFIA is responsible for protecting Canada’s crops and forests from pests and diseases, such as Emerald Ash Borer and Potato Wart. The Agency has detection and control strategies to identify, assess and control or eradicate pests and diseases. This includes strategies for preventing pests and diseases from entering Canada, spreading within Canada, and being exported to other countries.
An estimated 300 species of tree-feeding insects have entered North American forests over the past century as part of commercial shipments and/or individual travellers’ effects. In response, the CFIA has developed import policies and standards to help prevent pests and diseases from entering Canada at its borders and other points of entry. The CFIA’s prevention efforts are supported by the Canadian Border Services Agency (CBSA), which enforces the CFIA’s import policies and standards at Canada’s borders and other points of entry. Within Canada, the CFIA works to control or eradicate pests. Keeping Canadian plants and plant products disease and pest-free is also critical to ensuring the safety and quality of Canadian plant resources and to protecting our export markets.
The stated purpose of the Plant Protection Act is to prevent pests and diseases injurious to plants from being imported into Canada, from spreading within the country and from being exported out of it. The Act also provides for controlling and eradicating pests and diseases and for certifying the pest and disease-free status of plants and plant material. To encourage reporting of plant pests, regulations under the Plant Protection Act allow for compensation to producers for the destruction of plants and plant products due to a specified regulated pest or disease.
The CFIA certifies the pest and disease-free status of plants and plant material, and encourages the reporting of pests to the CFIA by compensating producers for any CFIA required destruction of infected products. For example, CFIA paid $661,322 in compensation26 to Ontario producers who reported the presence of Emerald Ash Borer in their ash trees.
|
Expected Result: Entry and domestic spread of regulated plant diseases and pests are controlled |
Ultimately, the CFIA’s goal is to mitigate the risk of entry of new regulated pests and plant diseases into Canada.
The CFIA undertakes a series of activities to mitigate the risk of imported plant pests and diseases. Importers who wish to bring plants and plant products into Canada must first obtain an import permit from the CFIA. Regulated commodities are examined by inspectors from the Canada Border Services Agency, and in some cases CFIA inspectors, to confirm that they comply with federal acts and regulations before they enter the country.
Inspections
In 2006–07, there were 13,003 inspections of regulated plant products being imported into Canada compared with 18,581 in 2005–06. Of these, 1,298 (approximately 10%) resulted in interventions,27 compared with 1,745 (approximately 9%) the previous year. Interventions include treatment orders, detentions, disposals, or refusals of entry for reasons including improper documentation and the presence of a regulated plant pest or disease. Significant year-to-year fluctuations in the number of inspections carried out is normal, as plant imports can fluctuate annually.
On-site verifications
To improve the effectiveness of its import program, and to enhance risk mitigation at origin, the CFIA increased on-site verifications of certification systems in the country of origin from five in 2005–06 to 10 in 2006–07. These activities help the Agency ensure that the certification systems of foreign countries are sufficiently stringent to generate a product that meets Canadian import requirements, ensuring protection of Canada’s plant base.
Three examples of on-site verification that took place during the 2006–07 reporting period are on-site verifications of Chinese fragrant pear production, grapevines from France and a systems verification of Enoki mushroomgrowing production practices in Taiwan. These examples are described below.
|
A CFIA audit of Chinese fragrant pear production revealed no major deficiencies, confirming that material originating from this program met Canadian standards in a consistent fashion. The CFIA tested 1,932 import samples of grapevines from France in 2006 in response to the Agency’s 2006 detection of the spread of two grapevine phytoplasmas in France. While no positive results were detected, several non-quarantine viruses were found, warranting further Agency monitoring. The CFIA also implemented additional treatment requirements for importation of grapevines from European sources. This included a mandatory hot water treatment and a follow-up field survey of European grapevines planted in Canada. The CFIA also conducted an on-site systems verification of Enoki mushroom-growing production practices in Taiwan, leading to Canada’s decision to allow the importation of this commodity from pre-approved sources in Taiwan. |
Restrictions
Another aspect of the Agency’s risk mitigation at origin is the implementation of restrictions when a pest has been identified in another country. For example, the CFIA placed restrictions on imports of potatoes, horticulture products and soil from Idaho as a result of a notification of Pale Cyst Nematode detection in April 2006. A scientific and technical review of the delimiting and containment strategies adopted in Idaho were then performed to assess if the phytosanitary risk measures adopted were sufficient to protect Canada from the introduction of Pale Cyst Nematode from Idaho. The United States Department of Agriculture and Animal and Plant Health Inspection Service (USDA–APHIS) and the CFIA have since agreed on a strategy for addressing Pale Cyst Nematode.
Pests and diseases
The globalized trade of plants and plant products makes it difficult for the CFIA to achieve absolute prevention of the entry of new pests and diseases into Canada.
![]() The current Agency performance target in this area is the absence of evidence that any new regulated plant pests and diseases have been detected in Canada over the last fiscal year. When the Agency confirms that the pest or disease has been detected in the country, and depending on the pest or disease, the
Agency responds quickly by investigating the risk posed to Canada’s plant resource base and by developing strategies for control and eradication, as appropriate.
The current Agency performance target in this area is the absence of evidence that any new regulated plant pests and diseases have been detected in Canada over the last fiscal year. When the Agency confirms that the pest or disease has been detected in the country, and depending on the pest or disease, the
Agency responds quickly by investigating the risk posed to Canada’s plant resource base and by developing strategies for control and eradication, as appropriate.
In 2006–07, the CFIA detected two new regulated pests28 in Canada: Chrysodeixis sp. and Bois noir phytoplasma, compared to four pests found in 2005–06. The Agency responded immediately to determine the extent of the introduction and to put control measures in place to prevent the spread of these pests. The following summarizes the CFIA’s actions in these two cases:
|
In August 2006, when chrysodeixis was discovered in greenhouses in Delta, British Columbia, the CFIA recognized the potential trade impacts of this organism on trade with the U.S. To mitigate the impacts, the Agency developed eradication protocols while establishing compliance agreements for impacted facilities. This allowed for continued shipping of host vegetables/fruit and transplants to the U.S., while mitigating the risk of spreading the pest. Negative trapping results confirmed the effective eradication of the pest. In September 2006, the Agency found one grapevine plant infected with Bois noir phytoplasma in a lot of 1,965 plants that had been imported from France and planted in B.C. In response, the entire lot was removed and destroyed prior to spring of 2007. The CFIA will be conducting confirmatory surveys over the next year to assess the effectiveness of these eradication measures. |
These instances are clear examples of the CFIA’s commitment and vigilance to ensuring that the entry and domestic spread of regulated plant diseases and pests are controlled.
Surveys
Various regions of Canada are surveyed routinely to detect foreign pests and diseases that may have entered this country, and to define the boundaries of any infestations. Some pest surveys are conducted in co-operation with other agencies. The Agency acts as a central repository for all data on regulated pests and diseases, regardless of which agencies are involved in carrying out the survey.
Pest surveys allow Canada to validate its claims of pest and disease-free status for certain areas, to detect any new pests, and to establish quarantine restricted zones to limit their spread. These surveys are also central to control and eradication programs as the survey data provide information needed for the CFIA to make decisions regarding further control measures.
![]() The CFIA 2006–07 target for pest survey is to complete 100% of planned pest surveys.
The CFIA 2006–07 target for pest survey is to complete 100% of planned pest surveys.
In 2006–07, the CFIA planned and delivered a total of 24 pest surveys29 for the fiscal year, meeting the Agency’s target of 100%. In 2005–06 the CFIA planned 52 pest surveys and conducted 60. Annual survey priorities are established in conjunction with the Plant Health Division commodity section according to policy needs. CFIA Plant Health Division, through policy development activities that identify key scientific, environmental, social and economic issues, developed risk-based needs assessments that identified the need for targeted and specific activities. Some of the issues focused on the requirements for surveys for pests in all commodity groups, such as: forestry; horticulture; potatoes and grains; and field crops. The largest surveys were conducted as part of the CFIA control or emergency response for Plum Pox Virus, Emerald Ash Borer, Asian Longhorned Beetle, Brown Spruce Longhorned Beetle, Sudden Oak Death and the newly discovered infestation of Pale Cyst Nematode in Quebec.
Controls
While it is impossible for the CFIA to control the spread of pests and diseases caused by natural mechanisms, such as wind or the movement of wildlife, the Agency can limit spread caused by humans through the movement of material such as logs, firewood or nursery stock from an infected area to a non-infected area.
![]() The target for control programs is to keep pests and diseases from spreading beyond quarantine zones or restricted areas.
The target for control programs is to keep pests and diseases from spreading beyond quarantine zones or restricted areas.
Of six identified high-priority plant pests and diseases that were part of the Agency’s surveys in 2006–07, the CFIA was successful in the control of three, or half of the cases. This is compared with 2005–06 when five identified highpriority plant and pest diseases were successfully controlled by the CFIA. The Agency’s actions in regards to each of the respective plant pests are detailed below.
|
Surveys conducted by the CFIA indicate that Potato Wart has not spread outside its quarantined area of central Prince Edward Island since 2005. Efforts to control the spread of Asian Longhorned Beetle have been successful; no newly infested trees were discovered outside the regulated area in 2006. Pest-mitigation activities continue in the Toronto area. Following the detection of new infestations of Emerald Ash Borer in June 2006, Ministerial Orders were put in place for Lambton and Elgin Counties to slow the spread of the pest in Ontario. Following the October 2006 detection of three infested trees, a quarantine zone was also established in London, Ontario. In 2006 there were 18 new Brown Spruce Longhorned Beetle finds outside the current regulated area in Nova Scotia. The CFIA has been working with industry to revise the regulated area in accordance with the new finds, which will be finalized before the flight season in 2007. The Agency is also working with its partners to develop more effective detection and control tools. In 2006, a survey for Apple Clearwing Moth in British Columbia indicated that the pest has spread to the British Columbia coast and interior fruit belt. Detections were also made in London, Ontario. The CFIA shared biological information with the U.S. Department of Agriculture and discussed further regulatory approaches to deal with this pest. The Agency will conduct additional surveys to determine the extent of distribution of this pest in Ontario in 2007. In 2006, as part of its seven-year program to eradicate Plum Pox Virus, the CFIA took samples from more than 940,000 susceptible trees in Canada. This resulted in the discovery of 610 Plum Pox Virus positive trees. To date, the Agency has eliminated the disease from five quarantine areas in Ontario and Nova Scotia with two areas in Ontario remaining to be actioned. In 2006–07, there was a minor expansion of one of the quarantine areas in Ontario; however, the overall trend continues to show lower virus levels inside the quarantine areas. |
Emergency response
The CFIA is committed to dealing with new pests and diseases in an efficient and timely manner. Three examples of the Agency’s rapid response to new pests and diseases in 2006–07 are presented below.
|
Phytophthora ranorum, the disease that causes Sudden Oak Death, was first detected in Canada in 2004. In 2006–07, the CFIA took samples from approximately 250 wholesale and retail nurseries across Canada that were either growers or importers of host plants to test for P. ramorum. Results indicated that the disease was detected at one wholesale and four retail nurseries in British Columbia. The disease has been eradicated from three retail nurseries and eradication efforts have been intensified in the two remaining nurseries. The Agency will continue to monitor for further signs of the disease. In 2006–07, the CFIA activated its Chrysanthemum White Rust Eradication Protocol to eradicate an infestation at a site in British Columbia. No further infestations were found. Following the detection of Golden Nematode in Quebec in August 2006, the Agency immediately undertook a major investigation and delimiting survey to determine the extent of the infestation and the steps to contain it. These efforts were successful in removing restrictions placed on certain agri-cultural products from most of Quebec. The CFIA is working with a committee of stakeholders and technical experts to identify a long-term management strategy to contain and mitigate the risk associated with Golden Nematode within the regulated area as well as explore viable business options for affected growers. |
|
Expected Result: Industry complies with federal acts and regulations regarding Canada’s crops and forests |
The CFIA verifies that domestic and imported fertilizer and supplemental products sold in Canada meet the required standards under the Fertilizers Act and Regulations. These products are also sampled by the Agency to: test their efficacy; confirm that product guarantees are met; and confirm that contamination does not exceed set maximums.
Bulk-blend fertilizer monitoring
![]() The target for the bulk-blend fertilizer monitoring program is 95% compliance.30
The target for the bulk-blend fertilizer monitoring program is 95% compliance.30
The Agency monitors approximately 1,179 bulkblend fertilizer facilities across Canada, from which it tests samples of fertilizer products to confirm that label guarantees for the levels of nitrogen, phosphorus and/or potassium fall within regulatory tolerances. In 2006–07, the CFIA inspections found that 78% of samples were in compliance as compared with 2005–06 when 82% of samples were found to be compliant.
There has been a slight decrease in the compliance rate for bulk-blend fertilizers; yet, this rating remains consistent with findings over the past five years, which ranged between 75% and 82%. The slight decrease may be attributed, in part, to the implementation of an adjusted sampling strategy, which aims for greater industry compliance in the long term and includes targeting higher risk facilities. The CFIA is currently in discussions with the industry-led Canadian Fertilizer Products Forum in an effort to identify root causes of non-compliance as well as options for bringing the product category back into compliance.
Pathogen, heavy metal and pesticide contamination testing
The CFIA regularly monitors fertilizer and supplement products for pathogens and heavy metal and pesticide contamination to help ensure the safety of the products for plants, animals, humans and the environment.
![]() In 2006–07, the target cumulative compliance rate for these testing programs was 95%.31 This compliance level is consistent with the 2005–06 result of 96%.
In 2006–07, the target cumulative compliance rate for these testing programs was 95%.31 This compliance level is consistent with the 2005–06 result of 96%.
Fertilizer-pesticide guarantee monitoring
Pesticide guarantees in fertilizer-pesticide products are monitored by the CFIA for their compliance with the tolerances set forth in the fertilizers regulations. In 2006–07, the Agency found that only 69% of samples tested were in compliance. The majority of non-compliant samples contained less than the amount of active pesticide ingredient indicated on the label and, as such, are considered non-compliant from an efficacy perspective. While low, the compliance rate of 69% represents an 8% improvement compared with sampling results in 2005–06, an increase that has been attributed to the redesign of the CFIA guarantee monitoring program. Further improvement is expected in upcoming years.
In 2006–07, the CFIA investigated 86 instances of fertilizer-pesticide non-compliance. Combined with investigations carried over from previous reporting periods, these investigations resulted in no convictions or fines. This is attributable to a number of factors including: investigations/prosecutions/cases are still pending; evidence did not support a conviction; charges were withdrawn due to either a plea bargain or the introduction of new evidence.
2.3.3b Program Sub-Activity: Protecting Canada’s livestock and aquatic animals
The CFIA works to protect Canada’s animal health status through the implementation of two main programs: Animal Health and Livestock Feeds.
Of the $140.1 million the Agency spent to achieve this Strategic Outcome, approximately $63.1 million was devoted to protecting Canada’s livestock and aquatic animals.
|
Expected Result: Entry and domestic spread of regulated animal diseases are controlled |
The compensation program is designed to encourage owners to report disease in their herds and flocks at the earliest signs, thereby preventing or reducing the spread of disease and assisting owners in rebuilding their herds. Under the Health of Animals Act, Canadians who care for or have control of an animal are required to report the presence or suspicion of a reportable disease listed in the Reportable Diseases Regulations to the CFIA. The Agency monitors, tests, inspects and orders quarantines to prevent, control or eradicate regulated animal diseases. To encourage early reporting of suspected diseased animals, the CFIA administers a compensation program. In 2006–07, the CFIA paid livestock owners $1.8 million in compensation compared with $5.1 million in 2005–06.32
The control of animal disease is a shared responsibility of the owner, the industry, and the federal government. In addition to the human and animal health benefits of reporting disease in farm animals, public confidence in Canada’s safe food supply is enhanced. Early reporting also helps Canada maintain its excellent international animal health status, which, in turn bolsters Canadian exports of animals and animal products. The CFIA provides annual reports to the World Organisation for Animal Health on the status of animal diseases in Canada. The Agency also belongs to the Canadian Animal Health Network, which links partners involved in monitoring animal diseases within Canada.
|
Table 2.3.3b.2 — Economic Value of Trade in Animals and Animal Products to Canada (2006) |
|
|
Total imports: $776.5 million |
Total exports: $2.99 billion |
|
Source: World Trade Atlas, Statistics Canada |
|
To control the entrance of regulated diseases into Canada, the CFIA, in partnership with the Canada Border Services Agency and the Canadian Wildlife Service, regulates the entry of all imported animals and animal products.33 The Agency also carries out scientific risk evaluations including evaluating the risks related to the commodity being imported, and the disease status of the country from which the product originates. Information garnered from the risk evaluations supports the CFIA’s decisions relating to regulation and the imposition of import controls.
![]() The target for keeping newly regulated diseases from entering Canada is the detection of zero cases. In both 2005–06 and 2006–07, no evidence was found indicating that any new foreign animal disease entered Canada, indicating that the CFIA target was met.34
The target for keeping newly regulated diseases from entering Canada is the detection of zero cases. In both 2005–06 and 2006–07, no evidence was found indicating that any new foreign animal disease entered Canada, indicating that the CFIA target was met.34
Despite the best efforts of the CFIA, foreign animal diseases occasionally do enter Canada. In such cases, the Agency implements control programs that are designed to prevent or mitigate the effects of disease outbreaks.
![]() The target for these control programs is to have no increase in the proportion of domestic animals with regulated animal diseases found in Canadian herds or flocks.35
The target for these control programs is to have no increase in the proportion of domestic animals with regulated animal diseases found in Canadian herds or flocks.35
The following provides a summary description of the CFIA’s actions in 2006–07 regarding the control programs for Chronic Wasting Disease, bovine tuberculosis and scrapie:
|
Chronic Wasting Disease is a transmissible spongiform encephalopathy (TSE) that affects deer and elk. It is controlled by the CFIA in farmed elk and deer. Eleven animals tested positive this year compared with one from last year. All the cases occurred in three infected farmed cervid herds. However, sampling and testing programs indicated that this disease is still present in wild deer and elk. Given the disease’s long incubation period, surveillance and testing of farmed animals will continue to ensure the disease does not spread from wild to farmed populations. Bovine tuberculosis is a bacterial disease capable of remaining dormant in animals for several years before emerging. Long-term surveillance by veterinary inspectors is therefore required before a country can declare its animals free from this disease. Most of Canada is free of bovine tuberculosisinfected animals. A total of 21 cases were detected this year as compared with eight cases detected in 2005–06. All were additional cases found during depopulation of one infected herd detected in January 2006. Surveillance also determined that two farms were exposed to animals with this disease and, as a result, both farms were depopulated. It is important to note that none of the infected carcasses entered the food chain and there was no risk to human health. Scrapie is another transmissible spongiform encephalopathy (TSE) affecting sheep and goats. The CFIA’s control program for scrapie requires that all animals exposed to the disease be destroyed and prevented from entering the food chain. In 2006–07, the CFIA identified and destroyed two animals from the same flock as compared with 1,217 animals from four different flocks affected by scrapie in 2005–06. |
Revised import policies for bluetongue and anaplasmosis
The CFIA’s import controls are designed to effectively mitigate the risks posed by foreign animal diseases. These measures are periodically reviewed to ensure that they reflect the most current scientific information, remain effective and do not impose unwarranted trade restrictions. In line with this approach, Canada has reviewed and revised its bluetongue and anaplasmosis import controls for animals from the United States. These ailments pose no human health implications.
Bluetongue and anaplasmosis are diseases of domestic and wild ruminants that can be transmitted by biting insects such as midges and ticks. Both bluetongue and anaplamosis are reportable diseases under the Health of Animals Act.
The CFIA reviewed its import policy for bluetongue in July 2006. In consultation with a range of stakeholders, including provincial governments, animal health experts and industry, a decision was reached, as announced in February 2007, that bluetongue testing or herd certification would no longer be required for animals imported from the U.S.
In December 2006, the CFIA reviewed the import conditions for anaplasmosis for cattle and other ruminant animals from the United States. Relevant scientific factors were reviewed and various options ranging from status quo to complete deregulation were examined. The consensus of opinion was to reduce the level of required testing for anaplasmosis in breeding stock to a single test, utilizing improved testing methodology.
Feeder cattle for introduction to approved feedlots in Canada were previously eligible to be imported year-round from several northern states without testing, but with the changes to the bluetongue and anaplasmosis requirements, animals may now be imported to approved feedlots from any U.S. state year-round without testing for either disease.
The CFIA has now finalized the administrative mechanisms needed to issue import permits under the new conditions.
|
Expected Result: Industry complies with federal acts and regulations for livestock |
Effective feeds contribute to producing and maintaining healthy livestock for the production of safe food. Under the authority of the federal Feeds Act and the Health of Animals Act and their respective regulations, the CFIA administers a national livestock feed program to confirm that livestock feeds — either manufactured and sold in Canada or imported into this country — are safe, effective and labelled appropriately.
Inspections
As part of this program, the Agency inspects feed mills, rendering facilities, and on-farm feed mixers to assess the extent to which feed products are in compliance with federal regulations. Feed product tests are conducted through various inspection programs, including: the traditional feed inspection system, which analyzes feed products for chemical contamination, drug residue, heavy metals and salmonella; testing of drugs in feed; and inspection related to controlling the feeding of mammalian proteins to ruminant animals, as stipulated under the 1997 Feed Ban.
Feed Ban36
In 1997, as part of a series of preventative measures to mitigate the spread of BSE in the Canadian herd, regulations were introduced for the rendering, feed production and distribution sectors. Referred to as the “Feed Ban,” these regulations prohibit feeding most mammalian proteins to ruminant animals, such as cattle, sheep and goats. The ban requires rendering facilities, feed manufacturers, feed retailers and livestock producers to follow and document production and feeding procedures to prevent the inclusion of prohibited materials (mammalian proteins) in feed and feed ingredients intended for ruminant animals, such as cattle, sheep and goats.
Ensuring that feed for these animals is free from prohibited mammalian proteins is a critical step in reducing the risk that new cases of BSE will occur. The CFIA conducts inspections at commercial and on-farm feed manufacturers, rendering facilities and retail outlets to verify compliance with the Health of Animals Regulations, with respect to the Feed Ban. When instances of non-compliance are identified, CFIA inspectors set out timeframes for corrective actions, based on health and safety considerations, after which they return to verify that the issue has been appropriately met.
Industry compliance with these regulations in relation to the targets set by the CFIA is presented in Table 2.3.3b.3 below. Inspections of commercial feed mills and rendering establishments are reported because they represent a higher risk in terms of potential contamination of non-prohibited material or ruminant feed with contaminated material.
It is important to note that the CFIA’s Report on Plans and Priorities 2006–07 included four performance indicators:
- Extent to which feed mills inspected comply with the Feeds Act, including the Feed Ban
- Extent to which renderers inspected comply with the Feeds Act, including the Feed Ban (under the Health of Animals Regulations)
- Extent to which feed mills inspected are without any major deviations from the Feeds Act, including the Feed Ban (under the Health of Animals Regulations)
- Extent to which renderers inspected are without any major deviations from the Feeds Act, including the Feed Ban (under the Health of Animals Regulations)
The compliance rates for indicators 1 and 2 include all (minor and major) deviations with respect to compliance to both the Feeds Act and the Feed Ban, whereas the compliance rates for indicators 3 and 4 are specific to major deviations. Major deviations are areas of noncompliance that have the potential to result in risk to either human or animal health and are relevant to the protection of Canada’s livestock. Minor deviations, such as absence of a signature or not retaining records for the required period, have little relevance to the risk mitigation objective of the applicable acts and regulations.
Review of these performance indicators resulted in a determination that the first two indicators, as now worded, are of little significance from a performance reporting perspective due to the inclusion of minor deviations. They are, therefore, not included in this report, nor will they be included in subsequent RPPs. At the same time, two indicators that were presented in last year’s Performance Report, but not in the 2006–07 RPP, do reflect CFIA’s risk mitigation focus and are meaningful from a performance perspective. As a result, they have been included in this report, along with trend data, and will be included in future reports. They are:
- Percentage of feed mills that are compliant with the Feed Ban (without major deviations)
- Percentage of feed renderers that are compliant with the Feed Ban (without major deviations)
![]() The CFIA 2006–07 target for percentage of commercial feed mills that are compliant with the Feed Ban (without major deviations) is 95%. This rate includes all feed mills that came into compliance throughout the reporting period, including those that were detected as being noncompliant at first inspection
but took action to successfully move into compliance during the reporting period. In 2006–07, the Feed Ban compliance rate for commercial feed mills was 94%, down from 96% in 2005–06. The performance target is considered met and the decrease in compliance is not considered significant. That said, the CFIA is committed to continue working with partners to promote compliance
with the Feed Ban.
The CFIA 2006–07 target for percentage of commercial feed mills that are compliant with the Feed Ban (without major deviations) is 95%. This rate includes all feed mills that came into compliance throughout the reporting period, including those that were detected as being noncompliant at first inspection
but took action to successfully move into compliance during the reporting period. In 2006–07, the Feed Ban compliance rate for commercial feed mills was 94%, down from 96% in 2005–06. The performance target is considered met and the decrease in compliance is not considered significant. That said, the CFIA is committed to continue working with partners to promote compliance
with the Feed Ban.
![]() The target for compliance of commercial feed mills with the Feeds Act including the Feed Ban (without major deviations) combined is 96%. In 2006–07, the compliance rate was 82%. As a new indicator, there is some question as to the relevance and reasonableness of the target compliance rate. This
will be reviewed in the coming year. A number of factors of potential significance may have contributed to this result. The CFIA employees directly responsible for the inspection of commercial feed mills across Canada were provided with significant additional training on the feed mills industry practices and products, as well as CFIA inspection procedures and the assessment of
compliance during 2006–07. There was also an increase in the frequency of inspection during the reporting year for this sector — from one full inspection per facility per year to one full inspection plus one to three partial inspections. These factors, individually and combined, may have affected the compliance rate. Also of potential significance, compliance issues not
associated with the Feed Ban may be perceived by both industry and inspection staff as being lower priority, based on risk. This may have resulted in a larger number of instances of non-compliance not being resolved in a timely manner. Finally, existing issues around the use of compounded drugs and the use of appropriate sequencing to manage drug residues in feed are being clarified
by Health Canada and CFIA. Once the required clarifications are fully communicated, it is expected that compliance will improve.
The target for compliance of commercial feed mills with the Feeds Act including the Feed Ban (without major deviations) combined is 96%. In 2006–07, the compliance rate was 82%. As a new indicator, there is some question as to the relevance and reasonableness of the target compliance rate. This
will be reviewed in the coming year. A number of factors of potential significance may have contributed to this result. The CFIA employees directly responsible for the inspection of commercial feed mills across Canada were provided with significant additional training on the feed mills industry practices and products, as well as CFIA inspection procedures and the assessment of
compliance during 2006–07. There was also an increase in the frequency of inspection during the reporting year for this sector — from one full inspection per facility per year to one full inspection plus one to three partial inspections. These factors, individually and combined, may have affected the compliance rate. Also of potential significance, compliance issues not
associated with the Feed Ban may be perceived by both industry and inspection staff as being lower priority, based on risk. This may have resulted in a larger number of instances of non-compliance not being resolved in a timely manner. Finally, existing issues around the use of compounded drugs and the use of appropriate sequencing to manage drug residues in feed are being clarified
by Health Canada and CFIA. Once the required clarifications are fully communicated, it is expected that compliance will improve.
![]() The CFIA 2006–07 target percentage of feed renderers that are compliant with the Feed Ban (without major deviations) is 93%. The compliance rate, for the purpose of this analysis, includes only major deviations. This rate includes all renderers that came into compliance throughout the reporting period,
including those that were detected as being non-compliant at first inspection but took action to successfully move into compliance during the reporting period. The actual compliance rate for renderers was 100%. The 2005–06 compliance rate was 93%. As there are only 31 inspection sights, including year over year comparisons, actual results could fluctuate greatly with only a few
non-compliant inspections. At the end of March 2007, there were no outstanding major deviations with the Feed Ban.
The CFIA 2006–07 target percentage of feed renderers that are compliant with the Feed Ban (without major deviations) is 93%. The compliance rate, for the purpose of this analysis, includes only major deviations. This rate includes all renderers that came into compliance throughout the reporting period,
including those that were detected as being non-compliant at first inspection but took action to successfully move into compliance during the reporting period. The actual compliance rate for renderers was 100%. The 2005–06 compliance rate was 93%. As there are only 31 inspection sights, including year over year comparisons, actual results could fluctuate greatly with only a few
non-compliant inspections. At the end of March 2007, there were no outstanding major deviations with the Feed Ban.
![]() The CFIA 2006–07 target percentage of feed renderers for the Feeds Act, including the Feed Ban (without major deviations), combined was 93%. The compliance rate was 100%. The total population of facilities subject to this inspection activity is relatively small (31), thus even a small change in
the number of facilities found to be in non-compliance on a year-to-year basis would have a significant impact on the overall compliance rate. The year-to-year increase in compliance from 93% to 100% demonstrates the CFIA’s commitment to continuously working with partners to improve compliance rates, thus protecting Canada’s livestock and ultimately contributing to the
safety of Canada’s food supply.
The CFIA 2006–07 target percentage of feed renderers for the Feeds Act, including the Feed Ban (without major deviations), combined was 93%. The compliance rate was 100%. The total population of facilities subject to this inspection activity is relatively small (31), thus even a small change in
the number of facilities found to be in non-compliance on a year-to-year basis would have a significant impact on the overall compliance rate. The year-to-year increase in compliance from 93% to 100% demonstrates the CFIA’s commitment to continuously working with partners to improve compliance rates, thus protecting Canada’s livestock and ultimately contributing to the
safety of Canada’s food supply.
Enforcement
In 2006–07, the CFIA investigated 933 instances of non-compliance compared with 875 in 2005–06. Of the 933 instances of non-compliance, 842 were for the Health of Animals Act and 91 were for the Feeds Act compared with 824 for the Health of Animals Act and 51 for the Feeds Act in 2005–06. Combined with investigations carried over from previous reporting periods, these investigations resulted in 12 convictions (three for the Health of Animals Act and nine for the Feeds Act) compared with one conviction in 2005–06. Total fines for 2006–07 were $207,000 ($197,000 for Health of Animals Act and $9,250 for Feeds Act). In 2005–06, the total fines totalled $90,000 ($75,000 for Health of Animals Act and $15,000 for Feeds Act). Fines have therefore nearly doubled from 2005–06 to 2006–07.
2.3.3c Program Sub-Activity: Assessing agricultural products
The strategies related to this sub-activity focus on assessing and approving new agricultural products to determine whether or not they meet standards set by federal acts and regulations.
Of the $140.1 million the Agency spent to achieve this Strategic Outcome, approximately $11.8 million was devoted to assessing agricultural products.
|
Expected Result: Agricultural products meet the requirements of federal acts and regulations |
The CFIA assesses and approves new feeds, fertilizers and supplements. The Agency also monitors the release of proposed new products for research purposes.
Feeds
The Feeds Act and Regulations require pre-market approval of all new ingredients in livestock feeds and the registration of specified mixed feeds. Products are approved by the CFIA only if this review has determined that the products pose minimal risk of adversely affecting the environment, animals, plants or humans. In 2006–07, the CFIA received and completed reviews of 532 submissions requesting approval for new products. Of these, 477 (90%)37 met legislative requirements and were approved. It is important to note that these data reflect applicant performance, as opposed to the CFIA’s performance.
Fertilizers and fertilizer supplements
Federal regulations require that all fertilizer and supplement products sold or imported into Canada be safe when used according to directions, efficacious for the intended purpose, and be properly labelled. Some fertilizers and most supplements are also subject to registration, which requires assessment by the CFIA prior to their importation and sale. Other fertilizers are exempt from the registration requirement, but must still meet prescribed safety, efficacy and labelling standards at time of sale. In 2006–07, the CFIA received 775 submissions and completed reviews of 176 submissions39 requesting approval for new products.
The CFIA samples biotechnology-derived fertilizer-supplement products from both retail and manufacturing outlets to help ensure that the products have the appropriate amount of viable cells in accordance with the purported guarantee.
![]() The CFIA’s target for fertilizer-supplements compliance is 95%.
The CFIA’s target for fertilizer-supplements compliance is 95%.
In 2006–07, the compliance rate was 96%, exceeding the 2005–06 compliance rate of 92%. The increase in compliance is attributed to the CFIA’s new stakeholder-engagement model, which likely led to increased industry awareness and willingness to comply with the regulations.
Approval of plants with novel traits and inspection of confined field trials
The CFIA regulates and authorizes Plants with Novel Traits (PNTs) that are imported or released into the natural environment. The CFIA’s confined field trial program allows developers to conduct research on their products and to determine how they behave in the environment, while allowing the CFIA to establish that the material is adequately controlled and confined. The Agency sets specific terms and conditions for conducting these trials.
![]() The target compliance rate for monitoring confined field trials is 90%. In both 2006–07 and 2005–06, the compliance rate for monitoring confined field trials was 94%,39 exceeding the 90% target compliance rate. When compliance problems were
identified (in the remaining 6%), the CFIA issued compliance letters outlining any corrective actions required and conducted followup inspections, where appropriate, to ensure that the required corrective actions had been taken. No incidence of non-compliance resulted in an environmental or safety concern. In 2006–07, the CFIA approved eight new PNTs for unconfined environmental
release. As of March 31, 2007, the total number of PNTs approved for unconfined environmental release was 57.
The target compliance rate for monitoring confined field trials is 90%. In both 2006–07 and 2005–06, the compliance rate for monitoring confined field trials was 94%,39 exceeding the 90% target compliance rate. When compliance problems were
identified (in the remaining 6%), the CFIA issued compliance letters outlining any corrective actions required and conducted followup inspections, where appropriate, to ensure that the required corrective actions had been taken. No incidence of non-compliance resulted in an environmental or safety concern. In 2006–07, the CFIA approved eight new PNTs for unconfined environmental
release. As of March 31, 2007, the total number of PNTs approved for unconfined environmental release was 57.
The CFIA is also engaged in consultation and policy development in key areas of plant biosafety, including plant molecular farming and adventitious presence. Plant molecular farming involves the growing of plants to produce pharmaceutical or industrial compounds, instead of for traditional uses such as for food, feed or fibre. Adventitious presence refers to the trace level of unintentionally present, biotechnologyderived material in seeds, grains/oilseeds, livestock feed and food, including material that is not approved.
Licensing veterinary biologics
In recent years, the animal health products industry has increasingly relied on veterinary biologics to prevent and diagnose disease. These include vaccines, antibody products, and diagnostic tests. Unlike some pharmaceutical products, most veterinary biologics leave no chemical residues in animals. In addition, most disease organisms do not develop a resistance to the immune response produced by a veterinary biologic.
The CFIA is responsible for licensing and regulating veterinary biologics in Canada. This licensing program is central to Canada’s national animal health program, which strives to protect the health of Canadian citizens, their domestic pets and animals used for food.
To meet Canadian licensing requirements, veterinary biologics must be shown to be pure, potent, safe and effective when used in accordance with the manufacturer’s label recommendations. In 2006–07, the CFIA received 40 new complete submissions, completed 52 initial reviews (including product files received in previous years), and licensed or registered 39 new products. In 2006, the average initial review time per product file was 86 days. Continuous efforts towards the development and implementation of service standards are being undertaken.
Porcine Circovirus vaccineIn 2006, the swine industry in 2006 experienced severe problems with Post-weaning Multisystematic Wasting Syndrome (PMWS) in pigs, also known as Porcine Circovirus Associated Disease (PCVAD). Porcine circovirus-associated diseases, including PMWS, are widespread in Canada and around the world. Factors such as genetic predisposition, the emergence of a new strain of porcine circovirus type 2 (PCV2) virus, interactions with other common infectious agents (such as viruses and Mycoplasma), and management factors are thought to also play a role in this swine disease. This syndrome can be devastating to individual producers and to the swine industry as a whole; however, it does not fall within the Canadian Food Inspection Agency's (CFIA) disease control and eradication mandate, which focuses on foreign animal diseases and those that present a public health or food safety threat. The CFIA regulatory officials worked, on a priority basis, on the review and approval of porcine circovirus type 2 (PCV2) vaccine submissions, and issuance of import permits for the available vaccine. Currently, three vaccines are available in Canada. These products are currently available for emergency use under veterinary supervision. |
2.3.4 Strategic Outcome: Security from deliberate threats to Canada’s food supply and agricultural resource base
|
|
The Government of Canada is committed to protecting Canadians from deliberate threats to their safety. Under the Emergency Preparedness Act, the CFIA is mandated to prepare for, and respond to, emergencies involving food safety, animal health, plant health and any other situation related to the Agency’s programs. Chemical, physical, and biological threats to humans can occur through the deliberate contamination of the environment or of food and water. Threats to Canada’s animal and plant resource base may occur through the deliberate introduction of significant plant pests or foreign animal diseases.
The CFIA’s emergency preparedness program focuses on strategies that help the Agency and its partners reach a state of readiness to provide and promote an effective and rapid response to food safety, animal or plant health emergencies, including deliberate threats. These strategies are part of the Government of Canada’s Public Security and Anti-Terrorism (PSAT) initiative, which is a key element of Canada’s National Security Policy.
To achieve this Strategic Outcome, the CFIA works in collaboration with a number of partners, including Public Safety Canada, the Public Health Agency of Canada, provincial and territorial governments, municipalities, and law enforcement authorities.
In 2006–07, the CFIA spent approximately 4% of its budget to achieve this Strategic Outcome.
2.3.4a Program Sub-Activity: Preparing for emergencies
The true level of preparedness can be known only when an emergency occurs. The CFIA continues to develop and update emergency response plans within its mandate, and to lead or participate in emergency exercises. Such exercises give the Agency the opportunity to test, assess and refine its approaches as necessary.
Of the $23.8 million the CFIA spent to achieve this Strategic Outcome, approximately $1.2 million was devoted to preparing for emergencies.
|
Expected Result: The Agency is in a state of readiness for an effective, rapid response to emergencies |
Responding to an emergency is a complicated process involving many partners. Launching an effective, integrated response to agricultural and food safety emergencies requires that all involved players understand their respective roles and responsibilities, and that information for making decisions flows quickly among them. Numerous federal departments, provinces, territories, municipal authorities as well as the United States government and others, play key roles in responding to an emergency. Therefore, effective intergovernmental links must be established.
In 2006–07, the CFIA continued to participate in the Trilateral Co-operation’s Emergency Preparedness and Response Working Group, established to enhance the ability of Canada, the United States, and Mexico to respond to emergencies, including those for food, which may affect more than one participating country. The group’s workplan supported the objectives of the Security and Prosperity Partnership of North America, a trilateral effort to increase security and enhance prosperity among the three countries through greater co-operation and information sharing.
![]() Funding from the Public Security and Anti-Terrorism
(PSAT) Strategy was used to improve the Agency’s ability to manage emergencies, specifically avian influenza. Three steps were taken to ensure the CFIA met this target. First, the Agency re-designed the epidemiology/ investigation questionnaire for avian
diseases to ensure complete and detailed information about the premises under investigation are collected in a systematic fashion. Second, the Agency added the questionnaire to the web-based Canadian Emergency Management Response System
(CEMRS) to facilitate access to and sharing of information during an emergency. Finally, the CFIA trained its field staff across the country in the
use of the questionnaire and CEMRS.
Funding from the Public Security and Anti-Terrorism
(PSAT) Strategy was used to improve the Agency’s ability to manage emergencies, specifically avian influenza. Three steps were taken to ensure the CFIA met this target. First, the Agency re-designed the epidemiology/ investigation questionnaire for avian
diseases to ensure complete and detailed information about the premises under investigation are collected in a systematic fashion. Second, the Agency added the questionnaire to the web-based Canadian Emergency Management Response System
(CEMRS) to facilitate access to and sharing of information during an emergency. Finally, the CFIA trained its field staff across the country in the
use of the questionnaire and CEMRS.
While these activities were targeted to improve the CFIA’s response to Notifiable Avian Influenza, staff training and the flexibility of CEMRS provided the additional benefits of strengthening CFIA’s overall ability to respond to additional foreign animal diseases and other emergencies.
2.3.4b Program Sub-Activity: Enhancing capacity to respond to emergencies
In addition to preparing for emergencies through joint exercises, the CFIA plays a significant role in emergency responses to deliberate threats. The Agency’s front-line investigation and scientific expertise, as well as its considerable, widely-dispersed laboratory system, have enhanced its capacity for testing for potential contaminants, thereby contributing to the CFIA’s emergency response capabilities.
Of the $23.8 million the CFIA spent to achieve this Strategic Outcome, approximately $22.6 million was devoted to enhancing the Agency’s capacity to respond to emergencies.
|
Expected Result: The Agency has the capacity to respond to emergencies |
In 2006–07, the CFIA continued to expand the capacity of its laboratories to deal with deliberate threats to the food supply and to plant and animal resources. Improvements include the enhancement of bio-security measures and procedures, greater laboratory capacity, and the enhancement of laboratory infrastructure.
Specifically, the CFIA developed guidelines and standards for the containment of plant and animal pathogens and worked with federal, provincial and international partners to enhance its animal health diagnostic network. The Agency also developed new and faster testing methodologies for microbial food contaminants. In addition, the CFIA funded equipment and infrastructure upgrades at CFIA laboratories across the country to enhance its laboratory emergency response capacity.
All health, safety and protection situations have a geographic dimension. Visual representations of geographic information are thus essential to the CFIA’s effective response to animal, plant and food-related incidents across Canada. In 2006–07, the CFIA used funding under the Public Security and Anti-Terrorism (PSAT) initiative to prepare a GIS Project Charter and Plan for the proposed development of a geographic information system. Funding was also used to maintain equipment, software licences and telecommunications needed in CFIA National Emergency Operation Centres, building on the upgrades achieved in 2005–06.
During the 2006–07 fiscal year, the Agency also moved to update and upgrade its system for maintaining contact lists. During emergencies, or when dealing with urgent issues, the Agency must be able to quickly contact industry and other government departments via e-mail. To facilitate better responses to emergency situations and urgent circumstances, the CFIA has begun the development of a website containing all emergency contact information, to aid rapid communications between stakeholders.
![]() The CFIA target for the implementation of the National Emergency Response System was full implementation of the system. The CFIA met this target, enabling the Agency to engage in a co-ordinated federal response to potential emergencies that could affect food safety, animal or plant health.
The CFIA target for the implementation of the National Emergency Response System was full implementation of the system. The CFIA met this target, enabling the Agency to engage in a co-ordinated federal response to potential emergencies that could affect food safety, animal or plant health.
4 HACCP: Hazard Analysis Critical Control Point, more details in Section 3.1
5 Data Systems and Controls: Reasonable
6 Data Systems and Controls: Good
7 Data Systems and Controls: Good
8 In order to further improve reporting to Canadians on
BSE, the Agency will develop outcome based performance targets as part of its review of the Agency’s Performance Measurement Framework in Fall, 2007.
9 Data Systems and Controls: Good
10 Although the Office International de Épizooties
(OIE) became officially known as the World Organisation for Animal Health in 2005, it has retained the common usage acronym,
OIE, which appears in this document.
11 Data Systems and Controls: Reasonable
15 The CFIA recognizes that this Expected Result is activity-based and not outcome-based. However, given that the expected result was presented in the Report on Plans and Priorities, the Agency must report on the results in this Performance Report. A recently conducted review and revision of the Agency’s
PAA,
including the development of outcome-based Expected Results, will address this issue in the future.
16 The CFIA recognizes that this Expected Result is activity-based and not outcome-based. However, given that the expected result was presented in the Report on Plans and Priorities, the Agency must report on the results in this Performance Report. A recently conducted review and revision of the Agency’s
PAA,
including the development of outcome-based Expected Results, will address this issue in the future.
17 In order to further improve reporting to Canadians on Fair
Labelling, the Agency will develop outcome based performance targets as part of its review of the Agency’s Performance Measurement Framework in Fall, 2007.
18 Data Systems and Controls: Good
19 Data Systems and Controls: Good
20 Data Systems and Controls: Good
21 Data Systems and Controls: Good
22 Data Systems and Controls: Reasonable
23 Data Systems and Controls: Good
26 Data Systems and Controls: Good
27 Data Systems and Controls: Good
28 Data Systems and Controls: Good
29 Data Systems and Controls: Reasonable
30 Data Systems and Controls: Reasonable
31 Data Systems and Controls: Reasonable
32 Data Systems and Controls: Good
33 Data Systems and Controls: Good
34 Data Systems and Controls: Good
35 This performance indicator will be reviewed to better reflect the gency’s risk mitigation activities.
36 Further information on the Feed Ban can be found in Section 2.3.2b.
37 Data Systems and Controls: Reasonable
38 Data Systems and Controls: Good