ARCHIVED - Patented Medicine Prices Review Board Canada
 This page has been archived.
This page has been archived.
Archived Content
Information identified as archived on the Web is for reference, research or recordkeeping purposes. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats on the "Contact Us" page.
2009-10
Report on Plans and Priorities
Patented Medicine Prices Review Board Canada
The original version was signed by
The Honourable Leona Aglukkaq
Minister of Health
Table of Contents
- Raison d’être
- Responsibilities
- Strategic Outcome and Program Activity Architecture (PAA)
- Planning Summary
- Risk Analysis
- Expenditure Profile
- Voted and Statutory Items
Section II — Analysis of Program Activities by Strategic Outcome
Section III — Supplementary Information
Chairperson's Message
I am pleased to present the 2009-2010 Report on Plans and Priorities for the Patented Medicine Prices Review Board (PMPRB).
The PMPRB is an independent, quasi-judicial body established by Parliament in 1987 under the Patent Act. Its mandate is two-fold: Regulatory – to ensure that prices charged by patentees for patented medicines sold in Canada are not excessive, thereby protecting consumers and contributing to Canadian health care; and, Reporting – to report on pharmaceutical trends of all medicines, and on R&D spending by pharmaceutical patentees, thereby contributing to informed decisions and policy-making.
The PMPRB contributes to the broader objective of improving the health of Canadians by reducing excessive patented drug prices and related cost pressures on the health care system.
The PMPRB’s capacity to carry out its statutory mandate is dependent on relevant and effective Excessive Price Guidelines (Guidelines) and on the ability to conduct hearings, when required.
In 2005, the Board initiated a process to review its Guidelines, including consulting with key stakeholders as required by the Patent Act. As a result of stakeholder input through consultation meetings and written submissions, significant progress in identifying potential issues, options and proposals for modernizing the Guidelines has occurred. One final round of consultations on final Guidelines’ proposals is planned for the spring of 2009. Next steps would include implementation of the revised Guidelines and monitoring and evaluation of the impact of the changes made.
In recent years, the PMPRB has experienced a significant increase in the number of hearings to determine if the price of a patented drug product is excessive. While some hearings have been completed, a number are still ongoing, and additional new hearings on other matters are likely in 2009-2010. It is hoped that the implementation of revised, modernized Guidelines will help mitigate this trend in the future.
In 2008 the PMPRB received on-going additional resources to more effectively and proactively provide educational outreach to patentees and other stakeholders, as well as implement and monitor the revised Guidelines, manage the increasing number of drugs coming under its jurisdiction, carry out ongoing price reviews and investigations within reasonable timelines, and hold timely hearings, as required.
A key activity under the PMPRB’s reporting mandate has been and will continue to be to work in collaboration with the Canadian Institute for Health Information (CIHI) and participating federal/provincial/territorial drug plans, to produce analyses and reports under the National Prescription Drug Utilization Information System (NPDUIS). Through critical analyses of price, utilization and cost trends, the PMPRB provides Canada’s health system with comprehensive, timely and accurate information on prescription drug trends and cost drivers. In October of 2005, in the context of the National Pharmaceuticals Strategy, the PMPRB also received direction from the federal Minister of Health to monitor and report on non-patented prescription drug prices. In 2008-2009, this work was folded under the broader umbrella of NPDUIS.
The PMPRB remains committed to predictability, fairness and transparency in the fulfilment of its regulatory and reporting responsibilities, to ongoing engagement of its key stakeholders, and to continual monitoring of developments within the larger pharmaceutical environment. The PMPRB continues to strive to ensure that the price regulatory regime is appropriate, efficient and effective.
Paper copy signed by
Brien G. Benoit, M.D.
Chairperson
Section I — Board Overview
Raison d’être
The Patented Medicine Prices Review Board (PMPRB) has a dual role:
Regulatory – To ensure that prices charged by patentees for patented drug products sold in Canada are not excessive, thereby protecting consumers and contributing to Canadian health care.
Reporting – To report on pharmaceutical trends of all drug products, and on R&D spending by pharmaceutical patentees, thereby contributing to informed decisions and policy-making.
Responsibilities
The PMPRB is an independent, quasi-judicial body created by Parliament as a result of revisions to the Patent Act (Act) in 1987 (Bill C-22) which increased patent protection for pharmaceuticals.
The PMPRB represents a strategic component of the federal government’s policy to balance consumer protection and health care with the trade and industrial development objectives of pharmaceutical patent legislation.
Subsequent revisions to the Act in 1993 (Bill C-91) further increased patent protection for pharmaceutical products by eliminating compulsory licensing. The amendments also gave the PMPRB increased remedial powers and shifted ministerial responsibility for the PMPRB to the Minister of Health. Prior to that, responsibility for the PMPRB rested with the Minister of Consumer and Corporate Affairs (now the Minister of Industry), who has overall responsibility for the Act. The Minister of Health is responsible for the pharmaceutical provisions of the Act as set out in sections 79 to 103.
Regulatory Role
Health Canada assesses new drugs to ensure that they conform to the Food and Drugs Act and the Food and Drug Regulations. Formal authorization to market or distribute a drug is granted through a Notice of Compliance (NOC). A drug may be temporarily distributed with specified restrictions before receiving an NOC, as an Investigational New Drug or under Health Canada´s Special Access Programme (SAP).
The PMPRB is responsible for regulating the prices that patentees charge — the factory-gate price — for patented drugs sold in Canada to wholesalers, hospitals, pharmacies or others, for human and veterinary use, to ensure that they are not excessive. The PMPRB regulates the price of each patented drug product, including each strength of each dosage form of each medicine sold in Canada. This is normally the level at which Health Canada assigns a Drug Identification Number (DIN).
The PMPRB has no authority to regulate the prices of non-patented drugs, and does not have jurisdiction over prices charged by wholesalers or retailers, or over pharmacists´ professional fees. Also, matters such as whether drugs are reimbursed by public drug plans, distribution and prescribing are outside the purview of the PMPRB.
Under the Patented Medicines Regulations (Regulations), patentees are required to file price (or revenue) and sales information twice a year for each strength of each dosage form of each patented drug sold in Canada for price regulation purposes. Patentees are also required to file R&D and expenditures for all drugs once a year for reporting purposes.
Patentees are also required to inform the PMPRB of their intention to sell a new patented drug. They are not required to obtain approval of the price of a patented drug before it is sold, but they are required to comply with the Act to ensure that prices of patented drugs sold in Canada are not excessive. In the event that the Board finds, after a public hearing, that a price is or was excessive in any market, it may order the patentee to reduce the price and take measures to offset any excess revenues it may have received.
Reporting Role
The PMPRB reports annually to Parliament, through the Minister of Health, on pharmaceutical trends relating to all drugs, and on the R&D spending by pharmaceutical patentees. In addition to these reporting responsibilities, under section 90 of the Act the Minister of Health has the authority to direct the
PMPRB to inquire into any other matter. Under this provision, the Minister has directed the Board to undertake two initiatives: the National Prescription Drug Utilization Information System (NPDUIS), and monitoring and reporting on Non-Patented Prescription Drug Prices (NPPDP).
National Prescription Drug Utilization Information System (NPDUIS)
Since 2001, pursuant to an agreement by the Federal/Provincial/Territorial Ministers of Health, the PMPRB has been conducting research under the NPDUIS. The purpose of the NPDUIS is to provide critical analyses of price, utilization and cost trends so that Canada´s health system has more comprehensive and accurate information on how prescription drugs are being used and on sources of cost pressures.
Non-Patented Prescription Drug Prices (NPPDP)
In 2005, the then Minister of Health, on his behalf and that of his federal and provincial and territorial colleagues, directed the PMPRB to monitor and report on non-patented prescription drug prices. This function is aimed at providing a centralized credible source of information on non-patented prescription drug
prices. As of April 2008, NPPDP studies are conducted under the umbrella of the NPDUIS.
Strategic Outcome and Program Activity Architecture (PAA)
The PMPRB has one Strategic Outcome (SO) and two program activities (PA) — representing its regulatory and reporting roles — which are illustrated in the following chart:
Compliance and enforcement of non-excessive pricing for patented medicines
The PMPRB is responsible for regulating the prices that patentees charge for patented drugs sold in Canada to wholesalers, hospitals, pharmacies or others, for human and veterinary use. Through this program activity, the
PMPRB reviews the prices that patentees charge for patented drugs, based on the price review factors in the Patent Act, to ensure that these prices are not excessive. In the event that the Board finds, following a public hearing, that a price is excessive in any market, it may order the patentee to reduce the
price and take measures to offset any excess revenues it may have received as a result of excessive prices.
Pharmaceutical trends
Through this program activity, the Patented Medicine Prices Review Board (PMPRB) provides analysis of pharmaceutical price trends and research and development spending by pharmaceutical patentees. It also provides critical analyses of price, utilization and cost trends for prescription drugs, and information on non-patented prescription
drug prices. The PMPRB reports on this information and its price review and enforcement activities as they relate to excessive pricing for patented medicines, both annually to Parliament, through the Minister of Health, and through special published studies.
Planning Summary
Financial Resources($ thousands)
| 2009–2010 | 2010–2011 | 2011–2012 |
|---|---|---|
|
11,358 |
11,983 |
11,608 |
Human Resources (FTE)
| 2009–2010 | 2010–2011 | 2011–2012 |
|---|---|---|
|
76 |
76 |
76 |
Planning Summary Table ($ thousands)
|
Strategic Outcome: Canadians and their health care system are protected from excessive pricing for patented medicines sold in Canada and are informed on pharmaceutical trends. |
|||||
|
Performance Indicator |
Target |
||||
| ProgramActivity1 | Forecast Spending 2008–2009 | Planned Spending | Alignment to Government of Canada Outcomes |
||
|---|---|---|---|---|---|
| 2009–2010 | 2010–2011 | 2011–2012 | |||
|
PA 1: Compliance and enforcement of non-excessive pricing for patented medicines |
6,539.2 |
7,045.0 |
7,645.0 |
7,645.0 |
Healthy Canadians: by ensuring that patented drug product prices are not excessive.
|
|
PA 2: Pharmaceutical trends |
3,099.6 |
1,593.0 |
1,593.0 |
1,593.0 |
Healthy Canadians: by creating awareness of pharmaceutical trends and cost drivers. |
|
PA 3: |
- |
2,720.0 |
2,745.0 |
2,370.0 |
|
|
Total Planned Spending |
9,638.8 |
11,358.0 |
11,983.0 |
11,608.0 |
|
|
Services received without charge |
- |
1,146.7 |
1,159.2 |
1,174.8 |
|
|
Total PMPRB Spending |
9,638.8 |
12,504.7 |
13,142.2 |
12,782.8 |
|
Contribution of Priorities to Strategic Outcome
| Operational Priorities | Type | Links to Strategic Outcome3 | Description |
|---|---|---|---|
|
Strengthen policy and economic analysis |
Ongoing |
PA 1 |
Policy and economic analysis is essential to ensuring that the PMPRB’s regulatory activities remain relevant, appropriate and effective in the context of the evolving pharmaceutical environment. The PMPRB will:
|
|
Improve the timeliness of ongoing price reviews and investigations |
Ongoing |
PA 1 |
It is essential that price reviews and investigations be conducted in a timely fashion so as to ensure that excessive prices are reduced with the least possible delay. The PMPRB will:
|
|
Hold efficient hearings into excessive prices as required |
Ongoing |
PA 1 |
Hearings have increased significantly in number and complexity in recent years. The ability to hold public hearings when needed is a core component of the Board’s mandate and authority. As a quasi-judicial body, the PMPRB must have the capacity to give patentees a fair and timely hearing. The PMPRB will:
|
|
Enhance the relevance and timeliness of analytical studies under NPDUIS |
Ongoing
|
PA 2 |
The Minister has directed the PMPRB to undertake analyses of prescription drug price, utilization and cost trends in support of decision-making by federal/provincial/territorial drug plan managers. The PMPRB will:
|
| Management Priorities | Type | Links to Strategic Outcome | Description |
|
Renew the database which stores and manipulates pricing data |
Ongoing |
PA 3 |
The database housing price and sales information filed by patentees is critical to the conduct of regulatory activities. The PMPRB will take further steps to improve the responsiveness of this tool to the needs of the PMPRB by:
|
|
Implement Human Resources Plans |
Ongoing |
PA 3 |
The PMPRB will focus its efforts on increasing the capacity of the organization by:
|
Risk Analysis
The PMPRB’s capacity to carry out its statutory regulatory mandate is focused on its ability to conduct timely price reviews and hearings, when needed. From its creation until 2005, the PMPRB had been largely able to carry out its mandate with limited recourse to public hearings due at least in part to the success of the Board’s Excessive Price Guidelines (Guidelines) and its Voluntary Compliance Policy.
In 2005-2006, the PMPRB identified a risk under its regulatory mandate stemming largely from an unprecedented number of investigations and anticipated hearings into excessive drug prices. This, coupled with renewed stakeholder attention to drug prices and cost trends, and concerns about high introductory drug prices and other issues, prompted the Board to initiate a comprehensive process to review its Guidelines, including consulting with key stakeholders as required by the Patent Act.
The PMPRB has been challenged by the significance of, and wide-ranging views of stakeholders on the issues being addressed in the Guidelines review. A key risk relates to the operationalization and impact of the revised Guidelines. The increased complexity of the scientific review (due to additional therapeutic improvement factors) and the price review (including new reviews at the level of sub-markets and a new CPI “Delinking” methodology when price increases are due to changes in benefits for customers) will add to workload demands. These areas will need to be closely monitored and assessed with adjustments in capacity and process, and potentially even the Guidelines, undertaken as needed.
Workload pressures will likely continue with respect to the number and complexity of price reviews, investigations and hearings. A surge in the number of patented drug products sold by generic companies is expected. The revised Guidelines may also lead to more, not fewer, investigations and hearings as patentees adjust to and may attempt to exploit the revised Guidelines.
Resource requirements have been identified to complete the review of the Guidelines, analyze and provide advice on emerging scientific and price review issues, implement new standard operating procedures relative to the revised Guidelines, manage an increasing volume of regulatory filings, continue to carry out price reviews and investigations within reasonable timelines, and hold hearings in a reasonably expeditious manner.
In terms of the National Prescription Drug Utilization System (NPDUS), there is a risk that studies may not be sufficiently timely or the conclusions readily pertinent to address the needs of federal/provincial/territorial drug plan decision-makers. It will be important to continue to engage them in setting research priorities and put in place improved project management processes.
The PMPRB shares with other small organizations the difficulty in attracting and retaining highly specialized subject matter experts and the length of time required to engage new staff. At the same time, being a smaller organization with a relatively “flat” hierarchy, offers ample opportunity for interaction with peers and inter-branch cooperation and support. The PMPRB benefits from a highly competent and dedicated staff, and a corporate structure that is well-aligned to the PMPRB’s program responsibilities.
The opportunity, and the challenge, in 2009-2010 will be to ensure that human resource planning and business planning remain in alignment, and that operational staffing activities and other support services are equipped to support the evolving organizational needs and directions.
The PMPRB has implemented an integrated business and human resources planning framework that ensures the identification, review and documentation of human resources requirements on a quarterly basis, and a weekly review of the status of classification and staffing initiatives. The PMPRB has increased its capacity in Human Resources in order to better support business requirements.
Expenditure Profile
Departmental Spending Trend
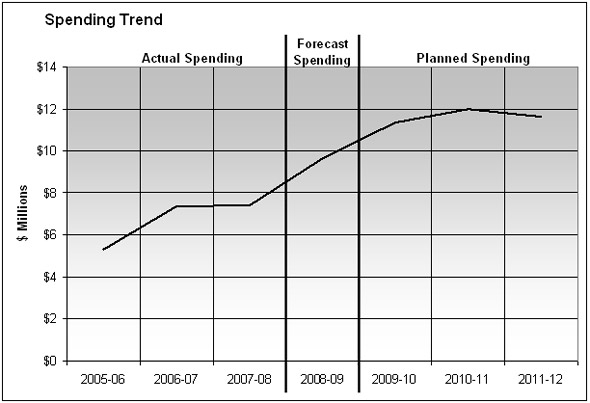
In recognition of increasing workload pressures, the PMPRB was provided program integrity funding in the amounts of $4.9 million (excluding employee benefit plans (EBP)) for 2006-2007 (in Supplementary Estimates A) and $5.0 million (including EBP) for 2007-2008 from the Treasury Board Risk Management Reserve to augment its $5 million A-base budget.
Those amounts included $4.4 million and $3.2 million for hearings for 2006-2007 and 2007-2008 respectively, which were placed in an existing Special Purpose Allotment (SPA) in the amount of $300 thousand. The ability to hold public hearings when needed is a core component of the Board’s mandate and authority. Due to the difficulty in forecasting the number and complexity of hearings in any given year, the amounts related to external hearing costs (legal counsel, expert witnesses, etc.) are placed in a Special Purpose Allotment so that they are reserved strictly for that purpose. Any unspent amount lapses at year end.
Although expenditures rose in each of 2006-2007 and 2007-2008, there were fewer hearings than anticipated. Some were settled through the submission of Voluntary Compliance Undertakings (VCUs). In addition, given the temporary nature of the funding, the staffing of some positions presented a challenge.
In anticipation of the sunsetting of the program integrity funding in 2008-2009, the PMPRB was granted additional funding in the amount of $4.7 million (excluding EBP), in addition to the core A-base of $5.8 million, to enable the PMPRB to meet its workload pressures and continue ongoing initiatives related to the delivery of its mandate.
The PMPRB expects to report expenditures of $9.6 million. Circumstances having delayed access to the additional funding until late January 2009, some positions remained unstaffed. In addition, some hearings moved into the less costly decision phase or were new hearings in the pre-hearing phase, which were also less costly.
Treasury Board approved authority to increase reference levels in Vote 35 (Program expenditures) by $5.6 million for 2009-2010, $6.2 million for 2010-2011, and $5.8 million for 2011-2012 and future years (including EBP and excluding Public Works and Government Services Canada accommodation charges).
As part of these increased resources, authority was given to increase the Special Purpose Allotment to conduct Public Hearings, in Vote 35 (Program Expenditures) by $1.9 million in 2008-2009, $2.2 million in 2009-2010, $2.8 million in 2010-2011, and $2.8 million in 2011-2012 and future years for public hearings and legal costs. This will provide assurance that the PMPRB will have the necessary resources to hold hearings even in the most exceptional years.
Voted and Statutory Items ($ thousands)
| Vote # or Statutory Item (S) |
Vote or Statutory Wording | 2008–2009 Main Estimates |
2009–20104 Main Estimates |
|---|---|---|---|
|
35 |
Program expenditures |
5,211 |
10,369 |
|
(S) |
Contributions to employee benefit plans |
631 |
989 |
|
|
Total PMPRB |
5,842 |
11,358 |
Section II — Analysis of Program Activities by Strategic Outcome
Strategic Outcome
Canadians and their health care system are protected from excessive pricing for patented medicines sold in Canada and are informed on pharmaceutical trends.
Program Activities
| Program Activity 1: Compliance and enforcement of non-excessive pricing for patented medicines | |||||
|---|---|---|---|---|---|
| Human Resources (FTEs) and Planned Spending ($ thousands) | |||||
| 2009–2010 | 2010–2011 | 2011–2012 | |||
| FTEs | Planned Spending | FTEs | Planned Spending | FTEs | Planned Spending |
| 45 | 7,045 | 45 | 7,645 | 45 | 7,645 |
| Program Activity Expected Results | Performance Indicators | Targets |
|---|---|---|
| Prices charged by Patentees for patented medicines in Canada are not excessive according to the factors of the Patent Act. | Percentage of patented medicines that are within the guidelines | 95% of patented medicines are within Guidelines. |
Program Activity Summary for PA 1:
The PMPRB is responsible for ensuring that the prices that patentees charge, the “factory-gate” price, for patented drugs sold in Canada to wholesalers, hospitals, pharmacies, or others, for human and veterinary use, are not excessive.
The PMPRB relies on voluntary compliance whenever possible since it is more effective, less time consuming, and less costly to all parties. Voluntary compliance by patentees is facilitated by published Guidelines, which are intended to assist patentees in setting prices that are not excessive by providing transparent and predictable information on how the price review will be carried out. The Guidelines are published in the PMPRB’s Compendium of Guidelines, Policies and Procedures (Compendium) and are available on the Web site http://www.pmprb-cepmb.gc.ca, under Legislation, Regulations, Guidelines.
The PMPRB reviews introductory and ongoing pricing information, as filed within 30 days of the first day of sale and thereafter semi-annually, pursuant to the Regulations, for all patented drugs sold in Canada, to ensure that they are not excessive.
The Guidelines outline the general approach to operationalizing the price determination factors in section 85 of the Act. They weredeveloped in consultation with stakeholders, including the pharmaceutical industry, federal, provincial and territorial Ministers of Health, and consumer groups (pursuant to subsection 96 (5) of the Act), and other stakeholders. The Guidelines are used by Board staff to conduct price reviews but are not binding on the Board or patentees in the context of a hearing.
Due to renewed stakeholder attention to drug prices and cost trends, and in order to respond to concerns about high introductory drug prices and other issues, the Board initiated a process in 2005, to review its Guidelines, including consulting with key stakeholders as required by the Act.
As a result of stakeholder input through consultation meetings and written submissions, significant progress in identifying issues, options and proposals for modifying the Guidelines has occurred. One final round of consultations on the final Guidelines proposals is planned for the spring of 2009. Next steps would include implementation of the revised Guidelines.
The PMPRB also plans to strengthen policy and economic analysis in order to:
- complete the review and modernization of the Guidelines;
- monitor the implementation of the revised Guidelines to ensure desired outcomes are being achieved;
- assess the impact on Canadian price trends; and
- enhance monitoring and impact assessment of legislative, regulatory and policy changes, both in Canada and in comparator countries, as well as changes in pharmaceutical innovation.
It is essential that price reviews and investigations be conducted in a timely fashion so as to ensure excessive prices are reduced with the least delay possible. In order to meet these objectives, the PMPRB will:
- strengthen internal scientific capacity and expand the membership of the Human Drug Advisory Panel to meet workload increases;
- enhance outreach activities to facilitate understanding by patentees of the regulatory reporting requirements and application of the Guidelines;
- undertake a process reengineering to improve the timelines and effectiveness of the price review process and investigations.
Another component of Program Activity 1 is the conduct of hearings into the prices of patented drugs. A significant increase in the number of hearings has created workload pressure on the Secretary to the Board, who manages the hearing process and provides Registrar Services, and the Legal Services Branch that provides internal legal research and advice and provides direction to external counsel in the prosecution function in hearings. The PMPRB will increase capacity in these areas to ensure timeliness of hearings, as required by statute.
| Program Activity 2: Pharmaceutical trends | |||||
|---|---|---|---|---|---|
| Human Resources (FTEs) and Planned Spending ($ thousands) | |||||
| 2009–2010 | 2010–2011 | 2011–2012 | |||
| FTEs | Planned Spending | FTEs | Planned Spending | FTEs | Planned Spending |
| 13 | 1,593 | 13 | 1,593 | 13 | 1,593 |
| Program Activity Expected Results | Performance Indicators | Targets |
|---|---|---|
| Stakeholders are more aware of pharmaceutical trends and cost drivers. |
|
|
Program Activity Summary for PA 2:
The PMPRB is responsible for ensuring that Canadians are informed about pharmaceutical trends. This program activity includes reporting annually on the PMPRB’s major activities, on analyses of pharmaceutical prices and price trends, and Research and Development (R&D) expenditures as reported by patentees. Under the Patent Act, the PMPRB monitors and reports on R&D spending in Canada, but has no regulatory authority over the amount or type of research spending by patentees.
Patentees are required, under the Regulations, to report their total sales of drugs and R&D expenditures in Canada, both patented and non-patented, to the PMPRB. Patentees are also required to submit detailed price or revenue and volume information, by class of customer5 in each province and territory, for patented drugs sold in Canada. This information allows the PMPRB to analyze trends in sales, utilization and prices among patented drugs. Results of this analysis are presented in the PMPRB’s Annual Report.
The PMPRB maintains the Patented Medicine Price Index (PMPI) to monitor trends in prices of patented medicines sold in Canada based on the Average Price across Canada. It is updated annually using price and sales information reported by patentees.
In 2001, the Minister directed the PMPRB, under section 90 of the Act, to play a role in the National Prescription Drug Utilization Information System (NPDUIS). This initiative involves preparing critical analyses of prescription drugs so that Canada’s health system has more comprehensive and accurate information on how prescription drugs are being used and on sources of cost increases. The Canadian Institute for Health Information (CIHI) and the PMPRB are partners in the NPDUIS. A steering committee, comprised of representatives of F/P/T public drug plans6, advises the PMPRB on the development of NPDUIS priorities for analytical studies.
Through the NPDUIS initiative, the PMPRB plans to produce updated editions of the Pharmaceutical Trends Overview Report (which analyses data from seven public drug plans and examines the trends of drug expenditures, price levels, per-unit costs, and drug utilization) and the New Drug Pipeline Monitor (which identifies drugs currently under development that may have an impact on F/P/T drug plan expenditures in the near future).
The NPDUIS’ future priorities include:
- the long term impact of population ageing on drug plan expenditures;
- the applicability of various international standards of drug use in the analysis of Canadian drug utilization;
- developing a cost driver methodology in the context of Canadian drug plan expenditures;
- the forecasting of drug plan expenditures;
- professional fees across jurisdictions;
- the impact of a generic launch on drug utilization; and
- price trends for non-patented prescription drugs
In order to enhance the continued relevance and timeliness of the analytical studies under the NPDUIS, the PMPRB will:
- strengthen internal capacity by staffing vacant positions; and
- ensure the priorities and results of analytical studies are relevant and timely for federal/provincial/territorial drug plan managers in terms of their decision-making needs.
| Program Activity 3: Internal Services | |||||
|---|---|---|---|---|---|
| Human Resources (FTEs) and Planned Spending ($ thousands) | |||||
| 2009–2010 | 2010–2011 | 2011–2012 | |||
| FTEs | Planned Spending | FTEs | Planned Spending | FTEs | Planned Spending |
| 18 | 2,720 | 18 | 2,745 | 18 | 2,370 |
Program Activity Summary for PA 3:
Internal Services are groups of related activities and resources that are administered to support the needs of programs and other corporate obligations of an organization. These groups are: Management and Oversight Services; Communications Services; Legal Services; Human Resources Management Services; Financial Management Services; Information Management Services; Information Technology Services; Real Property Services; Materiel Services; Acquisition Services; and Travel and Other Administrative Services. Internal Services include only those activities and resources that apply across an organization and not to those provided specifically to a program.
Section III — Supplementary Information
List of Supplementary Information Tables
The following tables are located on the Treasury Board Secretariat web site:
Sources of Non-Respendable Revenue (http://www.tbs-sct.gc.ca/rpp/st-ts-eng.asp)
Other Items of Interest
PMPRB Annual Report 2007 (http://www.pmprb-cepmb.gc.ca/English/View.asp?x=91&mp=68)
Quarterly NEWSletters (http://www.pmprb-cepmb.gc.ca/english/View.asp?x=287&mp=68)
Patentee’s Guide to Reporting (http://www.pmprb-cepmb.gc.ca/english/view.asp?x=146)
Compendium of Guidelines, Policies and Procedures
(http://www.pmprb-cepmb.gc.ca/english/view.asp?x=1034)
Patent Act (http://laws.justice.gc.ca/en/P-4/index.html)
Patented Medicines Regulations (http://laws.justice.gc.ca/en/P-4/SOR-94-688/index.html)
1 For program activity descriptions, please see Page 5.
2 Prior to 2009-2010, Internal services costs were distributed by Program Activities 1 and 2.
3 The PMPRB has only one Strategic Outcome (SO) (see page 7), and all priorities are linked to that SO. Links to Program Activity (PA) are indicated in this column.
4 Treasury Board approved authority to increase reference levels in Vote 35 (Program expenditures) by $5.6 million for 2009-2010. This funding will allow the Patented Medicine Prices Review Board to: 1) hold necessary hearings into excessive prices in a timely manner, as required by the Patent Act; 2) address the increase in workload anticipated as significantly more patented drug products sold by generic companies come under its jurisdiction; 3) modernize its Excessive Price Guidelines, to better deal with new forms of incremental pharmaceutical innovation; 4) replace its outdated data management system, which houses and manipulates all of the confidential sales and price information filed by patentees; 5) strengthen its policy and economic analysis capacity to better anticipate and respond to emerging and evolving developments in the regulatory environment and the pharmaceutical sector; and 6) maintain the timeliness of ongoing price reviews and investigations.