ARCHIVED - Health Canada
 This page has been archived.
This page has been archived.
Archived Content
Information identified as archived on the Web is for reference, research or recordkeeping purposes. It has not been altered or updated after the date of archiving. Web pages that are archived on the Web are not subject to the Government of Canada Web Standards. As per the Communications Policy of the Government of Canada, you can request alternate formats on the "Contact Us" page.
2008-2009
Reports on Plans and Priorities
Health Canada
The Honourable Tony Clement
Minister of Health and Minister for the Federal Economic Development Initiative for Northern Ontario
Table of Contents
Section 1 - 2008 - 2009 Departmental Overview
- Program Activity Architecture (PAA) Crosswalk
- Voted and Statutory Items Displayed in the Main Estimates
- Planned Spending and Full-time Equivalents
- Summary Information
- Departmental Priorities
- Program Activities by Strategic Outcome
- Departmental Plans and Priorities
Section 2 - Analysis of Program Activities by Strategic Outcome
- Accessible and Sustainable Health System Responsive to the Health Needs of Canadians
- Program Activity - Canadian Health System
- Program Activity - Canadian Assisted Human Reproduction
- Program Activity - International Health Affairs
- Access to Safe and Effective Health Products and Food and Information for Healthy Choices
- Program Activity - Health Products
- Key Program / Service: Pharmaceutical Human Drugs
- Key Program / Service: Medical Devices
- Reduced Health and Environmental Risks from Products and Substances, and Healthy, Sustainable Living and Working Environments
- Program Activity - Sustainable Environmental Health
- Better Health Outcomes and Reduction of Health Inequalities Between First Nations and Inuit and Other Canadians
-
Program Activity - First Nations and Inuit Health Programming and Services
- Key Program / Service: First Nations and Inuit Community Programs
- Key Program / Service: First Nations and Inuit Health Protection and Public Health
- Key Program / Service: First Nations and Inuit Primary Care
- Key Program / Service: Non-Insured Health Benefits (Supplementary) for First Nations and Inuit
- Key Program / Service: Governance and Infrastructure Support to First Nations and Inuit Health System
- First Nations and Inuit Programs and Services and Web Links
Section 3 - Supplementary Information
- Table 1: Departmental Links to the Government of Canada Outcomes
- Table 2: Sustainable Development Strategy
Section 4 - Other Items of Interest
- Health Canada's Regional Operations - An Overview
- Regionally-Responsive Service Delivery
- Outreach and Information
- Collaboration with Regional Partners
- Advancing the Science Agenda
- Science Advice
- Science Management
- Science Promotion
2008 - 2009 Departmental Overview
1. Minister's Message

It is my pleasure to present to you Health Canada's 2008-2009 Report on Plans and Priorities. Health Canada is committed to making this country's population among the healthiest in the world.
Health continues to be important for the Government of Canada. As Minister of Health, I am pleased to see that this report builds on the progress we made in 2007. In the last planning period, we followed through on our commitment to establish Patient Wait Times Guarantees. All ten provinces and three territories agreed to establish guarantees by 2010, supported by more than $600 million in federal funding. In the coming period, we will support provinces and territories as they work to reach their goals.
In 2007, we successfully created the Mental Health Commission of Canada. This Government committed $55 million over five years, which will enable the Commission to serve as the national focal point for mental health and mental illness. In 2008-09, the Government of Canada will support the Commission as it builds the national approach we need to improve the quality of life for Canadians and their families dealing with mental illness.
Canadians have told us they are concerned about the safety of the food and products they buy and use, and our government shares these concerns. The 2007 Speech from the Throne committed to introducing measures on food and product safety to ensure that families can have confidence in the quality and safety of what they buy. That is why this government announced the Food and Consumer Safety Action Plan, a comprehensive set of proposed new measures that will make Canadians safer by introducing tougher federal legislation and regulation of food, health and consumer products.
Our government is keenly aware of the important link between health and the environment. As highlighted in the 2007 Speech from the Throne, Health Canada will continue to work with its partners to continue carrying out health and environment initiatives such as the Chemicals Management Plan, Clean Air Agenda and National Water Strategy.
On a related note, our Government will continue efforts to improve clean drinking water in Aboriginal communities. We will also work to improve First Nations and Inuit health outcomes by continuing to develop the first-ever Patient Wait Times Guarantees for federally funded health services in First Nations communities. We will continue working to improve the overall health of First Nations and Inuit people, support the availability of and access to quality health programs and services, and develop capacity to support delivery of health services by First Nations and Inuit communities.
This planning period will also include continuing efforts to inform and protect Canadian families against the perils of controlled and illicit substances. We will work toward further driving down the prevalence of smoking among Canadian youth and implementing the National Anti-Drug Strategy to reduce both the supply and demand of illicit substances to safeguard our communities and benefit individual health.
In striving to get results for Canadians on all health matters, sound science is the basis of all of our Department's decisions and actions. As a result, Health Canada is working to develop a Science and Technology Strategy (S&T) that will strengthen the role of S&T within the Department, position Health Canada's S&T efforts to support government-wide activities, and allow the Department to develop a consistent set of science priorities over the next five to ten years.
This report is forward looking and responsive to Canadians' health needs. Health Canada remains committed to working to protect the health and safety of Canadians.

Tony Clement
Minister of Health, and Minister for the Federal
Economic Development Initiative for Northern Ontario
2. Management Representation Statement
I submit for tabling in Parliament the 2008-09 Report on Plans and Priorities for Health Canada.
This document has been prepared based on the reporting principles contained in the Guide to the Preparation of Part III of the 2008-09 Estimates: Reports on Plans and Priorities and Departmental Performance Reports:
- it adheres to the specific reporting requirements outlined in the Treasury Board of Canada Secretariat guidance;
- it is based on the department's strategic outcomes and program activities that were approved by the Treasury Board;
- it presents consistent, comprehensive, balanced, and reliable information;
- it provides a basis of accountability for the results achieved with the resources and authorities entrusted to it; and
- it reports finances based on approved planned spending numbers from the Treasury Board of Canada Secretariat.
Morris Rosenberg
Deputy Minister of Health
Health Canada's 2008-2009 Report on Plans and Priorities
The Report on Plans and Priorities is Health Canada's key planning and priority-setting document. It provides an overview of the work of the Department, the external and internal challenges it faces and the expected results that will be delivered to Canadians in the coming three years. It also summarizes planned expenditures and outlines performance measurement activities.
3. Raison d'être
Health Canada was established in 1996 through the Department of Health Act. It is the federal department responsible for helping Canadians maintain and improve their health. Health Canada is committed to improving the lives of all Canadians and to making this country's population among the healthiest in the world as measured by longevity, lifestyle and effective use of the public health care system.
4. Organizational Information
Health Portfolio Overview
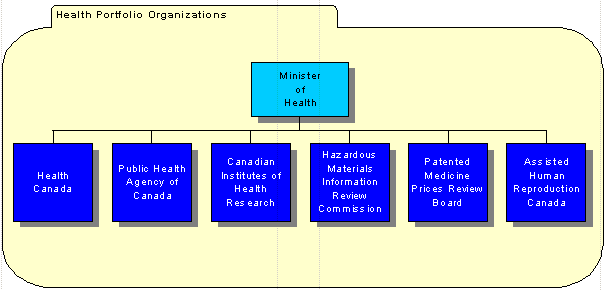
The Minister of Health is supported by the Health Portfolio which comprises Health Canada, the Public Health Agency of Canada, the Canadian Institutes of Health Research, the Hazardous Materials Information Review Commission, the Patented Medicine Prices Review Board, and the Assisted Human Reproduction Canada. Each member of the Portfolio prepares its own Report on Plans and Priorities.
The Health Portfolio consists of approximately 12,000 full-time equivalents and an annual budget of over $3.8 billion. Health Canada provides policy leadership and coordination among portfolio members as appropriate to ensure a coherent approach to addressing health priorities.
Health Canada fulfills its mission by supporting activities that preserve Canada's health care system; enhance and protect the health of Canadians; and communicate with Canadians about health promotion, disease prevention and safety messaging through the following roles and responsibilities:
- Leader/partner through the administration of the Canada Health Act, which embodies national principles to ensure a universal and equitable publicly-funded health care system, and through collaboration with provincial and territorial partners and stakeholders on reform initiatives;
- Funder through policy support for the federal government's Canada Health Transfer to provinces and territories, the transfer of funds to First Nations and Inuit organizations and communities for community health services delivery, and grants and contributions to various organizations to help meet Health Canada's objectives;
- Guardian/regulator through legislation and regulation of drugs and medical devices, consumer products and food, pesticides, chemicals, nuclear and radiological safety, illicit drugs, and through the use of science to assess health risks to Canadians and put into place mechanisms to mitigate these risks;
- Service provider through the provision of health services to First Nations and Inuit (public health and community health programs on-reserve and in Inuit communities outside of the territories, non-insured health benefits coverage regardless of residence, select primary care services on-reserve in remote and isolated areas where no provincial services are readily available); and,
- Information provider through the generation and sharing of knowledge and information on which personal decision-making, regulations and standards, and innovation in health rely.
Health Canada: A Partner in an Interwoven Community of Stakeholders
The responsibility for promoting, protecting and improving the health of Canadians does not rest solely with a specific level of government, the health professions or Canadians themselves. The responsibility rests within an interwoven community of collaborating stakeholders that each contributes to this goal. Outside of the Health Portfolio, Health Canada frequently relies on collaboration with many other groups and organizations, including:
- provinces and territories - who bear primary responsibility for health care administration and delivery, and have their own roles in health protection and promotion. A strong relationship with provincial and territorial counterparts is a critical factor in achieving our mandate;
- Aboriginal organizations - at the national, regional and local levels. To set priorities, deliver programs and services effectively, it is essential to work with these organizations in several ways, from shaping overall policy to devolving program responsibility where local capacity is in place;
- national health organizations - in recent years, the institutional landscape in health has evolved with the establishment of new organizations to advance key priorities in health care, typically involving shared governance between the federal and provincial and territorial governments;
- scientific organizations - both nationally and internationally, ranging from the universities and academic research institutes and networks to specialized agencies such as the World Health Organization; and,
- stakeholders - from industry groups to health charities to public policy advocates to health professionals such as physicians and nurses, the range of stakeholders relating to the Department is enormous. Some of the Minister's and Department officials' time will be given to stakeholder relations; they are a source of ideas, advice, criticism, support, information, and pressure for action.
Health Canada: A Science-based Department
Health Canada depends on sound science and research to fulfill its regulatory and policy mandate. In 2006-07, the Department spent an estimated $331million on science and technology (S&T), consisting of $63 million on research and development (R&D) and $268 million on related scientific activities (RSA). Of the $331 million S&T expenditures, $307 million was for internal S&T activities and $24 million was for S&T activities conducted outside of Health Canada.
Because of the scope of Health Canada's RSA and R&D responsibilities, the Department depends on the work of scientists in a range of fields, including the natural and life sciences and social sciences. These scientists conduct the RSA and R&D that the Department needs to address science challenges in support of its mandate. In 2006-07, Health Canada employed 2,900 science personnel, one quarter of whom undertake R&D with the remainder undertaking RSA activities (e.g. using science for policy and regulatory decision making). Departmental scientists work at the Department's headquarters and in various laboratories located across the country.
Departmental scientists perform a wide variety of essential functions, they provide leading-edge science, sound policy research, and effective program and service development; provide knowledge to Canadians, health care workers and other public and private sector stakeholders to enable them to make sound choices to protect health and the environment, through research, evaluation, risk assessment, risk management and surveillance; monitor and research the health threats from environmental factors such as toxic substances, air and water pollution, climate change; and foster sound decision-making and policy-development by all stakeholders to help reduce health risks.
Health Canada is working to further strengthen and enhance its science activities. For example, the Department is working to develop a S&T Strategy that will strengthen the role of S&T within the Department, ensure responsiveness to current and future needs of Canadians, strategically position Health Canada's S&T efforts relative to government-wide S&T activities, and allow the Department to develop a consistent set of science priorities over the next five to ten years.
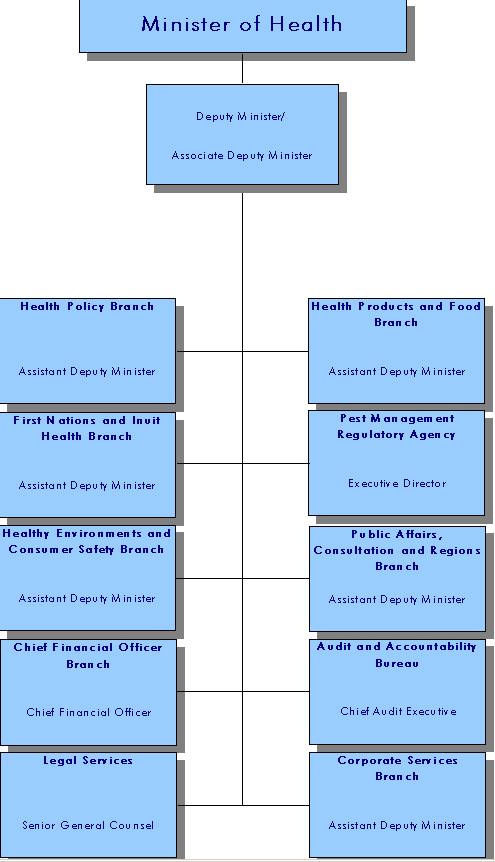
Health Canada's Organizational Structure
Health Canada has approximately 8,700 full-time equivalents across Canada. Roughly one third work outside the National Capital Region, providing close proximity to communities and clients, thereby facilitating the design and delivery of programs and services that are responsive to local issues, priorities, and needs of the diverse regions of Canada.
Health Canada's Organizational Chart
Health Canada At A Glance
| Type of Organization | Federal department, funded by Parliament |
| Mission | To help the people of Canada maintain and improve their health |
| Vision | Improving the lives of all Canadians and making this country's population among the healthiest in the world as measured by longevity, lifestyle and effective use of the public health care system |
| Strategic Outcomes |
|
| Government of Canada Outcomes Directly Supported | Healthy Canadians A safe and secure world through international co-operation A clean and healthy environment |
| Enabling Legislation | Department of Health Act |
| Acts Administered | Assisted Human Reproduction Act Canada Health Act Controlled Drugs and Substances Act Food and Drugs Act Hazardous Products Act Pest Control Products Act Pesticide Residue Compensation Act Radiation Emitting Devices Act Tobacco Act Canadian Environmental Protection Act, 1999 (co-administered) |
|
Program Activities |
Canadian Health System |
| Reporting to Parliament |
Health Canada reports to Parliament through the Minister of Health. |
5. Program Activity Architecture (PAA) Crosswalk
| 2008 - 09 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ($ millions) | Canadian Health System | Canadian Assisted Human Reproduction | International Health Affairs | Health Products | Food and Nutrition | Sustainable Environmental Health | Consumer Products | Workplace Health | Substance Use and Abuse | Pesticide Regulation | First Nations and Inuit Health Programming and Services | Total |
| Health Policy, Planning and Information | 306.3 | 2.1 | 28.7 | 337.1 | ||||||||
| Health Products and Food | 186.5 | 77.9 | 264.4 | |||||||||
| Healthy Environments and Consumer Safety | 165.4 | 24.2 | 34.8 | 152.9 | 377.3 | |||||||
| Pest Control and Product Regulation | 60.5 | 60.5 | ||||||||||
| First Nations and Inuit Health | 2,156 | 2,156 | ||||||||||
| Total | 306.3 | 2.1 | 28.7 | 186.5 | 77.9 | 165.4 | 24.2 | 34.8 | 152.9 | 60.5 | 2,156 | 3,195.3 |
Note: These figures represent planned spending adjustments to main estimates.
6. Voted and Statutory Items Displayed in the Main Estimates
| Vote or Statutory Item ($ millions) | Truncated Vote or Statutory Wording | 2008-09 Main Estimates | 2007-08 Main Estimates |
|---|---|---|---|
| 1 | Operating expenditures | 1,661.6 | 1,691.0 |
| 5 | Capital expenditures | 60.0 | 0.0 |
| 10 | Grants and contributions | 1,358.1 | 1,225.9 |
| (S) | Minister of Health - Salary and motor car allowance | 0.1 | 0.1 |
| (S) | Contributions to employee benefit plans | 110.9 | 111.3 |
| Total Department | 3,190.7 | 3,028.3 |
The difference between the 2008-09 Main Estimates and the 2007-08 Main Estimates is due to various increases and decreases to the reference levels, of which some of the major increases are related to: the Canadian Institute for Health Information, the National Anti-Drug Strategy, the Mental Health Commission of Canada, the implementation of the Patient Wait Times Guarantee Pilot Project Fund, and the yearly growth of the Indian Envelope.
7. Planned Spending and Full-time Equivalents
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Canadian Health System | 225.3 | 306.3 | 304.9 | 291.7 |
| Canadian Assisted Human Reproduction | 2.1 | 2.1 | 2.1 | 2.1 |
| International Health Affairs | 28.8 | 28.7 | 28.4 | 28.1 |
| Health Products | 222.6 | 222.4 | 219.8 | 216.5 |
| Food and Nutrition | 77.5 | 79.3 | 77.6 | 77.2 |
| Sustainable Environmental Health | 147.9 | 166.8 | 181.1 | 157.9 |
| Consumer Products | 24.0 | 24.6 | 25.1 | 24.8 |
| Workplace Health | 48.4 | 48.3 | 46.8 | 46.3 |
| Substance Use and Abuse | 134.0 | 152.9 | 151.5 | 148.0 |
| Pesticide Regulation | 60.1 | 67.4 | 68.1 | 68.2 |
| First Nations and Inuit Health Programming and Services | 2,126.9 | 2,161.5 | 2,206.8 | 2,039.1 |
| Budgetary Main Estimates (gross) | 3,097.6 | 3,260.3 | 3,312.2 | 3,099.9 |
| Less: Respendable revenue | 69.3 | 69.6 | 69.6 | 69.6 |
| Total Main Estimates | 3,028.3 | 3,190.7 | 3,242.6 | 3,030.3 |
| Adjustments:(1) | ||||
| Supplementary Estimates: | ||||
| Funding to administer programs that support the delivery of health services to First Nations and Inuit populations | 74.8 | |||
| Providing funding to the Canadian Institute for Health Information through the Health Information Initiative | 56.8 | |||
| Funding for program integrity pressures: Natural Health Products and Public Service Health Program | 25.9 | |||
| Funding to implement the Prevention and Treatment Action Plans of the National Anti-Drug Strategy focussing on educating, marketing and treatment initiatives (horizontal item) | 10.6 | |||
| Funding for the Indian Residential Schools Resolution Health Support Program, a component of the Settlement Agreement to address the Indian Residential Schools Legacy | 7.5 | |||
| Funding for the Phase II of the Response to Bovine Spongiform Encephalopathy (BSE) in the areas of Risk Assessment and Targeted Research | 5.9 | |||
| Contribution towards the start-up of the Mental Health Commission of Canada | 5.5 | |||
| Funding related to government advertising programs (horizontal item) | 5.1 | |||
| Funding for pest management controls for the evaluation and registration of new low-risk pesticides and increasing the use of evidence-based risk assessments | 3.8 | |||
| Funding to support the implementation of a Patient Wait Times Guarantee Pilot Project Fund to provide an accessible and sustainable health system responsive to the needs of Canadians | 2.6 | |||
| Funding for enhanced enforcement activities relating to the National Anti-Drug Strategy that are aimed at reducing the supply and demand for illicit drugs (horizontal item) | 1.9 | |||
| Transfer from National Defence - For the participation in the Chemical, Biological, Radiological, and Nuclear Research and Technology Initiative | 0.8 | |||
| Funding to undertake projects related to the development and application of biotechnology (Canadian Biotechnology Strategy) (horizontal item) | 0.7 | |||
| Funding related to the assessment, management and remediation of federal contaminated sites (horizontal item) | 0.6 | |||
| Funding for the environmental clean up of the Sydney Tar Ponds and Coke Ovens Sites in the Muggah Creek Watershed | 0.5 | |||
| Funding for activities that are essential to the continued implementation of the Public Service Modernization Act (horizontal item) | 0.4 | |||
| Funding in support of the Federal Accountability Act to evaluate all ongoing grant and contribution programs every five years (horizontal item) | 0.4 | |||
| Transfer from the Public Health Agency of Canada - to support the public health library which, as part of the Science Library Network, provides information and document services to support research and regulatory activities | 0.3 | |||
| Funding for the costs of hosting the 12th Summit of la Francophonie in Quebec City in 2008 (horizontal item) | 0.2 | |||
| Funding to strengthen the central agency regulatory function of the Treasury Board Secretariat and to establish a Centre of Regulatory Expertise (horizontal item) | 0.1 | |||
| Transfer from Canadian Heritage - for the development of Official Language Minority Communities (Interdepartmental Partnership with the Official Language Communities) | 0.1 | |||
| Transfer to the Public Health Agency of Canada - to support the Canadian Health Services Research Foundation's Community Health Nursing Study | -0.1 | |||
| Transfer to Public Service Human Resources Management Agency of Canada - to support the National Managers' Community | -0.1 | |||
| Transfer to Environment Canada - to assess the health and economic impacts of air pollution across Canada | -0.1 | |||
| Transfer to the Canadian Institutes of Health Research - to support Aboriginal youth suicide prevention research | -0.2 | |||
| Transfer to Foreign Affairs and International Trade - to provide support to departmental staff located at missions abroad | -0.3 | |||
| Transfer to the Canadian Institutes of Health Research - to provide funding and support for grants and awards through the Open Grants competition | -0.3 | |||
| Transfer to Indian and Northern Affairs Canada - for amendments to the self-government financial transfer agreements with ten Yukon First Nations | -0.5 | |||
| Transfer to the Public Health Agency of Canada - to adjust for the allocation of resources following the transfer of the control and supervision of the Population and Public Health Branch | -0.6 | |||
| Transfer to Justice - to implement the Prevention and Treatment Action Plans of the National Anti-Drug Strategy focussing on educating, marketing and treatment initiatives (horizontal item) | -0.6 | |||
| Less: Spending authorities available | -47.9 | |||
| Other adjustments: | ||||
| Operating budget carry forward (horizontal item) | 54.3 | |||
| Collective Agreements | 16.6 | |||
| Implementation of the Policy on Internal Audit (TB Vote 10) | 1.1 | |||
| Biotechnology Budget 99 Genomics | 4.0 | 4.0 | 4.0 | |
| Fulfilling outstanding obligations under Canadian Environmental Protection Act, 1999 | 25.0 | |||
| Newfoundland Indians - to recognize a landless band and for the registration of Newfoundland Indians | 8.1 | 8.4 | ||
| Funding for Winter Olympics - includes operations for Paralympic Games | 0.6 | 2.0 | ||
| Total Adjustments | 225.6 | 4.6 | 14.1 | 37.4 |
| Total Planned Spending (2) | 3,253.9 | 3,195.3 | 3,256.7 | 3,067.7 |
| Less: Non-respendable revenue | 8.9 | 8.9 | 8.9 | 8.9 |
| Plus: Cost of services received without charge (3) | 93.5 | 93.3 | 94.3 | 93.5 |
| Total Departmental Spending | 3,338.5 | 3,279.7 | 3,342.1 | 3,152.3 |
| Full-time Equivalents (4) | 9,178 | 9,106 | 9,141 | 8,951 |
(1) Adjustments reflect Supplementary Estimates for 2007-08 and future year approvals not reflected in the 2008-09 Main Estimates.
(2) Refer to Section 2 for an explanation by program activity of year-over-year fluctuations.
(3) Includes the following services received without charge: accommodation charges (Public Works and Government Services Canada); Contributions covering employers' share of employees' insurance premiums and expenditures (Treasury Board Secretariat); Workers' Compensation (Human Resources and Skills Development Canada); and Legal Services
(Department of Justice Canada).
(4) Full-time equivalents reflect the human resources that the department uses to deliver its programs and services. This number is based on a calculation that considers full-time, term, casual employment, and other factors such as job sharing.
8. Summary Information
| 2008-09 | 2009-10 | 2010-11 |
|---|---|---|
| 3,195.3 | 3,256.7 | 3,067.7 |
| 2008-09 | 2009-10 | 2010-11 |
|---|---|---|
| 9,106 | 9,141 | 8,951 |
| Name | Type |
|---|---|
| 1.Contributing to the improvement of the health of Canadians | Ongoing |
| 2.Reducing the risks to the health of the people of Canada | Ongoing |
| 3.Working with others to strengthen the efficiency and effectiveness of the publicly- funded health care and health system | Ongoing |
| 4.Strengthening accountability to Parliament and the public | Ongoing |
Program Activities by Strategic Outcome
| Planned Spending | |||||
|---|---|---|---|---|---|
| ($ millions) | Expected Results | 2008-09 | 2009-10 | 2010-11 | Contributes to the following priority |
| Strategic Outcome # 1: Accessible and Sustainable Health System Responsive to the Health Needs of Canadians | Priorities 1, 2, 3, and 4 | ||||
| Program Activity: Canadian Health System |
|
306.3 | 304.9 | 291.8 | |
| Program Activity: Canadian Assisted Human Reproduction |
|
2.1 | 2.1 | 2.1 | |
| Program Activity: International Health Affairs |
|
28.7 | 28.4 | 28.2 | |
| Strategic Outcome # 2: Access to Safe and Effective Health Products and Food and Information for Healthy Choices | Priorities 1, 2, and 4 | ||||
| Program Activity: Health Products |
|
186.5 | 184.1 | 180.9 | |
| Program Activity: Food and Nutrition |
|
77.9 | 76.3 | 76 | |
| Strategic Outcome # 3: Reduced Health and Environmental Risks from Products and Substances, and Sustainable Living and Working Environments | Priorities 1, 2 and 4 | ||||
| Program Activity: Sustainable Environmental Health |
|
165.4 | 179.9 | 177.6 | |
| Program Activity: Consumer Products |
|
24.2 | 24.6 | 26.6 | |
| Program Activity: Workplace Health |
|
34.8 | 34.1 | 32.2 | |
| Program Activity: Substance Use and Abuse |
|
152.9 | 151.5 | 148.2 | |
| Program Activity: Pesticide Regulation |
|
60.5 | 61.2 | 61.3 | |
| Strategic Outcome # 4: Better Health Outcomes and Reduction of Health Inequalities Between First Nations and Inuit and Other Canadians | Priorities 1, 2, 3, and 4 | ||||
| Program Activity: First Nations and Inuit Health Programming and Services | Strengthened community programs; better health protection; improved primary health care; and access to non-insured health benefits to contribute to improved health status of First Nations and Inuit individuals, families and communities | 2156 | 2209.6 | 2042.8 | |
Note: Figures include amounts for other departmental and regional infrastructure costs supporting program delivery.
9. Departmental Plans and Priorities
Our Operating Environment
Health Canada must respond to an ever-changing operating environment, one becoming more complex due to the rapid evolution of technology and the increasingly interconnected global community and marketplace. Protecting the health of Canadians in the face of these rapid changes becomes more challenging as we face many critical issues in Canada including demographic changes and stresses to our health care system. There are several key themes that will likely dominate the complex policy landscape for Health Canada for this planning period and beyond.
The Safety of Food, Health Products and Consumer Products
Recent incidents related to food, health and consumer products highlight gaps in protecting the health and safety of Canadians. The number of products and producers is vast and growing. Globalization has expanded supply chains across multiple boundaries, blurring the lines between imported and domestic products. Non-traditional suppliers in developing and emerging economies are now major players. Increasing the pace of innovation is critical for companies to survive. Consumers have more sources of information and have high expectations for industry and government.
Health and the Environment
On a daily basis, Canadians are exposed to a cocktail of environmental contaminants which pose risks to human health. Exposure to environmental contaminants is linked to a spectrum of health problems including cancers, asthma and other respiratory illnesses, as well as cardiovascular disease and developmental disorders in children. In addition, certain populations (children and those living in rural and remote populations) have been identified as being particularly at risk to health problems associated with exposure to environmental contaminants. While the Government of Canada has taken action to improve the environment, for example with the Chemicals Management Plan, there continue to be gaps with respect to understanding and reducing the risks that environmental contaminants may pose to human health (e.g. helping foster awareness in Canadians so that they can make wise choices to reduce their own health risks, better understanding what contaminants are finding their way into the Canadian population, and building a scientific evidence base that will answer questions about how exposure to contaminants may contribute to disease).
Shifting Demographics and the Health of Vulnerable Populations in Canada: Youth, Seniors and First Nations and Inuit
Climbing child obesity rates coupled with risky health behaviours lead many experts to believe that this may negatively impact Canada's youngest generation. By 2050, well over one-third of Canadians will be seniors (65+ years of age), above the forecasted Organisation for Economic Co-operation and Development (OECD) average. The Aboriginal population is younger and growing at twice the rate of the total population. Additional challenges remain as these populations face unique health challenges. For example, while Aboriginal Canadians are living longer, the status of their health continues to lag behind that of other Canadians.
The Capacity and Efficiency of the Health Care System
The sustainability of Canada's publicly-funded universal health care system is of great importance to Canadians. Demographic trends show that Canada's aging population could have large implications for the health care system. Looking at ways to meet the growing demand for health care as well as strengthening health promotion and prevention activities will be key to ensuring its sustainability.
Evolving Threats to Health from Infectious Disease, Both New and Re-emerging
With the rapid movement of people and consumer products around the world, potential infectious diseases can be within our borders in a matter of hours. Known diseases such as tuberculosis are re-emerging or are continuing to spread in developing countries. Prevention, preparedness and response for infectious diseases will remain a key necessity for safeguarding the health of Canadians.
The Opportunities and Risks for the Health of Canadians from the Rapid Pace of Technological Development
New technologies are redefining all sectors, with the medical field poised to harness major benefits due to the application of biotechnology in treatment and diagnostics. New technologies are also finding their way into consumer markets, posing potential new risks to consumers and challenging regulatory agencies. Alternative energy technologies and new fuels are becoming more economical and popular worldwide, though their health and environmental impacts are not always considered.
Departmental Priorities
Given the environment in which we operate and our key areas of interest for the planning period, Health Canada has established four departmental priorities to guide our responses to the challenges and opportunities facing the health of Canadians. These priorities reflect the Government of Canada's direction and commitments and serve as the drivers of our strategic outcomes and expected results for Canadians as explained in Section II.
Our four departmental priorities are as follows:
- Contribute to the improvement of the health of Canadians
- Reduce the risks to the health of the people of Canada
- Work with others to strengthen the efficiency and effectiveness of the publicly-funded health care and health system
- Strengthen accountability to Parliament and the public
Priority #1: Contribute to the improvement of the health of Canadians (ongoing)
Linkages to Strategic Outcomes:
Strategic Outcome #1 - Accessible and sustainable health system responsive to the health needs of Canadians
Strategic Outcome #2 - Access to Safe and Effective Health Products and Food and Information for Healthy Choices
Strategic Outcome #3 - Reduced Health and Environmental Risks from Products and Substances, and Healthy, Sustainable Living and Working Environments
Strategic Outcome #4 - Better Health Outcomes and Reduction of Health Inequalities Between First Nations and Inuit and Other Canadians
In order to meet the Government of Canada's objective to ensure healthy Canadians, Health Canada will continue to collaborate with provinces and territories and other partners to improve health system performance and health outcomes. For example, pilot projects on Patient Wait Times Guarantees are expected to yield lessons that will facilitate best outcomes for patients needing health services.
We will work to enable Canadians to make informed decisions and healthy choices about food and nutrition. We will also move forward with a number of initiatives to reduce and prevent tobacco consumption, and to combat alcohol and drug abuse. For example, Health Canada will work to increase the number of smokers who quit and develop national alcohol guidelines to support a moderate and sensible use of alcohol.
Health Canada will continue to collaborate with its partners to reduce the health gap that exists between Aboriginal and non-Aboriginal Canadians. We will do this through continued implementation of health programs, initiatives and strategies in First Nations and Inuit communities that aim to improve health outcomes in areas such as children and youth, chronic disease (e.g. diabetes) and injury prevention, and mental health and addictions.
Priority #2: Reduce the risks to the health of the people of Canada (ongoing)
Linkages to Strategic Outcomes:
Strategic Outcome #1 - Accessible and sustainable health system responsive to the health needs of Canadians
Strategic Outcome #2 - Access to Safe and Effective Health Products and Food and Information for Healthy Choices
Strategic Outcome #3 - Reduced Health and Environmental Risks from Products and Substances, and Healthy, Sustainable Living and Working Environments
Strategic Outcome #4 - Better Health Outcomes and Reduction of Health Inequalities Between First Nations and Inuit and Other Canadians
For the planning period, Health Canada will work with international partners to strengthen responses to possible global health threats to reduce risks to the health of Canadians.
As highlighted in the 2007 Speech from the Throne and in the Food and Consumer Safety Action Plan announced in December 2007, Health Canada will introduce measures on food and product safety to ensure that families have confidence in the quality and safety of what they buy. We will modernize our system to better inform and protect Canadians by legislating tougher federal government regulation of consumer, food and health products that is designed to prevent, and not merely react to, problems. We will also ensure better safety information for consumers and guidance to industries.
Health Canada will work to identify, assess and manage health risks from environmental factors, such as air pollutants and chemicals. We will also implement a new policy to address drinking water quality in First Nations communities to ensure access to safe drinking water.
Priority #3: Work with others to strengthen the efficiency and effectiveness of the publicly-funded health care and health system (ongoing)
Linkages to Strategic Outcomes:
Strategic Outcome #1 - Accessible and sustainable health system responsive to the health needs of Canadians
Strategic Outcome #4 - Better Health Outcomes and Reduction of Health Inequalities Between First Nations and Inuit and Other Canadians
Strengthening the efficiency and effectiveness of the publicly-funded health care system is essential to ensuring that it meets the needs of all Canadians. Health Canada will continue to work closely with provincial and territorial governments, as well as health organizations and other stakeholder groups to examine new and innovative ways to improve the sustainability and
responsiveness of the health system so that Canadians have timely access to appropriate and quality care. This includes efforts to improve the delivery of health care in First Nations and Inuit communities as well as the integration of health programs and services to better meet their needs.
Priority #4: Strengthen accountability to Parliament and the public (ongoing)
Linkages to Strategic Outcomes:
Strategic Outcome #1 - Accessible and sustainable health system responsive to the health needs of Canadians
Strategic Outcome #2 - Access to Safe and Effective Health Products and Food and Information for Healthy Choices
Strategic Outcome #3 - Reduced Health and Environmental Risks from Products and Substances, and Healthy, Sustainable Living and Working Environments
Strategic Outcome #4 - Better Health Outcomes and Reduction of Health Inequalities Between First Nations and Inuit and Other Canadians
Health Canada will continue to work towards improving accountability to Parliament and the public, enhancing the transparency of its decision-making and improving communication with Canadians. Health Canada recognizes the need for appropriate performance measurement to evaluate its work and report to Canadians.
Our Operating Priorities
Health Canada will continue to transform its human resources services in an effort to address the on-going challenges that it faces in the recruitment and retention of qualified staff, particularly in the areas of science and technology. These challenges, which result in part from skills shortages in related areas of expertise and an ever-changing marketplace, make it difficult for the Department to keep up with rapidly evolving trends and knowledge in these areas, and to maintain the relevancy of related legislation and regulations. We will continue to implement innovative approaches to human resources services, including targeted recruitment and retention strategies, and various approaches to address healthy workplace enablers, such as continuing education, professional development and flexible hours of work.
In addition, Health Canada will develop a long-term laboratory strategy to ensure that its laboratory facilities are aligned with its business processes and to identify opportunities to establish partnerships with other federal departments, provincial and territorial governments, and private sector stakeholders. The immediate focus will be on evaluating existing laboratory facilities in order to identify those that require refurbishing. Similarly, we will develop a long-term office accommodation strategy to ensure that the Department's office accommodation and other non-laboratory facilities are also aligned with its business processes. Both strategies will be based on the overall Real Property Management Framework which the Department is currently developing.
Health Canada will also further integrate human resources planning into its business planning processes, with the objective of achieving full integration by 2009-10, a commitment which is highlighted in the Department's response to Public Service Renewal. The integration of strategic, operational and human resources planning will link human resources opportunities, risks and priorities directly to operational business objectives, which in turn flow from the Department's strategic outcomes.
Finally, Health Canada will continue to work with Health Portfolio partners to transform other internal services in support of more efficient program delivery, with the first focus being on an enterprise IT approach. We will continue our involvement in the Shared Services Initiative, following the recent move of some of our IT Helpdesk support services to Public Works and Government Services Canada (PWGSC), including operational alignment for network services. We will also explore the possibility of moving our e-mail services and other departmental distributed computing environment services to PWGSC. As well, we will continue to implement management improvement initiatives as necessary, including in the areas of project management, and financial management and control. Health Canada is also currently exploring with the Canada School of Public Service, the feasibility of the department providing certain financial management services to the School. In addition, we will continue to apply the Management Accountability Framework (MAF) at all management levels, and to integrate MAF expectations into the management culture of the Department. In December 2007, a new independent Departmental Audit Committee was launched, composed of a majority of members from outside of the Public Service. The governance of Health Canada's operations will be further strengthened when this Committee becomes fully functional in the coming year.
Sustainable Development
For the period of 2007 to 2010, the focal point of Health Canada's efforts to achieve sustainable development is in the implementation of the department's fourth Sustainable Development Strategy (SDS). Departmental SDS targets will contribute to strategic outcomes of the departments in areas such as clean water, clean air, food safety, sustainable communities, and greening of operations. During this period, Health Canada will also be working with other federal departments to ensure policy and program integration efforts across government are properly taking social elements, such as the determinants of health, into consideration. For more information on Health Canada's Sustainable Development Strategy, please see Table 2 in Section 3 (p.77) of this Report.
Analysis Of Program Activities By Strategic Outcome
Strategic Outcome: Accessible and Sustainable Health System Responsive to the Health Needs of Canadians
Under this program activity, we strive to develop effective policy responses to a range of priority, emerging and cross-cutting issues that impact the health of Canadians. There are many governmental and non-governmental organizations working towards improved health outcomes in Canada, including provincial and territorial departments of health, professional organizations, research organizations, the program branches of Health Canada, and the agencies within the federal Health Portfolio. Our objective is to promote the national coordination and development of a strong, shared knowledge base to address health and health care priorities for all Canadians. We also aim to facilitate health system adaptation to changes in technology, society, industry, and the environment, so that Canadians will continue to be protected from heath risks, have access to quality health care, and gain positive health benefits from information and innovation.
Serving as a regulator, a trustee, a partner, an enabler, an innovator, a knowledge broker, and a proponent of transparency, we help support the sustainability of Canada's health system. To Canadians, sustainability means the availability, maintenance and advancement of key attributes of Canada's publicly funded health care system such as accessibility, efficiency and effectiveness. In the face of shifting and growing health system demands, we develop policies to respond to these changes in supporting a sustainable health system for Canadians.
The following program activities help support the Strategic Outcome entitled "Accessible and sustainable health system responsive to the health needs of Canadians":
- Canadian Health System
- Canadian Assisted Human Reproduction
- International Health Affairs
Program Activity - Canadian Health System
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Net expenditures | 272.2 | 306.3 | 304.9 | 291.7 |
| FTEs | 415 | 415 | 414 | 412 |
| Notes: The increase in forecast/planned expenditures from 2007-08 to 2008-09 is due mainly to the reprofile of funding for the Canadian Strategy for Cancer Control as well as an increase in the funding level for the establishment of the Mental Health Commission and the implementation of the Patient Wait Times Guarantee Pilot Project Fund. The
decrease in planned expenditures from 2009-10 to 2010-11 is mainly due to the sunset of the Patient Wait Times Guarantee Pilot Project Fund. Figures include an amount for departmental and regional infrastructure costs in support of program delivery. |
||||
Under this program activity, we provide strategic policy advice on health care issues such as improved access, quality and integration of health care services to better meet the health needs of Canadians wherever they live or whatever their financial circumstances. This objective is pursued mindful of long-term equity, sustainability and affordability considerations and in close collaboration with provinces and territories, health professionals, administrators, other key stakeholders, and citizens.
We are achieving this objective by investing in the health system and in health system renewal. This includes: working with provinces and territories to reduce wait times for essential services and ensure that the principles of the Canada Health Act are respected, developing health information and health measures for Canadians, and meeting the health and health access needs of specific groups including women and official language minority communities.
| Expected Results | Performance Indicators |
|---|---|
|
|
| Description: Through this program, Health Canada helps shape policy and programs related to health care renewal that benefit all Canadians through better access to quality health care services. We provide policy leadership on issues related to broader health care system renewal, such as improving access to quality health care, increasing the supply and improving the mix of health care professionals, reforming primary health care and enhancing the accountability of the health care system to Canadians. In the area of Pharmaceuticals Management, we identify the key policy issues, trends and implications with respect to pharmaceuticals access, affordability and equity; safety, effectiveness and appropriate prescribing and utilization, as well as drug prices, and the appropriate role of pharmaceuticals in health care. With provincial and territorial (P/T) partners, we work to advance federal, provincial and territorial (F/P/T) policy and mechanisms for better pharmaceuticals management. In 2008-09, Health Canada will advance federal initiatives and continue to foster the improved management and use of pharmaceuticals in the health care system, in collaboration with P/T governments where appropriate. Health Canada will continue to assist the implementation of Patient Wait Times Guarantees (PWTGs), including a $30 million PWTG Pilot Project Fund to support provinces and territories in the testing, advancement and establishment of PWTGs and facilitate best outcomes for patients through innovation and shared best practices by 2010. There are also four ongoing federal PWTG pilot projects. Three of these projects address diabetes and prenatal care in selected First Nations communities and one addresses wait times for paediatric surgery. Through the Health Human Resources (HHR) Strategy, we are focusing on securing and maintaining a stable and optimal health workforce in Canada and supporting overall health care renewal. Health Canada is working closely with the provinces and territories to develop priority objectives for the Pan-Canadian HHR Strategy for 2008-2013, based on the Action Plan from the Framework for Collaborative Pan-Canadian HHR Planning. For more information on the HHR Strategy, see: http://www.hc-sc.gc.ca/hcs-sss/hhr-rhs/strateg/index_e.html |
|||||
| Expected Results | Performance Indicators | ||||
|---|---|---|---|---|---|
Pharmaceuticals Management
|
Pharmaceuticals Management
|
||||
| 2008-09 | 2009-10 | 2010-11 | |||
| ($ millions) | FTEs | ($ millions) | FTEs | ($ millions) | FTEs |
| 181.5 | 239 | 180.3 | 238 | 167.0 | 237 |
Program Activity - Canadian Assisted Human Reproduction
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Net expenditures | 2.2 | 2.1 | 2.1 | 2.1 |
| FTEs | 14 | 13 | 13 | 13 |
| Notes: Figures include an amount for departmental and regional infrastructure costs in support of program delivery. | ||||
Health Canada is responsible for developing the licensing framework and regulations (laboratory and clinical) that will ensure the health, safety, dignity, and human rights of the Canadians who access the services of the Assisted Human Reproduction (AHR) sector and of the children born from the use of AHR technologies. Because the science of AHR evolves rapidly, we must engage stakeholders on an ongoing basis to find a balance between the needs of patients who use these technologies to help them build their families, the children born from these technologies, and the providers of these services - with health and safety as overriding goals. The goal of these policies and regulations is to develop a responsive regulatory regime that is a leader both domestically and in the international AHR community reflecting the objectives set forth in the AHR Act. We also gather input from stakeholders, including the provinces, to ensure a pan-Canadian approach.
We have completed public consultations on the first series of regulatory initiatives under the AHR Act and are drafting regulations in areas such as licensing administration, requirements for clinical and laboratory activities (i.e., in vitro fertilization procedures using one's own gametes), and health reporting information. These regulations will be pre-published in the Canada Gazette in 2008.
In addition, we will reviewthe Processing and Distribution of Semen for Conception Regulations, presently under the Food and Drugs Act, to incorporate these regulations under the AHR Act. We will continue developing regulatory policy and gathering input from stakeholders on the following issues: the use of donated gametes (semen, ovum and embryos), undertaking preimplantation genetic diagnosis, research, and manipulation of human reproductive materials. We intend to bring the new regulations into force over the next five years.
| Expected Results | Performance Indicators |
|---|---|
|
|
Program Activity - International Health Affairs
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Net expenditures | 29.8 | 28.7 | 28.4 | 28.2 |
| FTEs | 80 | 80 | 81 | 80 |
| Notes: Figures include an amount for departmental and regional infrastructure costs in support of program delivery. | ||||
Health Canada works internationally through leadership, partnerships and collaboration to fulfill its federal mandate of striving to make Canada's population among the healthiest in the world. The Health Policy Branch serves as the department's focal point to initiate, coordinate and monitor departmental policies, strategies and activities that help promote Canadian priorities and values on the international health agenda. International collaboration on global health issues is important because the health of Canadians is influenced significantly by public health risks originating in other countries. Global issues such as pandemic influenza preparedness, HIV/AIDS strategies and global health security are critical initiatives about which we consult with key external health partners such as the World Health Organization (WHO) and the Pan-American Health Organization (PAHO).
Health Canada will focus on three major activities/programs during fiscal year 2008-09: the Government of Canada's Americas Strategy, the development of a WHO framework and the Global Health Security Initiative.
Health Canada will contribute to the Government of Canada's Americas Strategy by: 1) supporting ongoing governance of PAHO and expanding engagement with other multilateral institutions; 2) encouraging bilateral and trilateral relations with key countries on health policy; and 3) promoting and using Canadian technical expertise to improve health outcomes in the Americas. This will result in a more focussed approach to health by the federal government and its partners in the region by advancing good governance, ensuring economic development and maintaining health security in the hemisphere.
We are also working to provide greater strategic direction on key global health and governance issues involving WHO. We are developing a framework that will provide a common set of principles and priorities for the activities of all Government of Canada departments and agencies and contribute to an effective and coherent Canadian approach to WHO. The framework will be finalized in 2008-09 and will be implemented thereafter.
The Global Health Security Initiative (GHSI) is an informal, international partnership to strengthen global public health preparedness and response to chemical, biological, radiological, and nuclear (CBRN) threats, as well as pandemic influenza. Member countries/organizations include the G7 countries, Mexico, the European Commission, and the WHO as a technical advisor. Health Canada leads the GHSI and plays an active role within the Initiative. In 2008-09, Health Canada will work with GHSI members to strengthen networks that: 1) facilitate improved preparedness and response to CBRN threats and pandemic influenza; and 2) increase dialogue and exchange on emerging health security issues to allow for cross-national learning.
| Expected Results | Performance Indicators |
|---|---|
|
|
Strategic Outcome: Access to Safe and Effective Health Products and Food and Information for Healthy Choices
Under this strategic outcome, Health Canada is committed to promoting and protecting the health and safety of Canadians. The Department achieves this outcome by continuing to work towards reducing health risks to Canadians from health products and food and providing information so that Canadians can make informed decisions and adopt healthy choices.
In 2008-09, the key activities that will contribute towards achieving results under this strategic outcome are mainly outlined in the Blueprint for Renewal II1 policy framework and Canada's Food and Consumer Safety Action Plan2 as announced in December 2007. The action plan reflects the need to modernize the health products and food's regulatory system. As part of its Comprehensive Review of resources and programs, Health Canada will determine the level of activities as well as resources the programs need to meet the Department's regulatory responsibilities, including the need for baselines, performance indicators and targets and program costs.
Scientific and technical expertise are of critical importance to the activities of this strategic outcome. They are used on a daily basis for policy development, the evaluation of health products and foods, risk assessments, compliance testing, and nutrition and contaminant surveillance to research on food pathogens and pharmacogenomics. We carry out research in support of regulatory activities and related activities that contribute to evidence-based decision making to protect the health and safety of Canadians.
As part of our new initiatives, we are preparing the first-ever comprehensive strategic science plan for health products and food. This plan includes the identification of current scientific practice, changes in the key methods for the generation and analysis of evidence, and the evolution of science and technology as it affects the nature of the products that fall within the health products and food mandate. The plan will guide our future work and support us in building science partnerships across the Health Portfolio and with the national and international community.
We anticipate that over the next three to five years, there will be major pressures on our science and operational infrastructure, and that we will need to adapt our practices as operational needs and related applied science changes. For example, nanotechnology is adding a new dimension to our fundamental understanding of the physical nature of the world around us. Maintaining and modernizing regulatory and analytical tools, process and practices also continue to drive our investment requirements. Strategies and frameworks to address infrastructure challenges are being developed and will serve as the basis for planning, investment, engagement, partnership, consultation, and communication.
Assessing and understanding the current and future human resources needs is key to ensure the right mix of skills and expertise is in place to deliver on our priorities. As we move forward, we will need to address the challenges in attracting and retaining highly qualified talent resulting from a highly competitive demand-driven environment. In addition, the potential retirement of 21% of the Health Products and Food Branch workforce within the next five years requires an aggressive strategy to address succession management, learning development and knowledge transfer to ensure management stability across the organization. We will implement succession planning strategies and knowledge transfer initiatives that are aligned with our business needs.
Health Canada will implement its Consumer Information Strategy and the Policy on Public Input in the review of regulated products to improve the way the department communicates information to consumers and to increase openness, transparency and accountability, thereby maintaining public confidence in the regulatory system for health products. As part of the Blueprint for Renewal II, Health Canada will conduct strategic outreach and consultations to increase consumer product safety. The department will collaborate with stakeholders and the public to seek input on product review, policy programming and legislative and regulatory proposals that will enhance the overall quality of decision-making and improve the safety of products for Canadians.
Health Canada is working with federal science and technology partners to promote and protect the health and safety of Canadians. Some of our work includes collaborating directly with Agriculture and Agri-food Canada (AAFC), Environment Canada (EC), the Public Health Agency of Canada (PHAC), and the Canadian Food Inspection Agency (CFIA).
Health Canada's regional offices develop and deliver key programs and services, including monitoring the risks of health products and the safety of food, and the investigation and inspection associated with the import, sale and manufacture of health products. They work with regional stakeholders and provincial and municipal governments to promote collaboration, participate in the monitoring of adverse reactions and assist in developing policy responses in areas including: food safety, nutrition, natural health products, antimicrobial resistance, and the efficacy of health products. As part of the national laboratory network, Health Canada's regional laboratories are key contributors to our work of developing and managing food safety research and surveillance projects on foodborne pathogens, persistent organic pollutants, natural toxins, genetically modified food, food allergen detection, and nutrition.
Internationally, Health Canada is working with the World Health Organization (WHO), other multilateral organizations and other governments on health products and food safety issues to strengthen and promote broader regulatory cooperation and encourage technical information exchange. Our partnerships enable Health Canada to ensure that its efforts meet the needs of all Canadians.
The following program activities help support the Health Products and Food's Strategic Outcome:
- Health Products
- Food and Nutrition
Program Activity - Health Products
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Gross expenditures Less: Expected respendable revenues |
266.7 39.9 |
226.3 39.9 |
224.0 39.9 |
220.7 39.9 |
| Net expenditures | 226.8 | 186.5 | 184.1 | 180.9 |
| FTEs | 2,211 | 2,038 | 2,021 | 2,007 |
| Notes: The decrease in forecast/planned expenditures from 2007-08 to 2008-09 is due in part to one-year funding received in 2007-08 for Natural Health Products, funding from the Department's operational budget carry-forward and a decrease in the funding level for the Therapeutic Access Strategy. The decrease in planned expenditures from 2008-09 to 2009-10 is mainly due to the sunset of Health Canada's Response to Bovine Spongiform Encephalopathy. The decrease is partially offset by an increase in the funding level for Protecting Canadians and the Environment from Toxic Substances through a Chemicals Management Plan. Figures for 2007-08 have been restated due to the change in the Program Activity Architecture for 2008-09. Figures include an amount for other departmental and regional infrastructure costs supporting program delivery. |
||||
The Health Products program activity is responsible for a broad range of health protection and promotion activities that affect the everyday lives of Canadians. As the federal authority responsible for the regulation of health products, the program activity evaluates and monitors the safety, quality
and effectiveness of drugs (human and animal), biologics, medical devices, and natural health products, under the authority of the Food and Drugs Act and Regulations, as well as the Department of Health Act.
The program activity also provides timely, evidence-based and authoritative information to key stakeholders (including but not limited to: health care professionals such as physicians, pharmacists and practitioners such as herbalists, naturopathic doctors, Traditional Chinese Medicine (TCM) practitioners) and members of the public to enable them to make informed decisions and healthy choices.
In its November 2006 report3, the Auditor General of Canada has identified a number of gaps in the regulatory system and questioned whether Health Canada has adequate tools and resources to fulfill its regulatory responsibilities and protect the health and safety of Canadians.
Although the current regulatory system has served Canadians well over the past decades, it needs to be modernized to address emerging health and safety risks, help ensure timely access to innovative health products, and reduce adverse effects. Pharmaceuticals, non-prescription medicines, vaccines, blood products, gene therapies, tissues, and medical devices all offer benefits to Canadians, but they also carry risks. Today, most of our efforts are spent on pre-market assessment. As part of Canada's Food and Consumer Safety Action Plan, a new legislation is being considered to better protect Canadians from unsafe health products by:
- Taking a life-cycle approach to regulation by keeping watch and gathering information on products like pharmaceuticals both before and after they are approved for sale. This would allow the most rapid action possible to address the health risks of products that don't come to light until they are on the market;
- Gaining new authorities to order needed changes to health product labels to provide product information to consumers and recall health products as soon as a safety problem is identified; as well as to compel manufacturing to conduct post-market studies and issue risk communication;
- In cooperation with provinces and territories, acquiring the power to make it mandatory for hospitals to report on serious adverse drug reactions, so dangers can be identified more effectively to inform corrective action as early as possible; and
- Raising fines to levels comparable with other industrial countries.
These measures under the new legislation will help Health Canada address some of our regulatory gaps. In addition, Health Canada will work on the next phases of the Blueprint initiatives, which will be guided by recommendations that resulted from stakeholder consultations. More of what will be accomplished in 2008-09 is described in the key programs below.
| Expected Results | Performance Indicators |
|---|---|
|
|
Program Activity - Food and Nutrition
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Gross expenditures Less: Expected respendable revenues |
84.7 1.3 |
79.3 1.3 |
77.6 1.3 |
77.4 1.3 |
| Net expenditures | 83.4 | 77.9 | 76.3 | 76.0 |
| FTEs | 699 | 706 | 695 | 692 |
| Notes: The decrease in forecast/planned expenditures from 2007-08 to 2008-09 is mainly due to the sunset of the Agriculture Policy Framework. The decrease in planned expenditures from 2008-09 to 2009-10 is mainly due to the sunset of Health Canada's Response to Bovine Spongiform Encephalopathy. Figures for 2007-08 have been restated due to the change in the Program Activity Architecture for 2008-09. Figures include an amount for departmental and regional infrastructure costs in support of program delivery. |
||||
Planned Spending and Full-Time Equivalents (FTEs)
The Food and Nutrition program activity establishes policies, regulations and standards related to the safety and nutritional quality of food. Food safety standards-quality are enforced by the Canadian Food Inspection Agency (CFIA). The legislative framework for food is found in the Food and Drugs Act and Regulations, the Canadian Food Inspection Agency Act and the Department of Health Act.
The program activity also promotes the nutritional health and well-being of Canadians by collaboratively defining, promoting and implementing evidence-based nutrition policies and standards. As the focal point and authoritative source for nutrition and healthy eating policy and promotion, the program activity disseminates timely, evidence-based and authoritative information to Canadians and stakeholders to enable them to make informed decisions and healthy choices.
Health Canada's activities to strengthen food safety are key parts of Canada's Food and Consumer Safety Action Plan. As part of the action plan, Health Canada will modernize its food safety system through an integrated and proactive approach. Health Canada will conduct legislative amendments to the Food and Drugs Act, which would help streamline and provide more consistent regulatory tools across all sectors and better position Health Canada to achieve its food safety mandate.
More specifically, Health Canada addresses the mitigation of risks to human health associated with foods that contain environmental contaminants (e.g. mercury in fish), chemicals resulting from food processing (e.g. furan and acrylamide in food), chemicals resulting from industrial (e.g. heavy metals, Persistent Organic Pollutants (POPs), and agricultural (veterinary drugs and pesticides) uses. Health Canada identifies and assesses potentially harmful health effects of these contaminants and determines appropriate ways for Canadians to reduce their exposure to them, while at the same time considering the benefits of consuming particular foods that may contain such contaminants.
In the area of nutrition, controlling the addition of vitamins and minerals to our foods (a process known as fortification) is one way that Health Canada ensures that Canadians receive the nutrients they need but are not exposed to levels that are dangerously high. With the increasing marketing of certain newly fortified foods (beverages) and highly fortified foods in other jurisdictions, Health Canada has an obligation to ensure the safety and safe use of some of these products. Currently, other jurisdictions such as Australia and New Zealand, and the Commission of European Communities, are in the process of developing policies on voluntary fortification of foods.
Healthy eating plays a vital role in promoting health and reducing the risk of obesity and other nutrition-related chronic diseases. Changes in social and physical environments are resulting in increasingly complex and influential forces affecting the eating practices of Canadians. Activities to advance knowledge about what Canadians are eating, the determinants of eating behaviour and effective strategies to promote healthy eating are essential to the development of meaningful and effective policies and programs to improve the nutritional health and well-being of Canadians.
To address some of these pressures, Health Canada plans to put in place a number of initiatives and measures, which are described in key programs below.
| Expected Results | Performance Indicators |
|---|---|
|
|
Endnotes
1.Blueprint for Renewal policy framework
http://www.hc-sc.gc.ca/ahc-asc/branch-dirgen/hpfb-dgpsa/blueprint-plan/blueprint-plan_ll_intro_e.html
2.The Government of Canada's Food and Consumer Safety Action Plan
http://healthycanadians.ca/pr-rp/plan_e.html
3.The Auditor General Report of November 2006 - Chapter 8
http://www.cbc.ca/news/background/auditorgeneral/report200611.html
4.The Auditor General Report of 2004
http://www.oag-bvg.gc.ca/domino/reports.nsf/html/20040302ce.html
5.Cabinet Directive on Streamlining Regulation
http://www.regulation.gc.ca/cdsr-dcrr_e.pdf
6.Food-related health advisories, warnings and other food safety information
http://www.hc-sc.gc.ca/ahc-asc/media/advisories-avis/2007/2007_08_e.html
http://www.hc-sc.gc.ca/ahc-asc/media/advisories-avis/2007/2007_58_e.html
http://www.hc-sc.gc.ca/ahc-asc/media/advisories-avis/2007/2007_142_e.html
http://www.hc-sc.gc.ca/fn-an/securit/handl-manipul/index_e.html
7.Novel foods (or also known as Genetically Modified Foods)
http://www.hc-sc.gc.ca/fn-an/gmf-agm/index_e.html
http://www.hc-sc.gc.ca/fn-an/gmf-agm/appro/index_e.html
http://www.hc-sc.gc.ca/fn-an/legislation/acts-lois/index_e.html
8.The Canadian Community Health Survey 2.2, Nutrition Focus
http://www.hc-sc.gc.ca/fn-an/surveill/nutrition/commun/index_e.html
9.Nutrition and healthy eating during pregnancy
http://www.hc-sc.gc.ca/fn-an/nutrition/prenatal/index_e.html
10.Nutrition labelling
http://www.hc-sc.gc.ca/fn-an/label-etiquet/index_e.html
11.Food Guide
http://www.hc-sc.gc.ca/fn-an/food-guide-aliment/index_e.html
Strategic Outcome: Reduced Health and Environmental Risks from Products and Substances, and Healthy, Sustainable Living and Working Environments
Program activities touch many aspects of Canadians' daily lives. These include: drinking water safety; air quality; chemicals in the workplace and the environment; substance use and abuse; tobacco and secondhand smoke; consumer product safety (including cosmetics); radiation exposure; workplace health; and, pest control products. Other health and safety related activities include: the Government's public safety and anti-terrorism initiative; chemical and nuclear emergency preparedness; inspection of food and potable water for the travelling public; and, health contingency planning for visiting dignitaries. Much of this work is governed through legislation including the Canadian Environmental Protection Act (CEPA), the Controlled Drugs and Substances Act, the Hazardous Products Act, the Tobacco Act, the Food and Drugs Act, the Radiation Emitting Devices Act, the Quarantine Act, the Pest Control Products Act, and others.
Health Canada will continue to advance science and use strong evidence-based research to formulate our healthy and safe living promotion and harm prevention programs, policies and regulations. Our experts work closely with colleagues in the federal government and beyond (e.g. academia) in the areas of both research and development and related scientific activities. Anticipatory, applied and novel research provide the evidence of emerging health issues through investigations along the continuum from exposure and hazard assessment, to mechanism of action and population studies, to contribute to the design and implementation of policies, regulations and legislation, as well as to decision making, aiming at protecting the health and safety of Canadians. In our role as a regulator, we extend our scientific research by contributing to the generation, dissemination and application of scientific and technological knowledge, including the assessment of products and processes for the purpose of regulation, as well as surveillance, testing and collection of information. In addition to our internal activities related to scientific research, health surveillance and foresight in the safe use of emerging and merging technologies (such as biotechnology and nanotechnology), we will also use the science conducted by external organizations to help identify risks to human health, and assess and manage these risks.
In response to the 2006 report of the Auditor General, a Comprehensive Review of programs was launched to determine the full costs of regulatory programs and identify opportunities to improve management and efficiency. An integrated change strategy will detail the actions to be undertaken immediately and over the coming years to address any gaps, risks, opportunities, and challenges identified by the Review. The findings of the Review will also be leveraged to support the development of an Integrated Planning and Reporting Strategy that will bring together planning tools and software, finances, risks, results, and performance indicators to improve business intelligence for management decision making and provide greater rigour, structure and integration to planning and accountability.
Activities within this strategic outcome require sustained partnerships with other federal government departments as well as provinces and territories, non-governmental organizations, academia, and the international community. We work closely, for example, with all branches of Health Canada as well as Environment Canada, Natural Resources Canada, the Public Health Agency of Canada, the Canadian Institute for Health Information, and others on health and environment issues. Through the Government's National Anti-drug Strategy aimed at tackling illegal drug use and associated crime, we are working with Justice Canada, Public Safety Canada, the Royal Canadian Mounted Police, and the Canada Border Services Agency. We are also engaged in the Government's public safety and anti-terrorism initiatives. In addition, we collaborate with provinces and territories through various committees to develop guidelines on issues such as tobacco cessation and safe drinking water, and to coordinate nuclear emergency preparedness activities. Our work with international partners allows us to better respond to domestic health and safety issues, meet our obligations and commitments, share best practices, and promote Canadian leadership globally.
The contributions of Health Canada's regional offices are integral to program delivery. They include: playing a leadership role on key national initiatives; conducting inspection, surveillance and educational activities related to consumer products, tobacco, controlled drugs and substances; conducting risk assessments and evaluations; and providing health advice to federal employees, provinces and municipalities related to chemical contaminants and exposure levels, and work environments.
The following program activities help support the Strategic Outcome:
- Sustainable Environmental Health
- Consumer Products
- Workplace Health
- Substance Use and Abuse
- Pesticide Regulation
Program Activity - Sustainable Environmental Health
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Gross expenditures | 152.1 | 166.8 | 181.3 | 179.0 |
| Less: Expected respendable revenues | 1.4 | 1.4 | 1.4 | 1.4 |
| Net expenditures | 150.7 | 165.4 | 179.9 | 177.6 |
| FTEs | 966 | 1,056 | 1,092 | 1,071 |
| Notes: The increase in forecast/planned expenditures from 2007-08 to 2008-09 is due mainly to an increase in the funding level for protecting Canadians and the environment from toxic substances through the Chemicals Management Plan. The increase in planned expenditures from 2008-09 to 2009-10 is due mainly to an increase in the funding level for the Chemicals Management Plan and for the implementation of the Clean Air Regulatory Agenda. Figures include an amount for departmental and regional infrastructure costs in support of program delivery. |
||||
The environment continues to be a key determinant of health for all Canadians. This program activity promotes and protects the health of Canadians, including vulnerable populations such as children, by identifying, assessing and managing health risks posed by environmental factors in living, working and recreational environments. The scope of activities includes: research on drinking water quality, air quality, contaminated sites, toxicology and climate change; clean air programming and regulatory activities; risk assessment and management of environmental contaminants, including new and existing chemical substances, environmental noise, environmental electromagnetic frequencies, products of biotechnology and products of other new and emerging technologies (including nanotechnology); solar ultraviolet radiation; preparedness for nuclear and environmental disasters as well as working with the passenger conveyance industry to protect the travelling public.
The Canadian Environmental Protection Act,1999, in concert with other federal statutes such as the Hazardous Products Act, the Food and Drugs Act, and the Pest Control Products Act, provide assessment and protection regimes for a variety of substances and products. Under the New Substances Notification Regulations of CEPA, 1999, new substances undergo a rigorous health and environmental risk assessment before they can be used in Canada. Under the Chemicals Management Plan, Health Canada and Environment Canada have used the results of the categorization of the 23,000 existing substances in Canada as of the mid-1980s to determine priority chemicals for assessment and then management of the risks associated with those substances deemed to pose a possible risk to human health and the environment. Other activities include: implementing a national bio-monitoring system; developing risk management performance agreements with industry sectors; and, strengthening the assessment and management of environmental health risks, including those posed by pharmaceuticals, personal care products, cosmetics, and food. Finally, enhanced communications and outreach activities allow Canadians to make better informed decisions about limiting their exposure to potential environmental hazards.
| Expected Results | Performance Indicators |
|---|---|
|
|
The financial information for Emergency Preparedness includes Healthy Environments and Consumer Safety Branch activities only.
Program Activity - Consumer Products
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Gross expenditures | 25.8 | 24.6 | 25.1 | 27.1 |
| Less: Expected respendable revenues | 0.5 | 0.5 | 0.5 | 0.5 |
| Net expenditures | 25.3 | 24.2 | 24.6 | 26.6 |
| FTEs | 195 | 203 | 208 | 216 |
| Notes: Figures include an amount for departmental and regional infrastructure costs in support of program delivery. | ||||
Health Canada identifies, assesses, manages, and communicates to Canadians the health and safety risks associated with consumer products (including domestic, industrial and clinical use products), cosmetics and radiation emitting devices. This is achieved through research, risk assessments and the development of risk management strategies to minimize the exposure of Canadians to potentially hazardous products. Also included are regulatory monitoring and compliance activities as well as information, education and guidance aimed at both industry and the public. Relevant acts include: Hazardous Products Act, Food and Drugs Act (cosmetics), and Radiation Emitting Devices Act.
| Expected Results | Performance Indicators |
|---|---|
|
|
Program Activity - Workplace Health
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Gross expenditures | 62.0 | 48.9 | 48.2 | 46.4 |
| Less: Expected respendable revenues | 13.8 | 14.1 | 14.1 | 14.1 |
| Net expenditures | 48.2 | 34.8 | 34.1 | 32.2 |
| FTEs | 463 | 437 | 436 | 434 |
| Notes: The decrease in forecast/planned expenditures from 2007-08 to 2008-09 is due largely to the one-year funding received in 2007-08 for the Public Service Health Program. Figures include an amount for departmental and regional infrastructure costs in support of program delivery. |
||||
This program activity provides services to protect the health and safety of the federal public sector, visiting dignitaries, and others. Specific programs include: the provision of occupational health services to federal employees; delivery of the Employee Assistance Program; emergency health services to Internationally Protected Persons; dosimetry services (the measurement of personal, occupational exposure to radiation); and, Workplace Hazardous Materials Information System (WHMIS) a national hazard communication standard, including worker education, inspector training and standards for cautionary labels
| Expected Results | Performance Indicators |
|---|---|
|
|
Program Activity - Substance Use and Abuse
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Net expenditures | 142.3 | 152.9 | 151.5 | 148.2 |
| FTEs | 555 | 575 | 586 | 586 |
| Notes: The difference between forecast and planned spending is due to a reprofile of funding from 2007-08 for the Drug Strategy Community Initiative Fund to 2008-09 and 2009-10 as well as an increase in funding for the National Anti-Drug Strategy which decreases slightly in 2010-11. Figures include an amount for departmental and regional infrastructure costs in support of program delivery. |
||||
Through regulatory, programming and educational activities, Health Canada seeks to improve health outcomes by reducing and preventing tobacco consumption and combatting alcohol and drug abuse.
Through the Tobacco Act and its regulations, Health Canada regulates the manufacture and sale of tobacco. Health Canada also leads the Federal Tobacco Control Strategy.
Health Canada administers the Controlled Drugs and Substances Act (CDSA) and its regulations. Through four regional labs, Health Canada provides expert scientific advice and drug analysis services to law enforcement agencies. The Marihuana Medical Access Regulations and related programs control the authorization for use and cultivation of marihuana by those suffering from grave and debilitating illnesses. Health Canada is a partner in the National Anti-Drug Strategy which includes: prevention programming aimed at youth; facilitating access to treatment programs; compliance and enforcement activities related to controlled substances and precursor chemicals; and, increased resources to drug analysis services commensurate with the increase in law enforcement resources; and the need to enhance the safety of procedures used in the dismantling of clandestine laboratories.
| Expected Results | Performance Indicators |
|---|---|
|
|
Program Activity - Pesticide Regulation
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|
Gross expenditures Less: Expected respendable revenues |
68.3 7.0 |
67.5 7.0 |
68.2 7.0 |
68.3 7.0 |
| Net expenditures | 61.3 | 60.5 | 61.2 | 61.3 |
| FTEs | 702 | 707 | 715 | 712 |
| Notes: Figures include an amount for departmental and regional infrastructure costs in support of program delivery. | ||||
To help prevent unacceptable risks to people and the environment, and facilitate access to sustainable pest management tools, Health Canada, through the Pest Management Regulatory Agency (PMRA), regulates the importation, sale and use of pesticides under the federal authority of the Pest Control Products Act (PCPA) and Regulations.
Protecting human health and the environment is an important priority for Health Canada and science is the foundation for Health Canada's activities related to the PCPA and its Regulations. We conduct assessments of risks to human health and the environment arising from exposure to chemical and biological pesticides as well as assessments of the value of these products. In support of this work, we develop assessment methodologies, pesticide testing protocols, risk reduction strategies, and risk management tools. Scientific expertise is in place in the following areas: toxicology, environmental toxicology, analytical chemistry, environmental fate and chemistry, biochemistry, endocrinology, ecology, crop science, plant pathology, entomology, occupational and bystander risk assessment, and aggregate and cumulative risk assessment.
The PMRA will work closely with other Health Canada branches and Health Portfolio members over the next fiscal year on a variety of initiatives. We will collaborate with Health Canada and Environment Canada to achieve re-evaluation targets established through the Government of Canada's Chemicals Management Plan. We will continue to work horizontally with Agriculture and Agri-Food Canada (AAFC) through our joint Pesticide Risk Reduction Program. This year, the program will focus on finding sustainable solutions to key areas of pesticide risk associated with grower identified pest management issues. Health Canada's National Pesticide Compliance Program (NPCP) will continue to work with national partners to promote, verify and enforce compliance with the PCPA. Compliance strategies, programs and activities will be modernized to align with new priorities and to improve transparency of the program and results to Canadians.
The PMRA co-chairs a multi-departmental working group known as the '6NR', involving six federal science-based departments and agencies (Agriculture and Agri-food Canada, the Canadian Food Inspection Agency, Environment Canada, Fisheries and Oceans Canada, Health Canada, and Natural Resources Canada). Over the next fiscal year, the 6NR will complete the summative evaluation of the
Horizontal Initiative: Building Public Confidence in Pesticide Regulation and Improving Access to Pest Management Products (also referred to as the BPC Initiative). Further information can be found at: http:// www.pmra-arla.gc.ca/english/pdf/ plansandreports/evaluation_bpcinitiative-e.pdf.
(BPC Initiative)
| Expected Results | Performance Indicators |
|
|
Human resources capacity to respond to the PMRA's needs continues to be an ongoing challenge. This requires that the PMRA continually re-prioritize work plans.
To address this, the PMRA has integrated Human Resources Planning within the Business Planning process and identified key priorities, including monitoring and reporting of these activities. The priorities are:
- Designation of Senior Manager Champion to develop a strategy with options/actions and monitoring and reporting;
- Working closely with Human Resources to build capacity; and,
- Continue to allocate funding to recruitment and staffing activities.
Strategic Outcome: Better Health Outcomes and Reduction of Health Inequalities Between First Nations and Inuit and Other Canadians
The objective of First Nations and Inuit health program activity is to improve health outcomes, by ensuring the availability of, and access to, quality health services, and by supporting greater control of the health system by First Nations and Inuit.
Health Canada works closely with our health partners and other federal departments to support First Nations and Inuit health. We support the Public Health Agency of Canada in its delivery of Children and Youth programming through the Aboriginal Head Start in Urban and Northern Communities program, as well as its work on a number of pan-Aboriginal programs and pandemic planning activities. We work closely with Indian and Northern Affairs Canada (INAC) through: the First Nations Water Management Strategy, to ensure that all First Nations communities across Canada have access to a safe and reliable water supply; the Home and Community Care program, to address the gaps in continuing care services available to First Nations and Inuit communities; and the Labrador Innu Comprehensive Healing Strategy, to support long term healing in the Labrador Innu communities. We also work with Indian Residential Schools Resolution Canada to ensure that eligible former students of Indian residential schools have access to mental health and emotional supports.
Program Activity - First Nations and Inuit Health Programming and Services
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
|---|---|---|---|---|
| Gross expenditures Less: Expected respendable revenues |
2,217.1 5.4 |
2,161.5 5.5 |
2,215.1 5.5 |
2,048.3 5.5 |
| Net expenditures | 2,211.7 | 2,156.0 | 2,209.6 | 2,042.8 |
| FTEs | 2,879 | 2,876 | 2,880 | 2,728 |
| Notes: The decrease in forecast/planned expenditures from 2007-08 to 2008-09 is due in part to the one-year funding received in 2007-08 to support the delivery of health services to First Nations and Inuit populations. The decrease is partially offset by an increase in the funding level for the Indian Residential Schools Resolution
Health Support Program and the yearly growth for the Indian Envelope. The increase in planned expenditures from 2008-09 to 2009-10 is due mainly to an increase in the funding level for the Indian Residential Schools Resolution Health Support Program, the National Anti-Drug Strategy, and the yearly growth of the Indian Envelope. The decrease in planned expenditures from 2009-10 to 2010-11 is due largely to the sunset of funding for the Follow-up to the Special Meeting of Ministers and Aboriginal Leaders in September 2004 and for the Territorial Medical Travel and Health Access Funds. The decrease is partially offset by the yearly growth of the Indian Envelope. Figures include an amount for departmental and regional infrastructure costs in support of program delivery. |
||||
Health Canada delivers a range of health services to First Nations and Inuit. A series of primary health care services are provided in approximately 200 First Nations communities, largely in rural or remote areas where access to provincial health care services is limited. In addition, home and community care services are provided in approximately 600 First Nations communities. Health Canada supports a range of community-based health programs, focussing on children and youth, mental health and addictions, and chronic disease and injury prevention. We also deliver a range of public health programs with a focus on communicable disease control, including tuberculosis, HIV/AIDS and immunization, and environmental health and research. The Non-Insured Health Benefits (NIHB) Program provides approximately 800,000 eligible First Nations and Inuit with a limited range of medically necessary health-related goods and services not provided through private insurance plans, provincial/territorial health or social programs or other publicly funded programs. Health Canada also maintains healthcare infrastructure, including nursing stations, treatment centres and community health centres. To support greater First Nations and Inuit control over their health system, many health programs and services are provided in collaboration with First Nations and Inuit communities, and a number of communities deliver their own health services through Contribution Agreements with Health Canada. Health Canada also works closely with other health partners, including provinces and territories, to support and coordinate health programs and services for First Nations and Inuit.
Health Canada uses information from evaluations and review studies to support program improvement on a continual basis. We will continue to work on three integrated program cluster evaluations for the Children and Youth, Environmental Health and Research and Communicable Disease Control program areas and on an implementation/process evaluation for the Maternal Child Health program in 2008-09.
| Expected Results | Performance Indicators |
|
|
Human Resource Challenges:
The Department continues to experience challenges in recruiting nurses into First Nations and Inuit communities, especially in remote and isolated areas. Health Canada is particularly concerned with the impact that nursing shortages have on the provision of services and the health and well-being of First Nations and Inuit, and we are undertaking a number of activities to address this challenge. Through the Aboriginal Health Human Resource Initiative, Health Canada is supporting Aboriginal students to pursue health careers, and working to improve retention of existing health care workers in First Nations and Inuit communities. We will continue to support the recruitment and retention of nurses in First Nations communities by pursuing the integration of Nurse Practitioners and Licensed Practical Nurses for health program delivery in remote and isolated First Nations communities. We will continue an aggressive nurse recruitment campaign to include piloting a Nurse Practitioner Student Incentive program to attract Nurse Practitioner students to First Nations communities. We will also continue to work with the Canadian Association of Schools of Nursing and other stakeholders to provide appropriate education programs to meet mandatory training requirements.
Key priorities:
- Continuing to provide health-related programs and services
- Improving quality of and access to health-related programs and services
- Promoting healthy living and disease prevention
- Improving accountability and performance measurement
Key Programs and Services
In addition to the four priorities, the Department has identified five key program areas from the Program Activity and Architecture (PAA).
Health Canada will continue to invest in the Maternal Child Health Program. This program provides pregnant First Nations women and families with infants and young children living on-reserve with home visits by nurses and trained, experienced mothers in the community. The program also provides case management for families living with more complex needs. In the north, program funding will continue to enhance existing health promotion programs. Key plans for 2008-09 are to increase the number of trained nurses and home visitation workers and enhance existing health promotion programs in Nunavik, Nunatsiavut and the Territories.
Work to expand the Aboriginal Head Start on Reserve (AHSOR) program will continue through increased training of AHSOR staff and a study of the status of AHSOR capital facilities in communities to better define capital requirements. The goal of the AHSOR program is to support locally controlled and designed early childhood intervention strategies for on-reserve First Nations children. Outreach activities will be strengthened and expanded to serve small communities that do not have enough children to run a centre-based program. In 2008-09, the results of 17 demonstration projects undertaken to improve integration and coordination among AHSOR, Human Resources and Social Development Canada's First Nations and Inuit Child Care Initiative, and INAC funded daycares in Alberta and Ontario will be used to inform future work on key components of the interdepartmental early childhood development strategy.
Implementation of the National Aboriginal Youth Suicide Prevention Strategy will continue with a focus on prevention, skills training, and the development of protocols to respond to suicide-related crises. Moreover, the Strategy will support greater awareness and knowledge through activities promoting mental wellness and resiliency among youth. Evaluation, data gathering and research initiatives will be undertaken to improve knowledge of effective Aboriginal youth suicide prevention activities and how suicide impacts Aboriginal youth living off reserve and in urban areas.
Health Canada and its partners have developed a strategic action plan for First Nations and Inuit mental wellness that aims to improve mental wellness through a coordinated continuum of mental health and addictions services respecting traditional, cultural and mainstream approaches to healing. Work with our partners, including the new Mental Health Commission of Canada, will continue in 2008-09 in order to validate and implement elements of the plan. In 2008-09, key aspects of the plan that can proceed within existing resource levels, such as raising awareness of the plan and aligning existing mental wellness activities with its objectives, will be implemented. The Department will also continue to offer a range of mental health and emotional support services to former students of residential schools and their families through the Indian Residential Schools Resolution Health Support Program.
Through Patient Wait Times Guarantees pilot projects in diabetes and prenatal care services on-reserve, Health Canada will test defined time frames and access to alternative care options if these time frames are exceeded. In 2008-09, pilot projects will begin data collection and testing of time frames.
Continuing work under the Aboriginal Diabetes Initiative will improve screening and care services and will strengthen community-based diabetes prevention and promotion activities through increased training and employment of community-based diabetes prevention workers. Participatory research will be undertaken to gather more data on pre-diabetes, diabetes and its complications, and to develop strategies to reduce the burden of the disease. Complementary activities to aid in diabetes prevention will also be undertaken including the continued promotion and dissemination of a Eating Well with Canada's Food Guide -- First Nations, Inuit, and Mé´©s, as well as ongoing partnerships with retailers in northern isolated communities to support healthy eating by increasing the availability, quality and promotion of healthy foods.
Employing a team approach which focuses on building First Nations' capacity, Health Canada works with First Nations to ensure that all First Nations communities south of 60Ì? have access to safe and reliable drinking water. Health Canada raises awareness in First Nations communities of the importance of using safe drinking water best practices on-reserve for public health through targeted risk communication materials.
In 2008-09, Health Canada will begin implementation of a new policy to: address the drinking water quality in individual wells and wells with two to four connections on-reserve; focus on public awareness and education; enhance quality assurance/quality control monitoring for contaminants; and, develop waterborne illness procedures.
As part of Health Canada's efforts to ensure that its nursing stations and health centres meet appropriate environmental standards, we will undertake remediation activities in at least 9 of 19 remaining contaminated sites in 2008-09. Health Canada will also continue its work with First Nations and Inuit to help them improve their health and well-being by building and supporting their capacity to identify, understand and control the impact of exposure to environmental hazards. In 2008-09, we will provide a total of $2 million in funding to at least 10 First Nations and Inuit communities to develop community-based research projects dealing with health-related environmental issues. In addition, Health Canada will continue to conduct regionally-based traditional food safety workshops, bringing together First Nations communities to increase their knowledge of the nutritional benefits and risks of contaminants so they can make informed decisions on food choices.
Health Canada will continue to work with our partners to support the development and implementation of quality improvement activities, including accreditation of First Nations and Inuit health organizations, and modernization and accreditation of addictions treatment centres. In 2008-09, $2.5 million will be invested in accreditation with an estimated 10% increase in the number of accredited treatment centres and health services.
By increasing the amount of bursary and scholarship funds available through the National Aboriginal Achievement Foundation and other Aboriginal organizations, we will exceed our target of doubling the numbers of First Nations, Inuit and Mé´©s students pursuing health career studies, and in the longer term, will increase the supply of Aboriginal health care workers. We will also continue to work with provinces, territories, colleges and universities to double the number of student support programs for First Nations, Inuit and Mé´©s health care students. Through work with health professional organizations and post-secondary school partners, health educational curricula will be adapted so that future health care workers have a better understanding of First Nations, Inuit and Mé´©s clients, leading to an improved quality of service to the clients and better health outcomes.
Health Canada supports the construction, operation, maintenance and environmental management of on-reserve health facilities and staff residences. In 2008-09, fourteen health facilities will be constructed or expanded, and recapitalization initiatives (repairs, replacements, upgrades) will improve the working environment of clients and staff, and enhance the quality of health care services offered at the community level. We will also invest $1.5 million in environmental audit, assessment and remediation activities to ensure operations of health facilities in First Nations communities meet environmental codes and requirements and are consistent with the Department's commitments to sustainable development.
The Aboriginal Health Transition Fund will continue to support federal, provincial and territorial governments, and Aboriginal communities and organizations, to improve the integration and adaptation of health services to better meet the needs of First Nations, Inuit and Mé´©s. Expected results for 2008-09 include the implementation of provincial and territorial plans and projects to better adapt health care services, and the continued implementation of Health Canada Regional plans and projects, to improve the integration and coordination of provincial, territorial and federal health programs and services. These projects, along with cross-jurisdictional First Nations, Inuit and Mé´©s projects supported by capacity funding, will contribute to improve partnerships between federal, provincial and territorial governments and Aboriginal peoples in the delivery of health programs and services for Aboriginal peoples. In addition, Health Canada will continue to pursue improvements to quality and accessibility of health services for First Nations through tripartite agreements with provincial governments, such as the First Nations Tripartite Health Plan with the Province of British Columbia and British Columbia First Nations Leadership Council.
In March 2004, the federal government directed Canada Health Infoway to facilitate the development of an electronic Pan-Canadian communicable disease management and public health surveillance system (now known as Panorama) in partnership with the provinces and territories. In 2008-09, plans will be completed and implementation begun to include First Nations health facilities and communities within the provincial implementations of Panorama.
Health Canada is developing plans for an enhanced and more effective use of information and communications technologies to support health care delivery and management. We will continue our collaborative work with various stakeholders towards the finalization of long-term Health Infostructure and e-Health strategy framework.
The First Nations Regional Longitudinal Health Survey is a national health survey of First Nations on-reserve. This survey collects information on medical and non-medical determinants of health and well-being of First Nations people living on reserves. Health Canada funds this survey and works collaboratively with the Assembly of First Nations on this project to build First Nations capacity and participation in health data collection and analysis. The data collection phase is expected to be completed in 2008-09.
First Nations and Inuit Programs and Services and Web Links
Non-Insured Health Benefits 2005/06 Annual Report
http://www.hc-sc.gc.ca/fnih-spni/pubs/nihb-ssna/2006_rpt/index_e.html
Non-insured Health Benefits Program
http://www.hc-sc.gc.ca/fnih-spni/nihb-ssna/index_e.html
Aboriginal Head Start On Reserve
http://www.hc-sc.gc.ca/fnih-spni/famil/develop/ahsor-papa_intro_e.html
Fetal Alcohol Spectrum Disorder
http://www.hc-sc.gc.ca/fnih-spni/famil/preg-gros/intro_e.html
Aboriginal Diabetes Initiative
http://www.hc-sc.gc.ca/fnih-spni/diseases-maladies/diabete/index_e.html
Injury Prevention
http://www.hc-sc.gc.ca/fnih-spni/promotion/injury-bless/index_e.html
Indian Residential Schools Resolution Health Support Program
http://www.hc-sc.gc.ca/fnih-spni/services/indiresident/index_e.html
National Native Alcohol and Drug Abuse Program
http://www.hc-sc.gc.ca/fnih-spni/substan/ads/nnadap-pnlaada_e.html
Drinking Water Quality
http://www.hc-sc.gc.ca/fnih-spni/promotion/water-eau/index_e.html
Immunization Schedule for Infants and Children
http://www.phac-aspc.gc.ca/im/is-cv/index.html
Targeted Immunization Strategy
http://www.hc-sc.gc.ca/ahc-asc/activit/strateg/fnih-spni_e.html#immuni
e-Health
http://www.hc-sc.gc.ca/fnih-spni/services/ehealth-esante/index_e.html
Aboriginal Health Human Resources Initiative
http://www.hc-sc.gc.ca/ahc-asc/activit/strateg/fnih-spni_e.html#ahhri-irrhs
New web-links to the Communicable Disease Control Division (CDCD)
http://www.hc-sc.gc.ca/ahc-asc/branch-dirgen/fnihb-dgspni/phcphd-dsspsp/cdcd-dcmt_e.html
Contribution Agreements
http://www.hc-sc.gc.ca/fnih-spni/finance/agree-accord/index_e.html
Supplementary Information
Table 2: Sustainable Development Strategy
Health Canada's current Sustainable Development Strategy (SDS) entitled "A Path to Sustainability" is a three-year strategic plan containing commitments to integrate sustainable development principles and practices into our policies, programs and operations - recognizing the complex interrelationships between health, the environment, the economy, and a range of social elements.
Maintaining continuity with the previous strategy, SDS IV builds on the same three themes:
- Helping to create healthy social and physical environments
- Minimizing the environmental and health effects of the department's physical operations and activities
- Integrating sustainable development into departmental decision-making and management processes
Health Canada's Strategy contains 51measurable targets under these three long-term themes. The targets contribute to the department's strategic outcomes such as access to safe and effective health products and information, reduced health and environmental risks, safer living and working environments, and better health outcomes for First Nations and Inuit and other Canadians. Specific targets address priorities such as clean air, clean water, sustainable communities, protection of the environment and human health, food safety, procurement, training and awareness, environmental stewardship, and integrating sustainable development into departmental decision-making and management processes.
Our Strategy reflects Health Canada's commitment to a coordinated federal approach for the fourth round of departmental SDSs, a government-wide initiative, led by Environment Canada, to strengthen coherence and accountability across departmental sustainable development strategies. This collaborative effort resulted in a set of common federal sustainable development goals and an associated reporting format that will enable government-wide reporting on key federal sustainable development issues for the first time since the establishment of the SDS process.
Select targets from Health Canada's Sustainable Development Strategy 2007-10: A Path to Sustainability, are documented in the table below in addition to performance measures, and expected progress for the fiscal year April 1, 2008 - March 31, 2009. The targets are numbered as they are in the Health Canada Sustainable Development Strategy for ease of reference. Priorities for 2008-09 include:
- Implementation of the Air Quality Health Index in 7 Census Metropolitan Areas
- Publication of a complete Climate Change and Health Vulnerability Assessment
- Publication of Guidelines for Canadian Household Reclaimed Water
- Revised policies on Product-Specific Health Claims
- Establishment of procedures and mechanisms to phase-out low fuel-efficient vehicles in Health Canada's fleet
For additional information on targets found in Health Canada's Sustainable Development Strategy 2007-10: A Path to Sustainability, please contact the Office of Sustainable Development at osd@hc-sc.gc.ca
Six Federal Long-Term Sustainability Goals
I. Clean and secure water for people, marine and freshwater ecosystems
II. Clean air for people to breathe and ecosystems to function well
III. Reduce greenhouse gas emissions
IV. Communities enjoy a prosperous economy, a vibrant and equitable society, and a healthy environment for current and future generations
V. Sustainable development and use of natural resources
VI. Strengthen federal governance and decision-making to support sustainable development
Legend for the following table:
| SDS Departmental Goal: | ||
| SDS Departmental Targets: | Performance Measure from Current SDS: | Department's Expected Results for 2008-09: |
| Clean Air | ||
| Between 2007-10, Health Canada will work to improve management and communication of the health risks of indoor and outdoor air pollution, including fact sheets, finalization of the Air Quality Health Index (AQHI) and further development of the Air Health Indicator. (1.1.3) Supports Federal Goal II |
# of Census Metropolitan Areas where AQHI is implemented Air Health Indicator (AHI) developed and tested |
Finalize development and test the AHI by March 31, 2009 Report on AHI in the final Canadian Environmental Sustainability Indicators Report |
| During the course of SDS IV, Health Canada will develop tools and information materials, including a summary of the Climate Change and Health Vulnerability Assessment, to better prepare Canadians and health professionals to deal with potential health impacts associated with a changing climate. (1.1.4) Supports Federal Goal II |
Risk management tools developed and implemented (e.g. Heat alert systems) with partners and stakeholders Publication of completed Climate Change and Health Vulnerability Assessment on Health Canada web site |
Assessment and summary document published and distributed |
| Clean Water | ||
| During the period 2007-10, Health Canada will establish and/or implement strategies, including a national waterborne contamination and illness response protocol, to help address and prevent incidences of drinking water contamination across jurisdictions, including for small systems and in small, rural and remote communities. (1.2.5) Supports Federal Goal I |
Real-time alert and reporting system for Boil Water Advisories developed (with the Public Health Agency of Canada), pilot tested, launched, and adopted progressively by federal, provincial and territorial (F/P/T) governments Successful completion of pilot projects or strategies to address current gaps specific to small systems, and testing and adoption by Canadian jurisdictions |
Promote use through F/P/T channels Identification/evaluation of tools and strategies for small systems Expected Result by 2010: Provinces adopt a common set of strategies to address current gaps specific to small drinking water systems, leading to improved drinking water quality, particularly in rural and remote communities |
| During the course of SDS IV, Health Canada will develop and update a minimum of 5 water quality guidelines for specific contaminants annually, including drinking and recreational water. (1.2.6) Supports Federal Goal I |
# of guidelines for water quality approved and adopted by F/P/T committees | 5 guidelines/guidance documents for water quality approved by F/P/T committees Guidelines for Canadian Recreational Water Quality posted on Health Canada website for public consultation Publication of final version of Guidelines for Canadian Household Reclaimed Water |
| Sustainable Communities | ||
| By March 2010, Health Canada commits to having completed the assessment of 16 sites requiring investigation of suspected petroleum hydrocarbon contamination, as well as the remediation of the 18 known petroleum hydrocarbon contaminated sites (as of March 2006) for which the department is responsible at health facilities on reserves. (1.3.2) Supports Federal Goal IV |
Sites suspected, or known to have petroleum hydrocarbon contamination assessed and remediated, if required Assessment and remediation reports developed |
Remediate 10 petroleum hydrocarbon contaminated sites |
| During the course of SDS IV, at least one of Health Canada's regional offices will explore the opportunity to collaborate with provincial and municipal organizations in the development of sustainable development practices or initiatives. (1.3.17 Supports Federal Goal IV |
Number of car-pooling registrations on the internet site | The Dé¡rche All駯 project in the Qu颥c Region will encourage and promote better commuting habits with the car-pooling initiative |
| Protection of the Environment and Human Health | ||
| During the 2007-10 period, develop a regulatory framework based on policy and technical analysis of issues related to the development of Environmental Assessment Regulations for new substances contained in products regulated under the Food and Drugs Act. (1.4.2) | International analysis conducted Development of the regulatory framework data requirements for the Environmental Assessment Regulations |
Advancing the development of the regulatory framework for the Environmental Assessment Regulations for new substances contained in products regulated under the Food and Drugs Act Consultation on the regulatory frameworks for the Environmental Assessment Regulations Development of data requirements for the Environmental Assessment Regulations Consultation on the data requirements for Environmental Assessment Regulations |
| Food Safety | ||
| By March 2010, continue to establish policies and standards related to the nutritional quality of foods (Trans Fat, Food Fortification, Product-Specific Health Claims). (1.5.2) | Transfat Implemented action plan Food Fortification Number and type of regulations and policies developed and published |
Transfat Stakeholder support for and implement the plan of action established in 2007-08 Food Fortification A comprehensive policy on the discretionary fortification of foods Product-specific Health Claims Regulatory framework: revised policies and recommendations |
By March 2010, continue to:
|
Food borne Pathogens Number of educational documents published (i.e., Codes of Practice, policies, Internet postings) |
Food borne PathogensIncreased consumer awareness of risk avoidance practices for:
Implement a risk communication plan on risks to mercury in fish and benefits of fish consumption (target population at risk, women of child-bearing age and young children) Develop effective risk management approaches to reduce Canadian dietary exposure to low-level genotoxic carcinogens and other trace contaminants in food (i.e., environmental sources and food processing-induced) Develop integrated Food Chemical Surveillance plan through Health Canada's Food Chemical Safety Network, and set standards to limit exposure to selected natural toxins (e.g. oxytoxin A and vomitoxin DON) |
| Fleet | ||
| By March 2010, reduce greenhouse gas (GHG) emission per vehicle per kilometre. (2.1.1) Supports Federal Goal III |
Annual average GHG emissions per vehicle per kilometre reduced | Establish procedures and mechanisms to phase-out low fuel-efficient vehicles, increasing the use of ethanol-blended vehicles and hybrids Standardizing key fleet management processes |
| Building Energy | ||
| By March 2009, an action plan to reduce GHG emissions in Health Canada's custodial laboratories will be developed and implemented (2.2.2) Supports Federal Goal III and GGO Goal 'Other custodian departments and agencies will establish and report on meaningful departmental targets in support of the overal FHIO target for building by 2010' |
Level of GHG emissions | With consultation and cooperation with RPFMD Custodial Portfolio, establish baseline data, tracking mechanisms and develop action plan |
| Procurement | ||
| By March 2010, all materiel managers and procurement personnel will attend a recognized training course on green procurement offered by Public Works and Government Services Canada (PWGSC), Canada School of Public Service (CSPS), or any other federal government department. (2.3.2) Supports Federal Goal III |
Number of materiel managers and procurement personnel trained Increase in level of knowledge and awareness on green procurement |
Advertise Green Procurement training courses offered by PWGSC and CSPS to all materiel managers and procurement personnel Updated internal procurement training material Review action plan on green procurement training for the department and conduct a follow-up survey to assess increase in knowledge and awareness of green procurement |
| Training | ||
| Health Canada will join with other government departments and the CSPS to design and deliver new Government of Canada Sustainable Development training material. Delivery to begin by December 2007. (2.4.1) Supports Federal Goal VI |
Formal inclusion of the SD course (#727) in the CSPS program of studies Number of Health Canada employees that successfully complete the course |
In 2008, the new course will be part of the regular course offerings of the CSPS Policy analysts will be better trained in the application of SD principles in their particular domain |
| Integration | ||
| By the end of 2008, as a pilot project, Health Canada will apply a sustainable development lens to select policies and programs. (3.1.1) Supports Federal Goal VI |
Final revision and printing of the SD lens following the completion of a pilot Presentation of the lens and pilot results to management committees as a tool to be applied in the development of all planning documents including Memorandum to Cabinet and Treasury Board Secretariat submissions Inclusion of the lens in planning and policy development process |
In 2008-09, final revisions and improvements to the lens will be made based on feedback received from the pilot with the Health Policy Branch The lens will be introduced to senior management to secure support for its inclusion in planning and policy development process |
Update on the Health Canada Sustainable Development Policy Suite: Policies the Department has adopted to advance Sustainable Development
Sustainable Development Policy
Since its first SDS, Health Canada has worked to create a culture that recognizes the importance of sustainable development in the planning and delivery of our strategic outcomes. Approval of a Sustainable Development Policy in December 2000 enabled the integration of five key principles (shared responsibility, integrated approaches, equity, accountability, and continuous improvement) into the Department's third SDS and acted as guiding principles for the development of the fourth strategy covering the 2007 to 2010 period.
Environmental Management Systems
The importance of sound environmental management of our operations and activities was underscored in the creation of the 2000 Health Canada Environmental Management Policy and the development of the Environmental Management Systems (EMS) Division. The use of an EMS, a tool for managing compliance, minimizing negative impacts and risks to the environment, and for continual improvement, supports the principles of sustainable development. The priorities of the EMS Division include:
- maintaining and improving the departmental EMS including its supporting policies and database
- implementing actions in support of its own SDS targets
- fostering environmental stewardship
Strategic Environmental Assessment
While environmental management of operations and activities is done through the EMS process, Strategic Environmental Assessment (SEA) offers a systematic and comprehensive process for evaluating the environmental effects of a policy, plan or program and its alternatives, at the earliest stage of planning. Health Canada has a requirement to complete SEAs in conformance with the 2004 Cabinet Directive on the Environmental Assessment of Policy, Plan and Program Proposals. The Department has developed a policy and guidelines for SEA to support departmental efforts to ensure proposals with important positive or negative environmental effects are identified and that these effects are either enhanced or mitigated in the implementation of the policy, plan or program. SEA training is currently offered to Health Canada analysts in both the National Capital Area and in the regions.
In 2008-09, the Office of Sustainable Development (OSD) will revise the 2004 Health Canada Policy and Guidelines on Strategic Environmental Assessment to formalize new protocols and practices to improve SEA management in response to the recommendations made by the Commissioner of the Environment and Sustainable Development. The policy changes combined with aggressive, professional SEA training will ensure that environmental considerations are better reflected in all departmental documents submitted to the Minister or to Cabinet for decision.
The following additional tables can be found on the Treasury Board of Canada Secretariat's website at http://www.tbs-sct.gc.ca/rpp/2008-2009/info/info-eng.asp.
Details of Transfer Payment Programs
Evaluations
Foundations (Conditional Grants)
Green Procurement
Horizontal Issues
Internal Audits
Progress Toward the Department's Regulatory Plan
Services Received Without Charge
Sources of Respendable and Non-respendable Revenue
Status Report on Major Crown Projects
User Fees
Other Items of Interest
Health Canada's Regional Operations - An Overview
Health Canada provides programs and services to Canadians in every province and territory in the country. Over one-third of Health Canada's employees work in communities outside the National Capital Region.
This cross-country presence allows Health Canada to deliver national services in a way that responds to regional issues and priorities; to develop relationships, partnerships and local knowledge that contribute to both the design and the delivery of effective programs and policies; and to build on opportunities for local collaboration with other governments, partners and stakeholders.
In 2008-09, Health Canada's regional operations will help the department realize these benefits through ongoing collaborative and consultative work, as well as by delivering on the Department's mandate through regional projects and initiatives.
Regionally-Responsive Service Delivery
Health Canada's regional staff will continue to deliver programs and services that respond to the needs of people and communities across Canada.
They will conduct inspection and surveillance activities related to consumer products, controlled drugs and substances, pesticides, and health products , building on local knowledge of manufacturing, importing and retail activities in each region.
Health Canada staff will conduct locally-based risk assessments and evaluations and provide health advice to federal employees, provinces and municipalities related to chemical contaminants and exposure levels, drinking water standards and work environments.
They will work with First Nations and Inuit communities to provide community-based health promotion and disease prevention programs and Home and Community Care Programs, and to help these communities develop emergency preparedness plans suited to their unique needs.
The ACCES Project in Quebec Region is an example of an initiative that will deliver Health Canada's services to First Nations in a way that responds to client needs and issues. The project will designate an officer to serve as a "single window" for each community to access Health Canada. It will help communities improve their capacity to plan and implement health programs and will support improved management and accountability.
Regions will also continue to implement projects using the Aboriginal Health Transition Fund to improve the integration of provincial/territorial and federal health services, and better meet the needs of First Nations and Inuit communities.
Outreach and Information
Through outreach and collaboration with governments, clients and partners in the regions, Health Canada's regional staff makes a critical contribution to the Department's knowledge base, as well as building awareness among clients in the region and providing opportunities for local stakeholder input.
The regions will continue to fund disease surveillance for First Nations and Inuit communities as well as provide intelligence on local health policy and health systems issues to support departmental policy and program development.
They will carry out consultations and share knowledge and intelligence, both to provide information to stakeholders, and to gather and assess regional views and interests in order to help shape national policies and regulations.
In the Atlantic Region, for example, Health Canada will work with a policy network of Health Portfolio partners to coordinate a horizontal approach to regional mental health initiatives. In addition, the Atlantic and Alberta regions will work together on a five-year initiative to develop and improve population health surveillance capacity for First Nations.
Collaboration with Regional Partners
In a number of regions, Health Canada is entering into collaborative agreements with partners to ensure that services are integrated to cover a range of clients' needs. The Tripartite First Nations Health Plan in the British Columbia (BC) region is an example of collaboration among the provincial government, Health Canada and First Nations that will move into its implementation phase in 2008.
The tripartite model is being adapted and pursued in other regions. For example, in the Ontario region, a First Nations Public Health Initiative will be established among the provincial Ministry of Health and Long-Term Care, the Chiefs of Ontario and Health Canada to promote an integrated approach to public health on reserve.
In the Northern region, similarly, collaboration will help improve accountability and client care. The Northern region will continue to collaborate with the Government of Nunavut, through a co-management forum, to identify priorities for the administration and delivery of the Non-Insured Health Benefits program in Nunavut, and to foster a dialogue on issues of shared concern.
The Product Safety Program is another area in which collaboration with local partners improves effectiveness. Health Canada's officers will continue to work with counterparts from Environment Canada and the Canada Border Services Agency to increase their scope for inspection and detection of unsafe products entering Canada.
The National Anti-Drug Strategy also creates opportunities for partners in the regions to work together to provide an appropriate range of services. In the BC region, Health Canada will work with provincial and municipal partners to develop innovative and integrated programming to address issues of addiction and substance abuse.
Through activities such as these, matching national directions to local conditions and opportunities, Health Canada's regional presence will continue to help the Department to maximize the reach and effectiveness of its programs and resources.
Advancing the Science Agenda
Over the past few years, Health Canada has strengthened its ability to perform and use science. Particular attention has been given to addressing the following priority areas:
- Science advice - Promoting the effective use of science in policy making: Assisting the Department in employing quality science advice in its policy and regulatory decisions;
- Science management - Enhancing science capacity and quality: Encouraging due diligence and ensuring Health Canada has the science capacity it needs to meet current and emerging challenges; and
- Science promotion - Raising awareness and understanding of science conducted at Health Canada: Improving stakeholder and public understanding of departmental science and its contribution to the health and safety of Canadians.
Science Advice
The Department will consider how best to implement the recommendations from a 2007-08 review of the Departmental Science Advisory Board to ensure the continued effectiveness of external science advice to the Minister.
As a science-based department, Health Canada must ensure that its research is conducted in a responsible manner. Health Canada's Research Ethics Board (REB), an independent body of experts, will continue to ensure that research involving humans that is funded or conducted by the Department meets the highest ethical standards. Other planned activities include the provision of research ethics training to researchers in the Department and in the Public Health Agency of Canada (PHAC), and the development of a Health Canada science integrity policy to strengthen the oversight and governance of departmental science and research.
Science Management
Mobilizing Science and Technology (S&T) to Canada's Advantage, the Government of Canada's new S&T Strategy (federal Strategy), provides a multi-year framework for action aimed at strengthening the contribution that S&T make to Canada's economic development and social well-being. Health and related life sciences and technologies is identified as a priority sector within the federal Strategy. Health Canada will continue to coordinate departmental and Health Portfolio (Canadian Institutes of Health Research and PHAC) implementation of the health-related policy commitments of the federal Strategy. In addition, Health Canada will continue to work with members of the federal S&T community to develop government-wide S&T initiatives. Health Canada will continue to champion the federal S&T community's pilot Scientists as Leaders Development Program. Health Canada is currently working with other departments to evaluate the pilot and determine the feasibility and value of continuing the Program.
The Department will finalize and implement a Health Canada S&T Strategy. The departmental Strategy, which provides a policy framework for the identification of science priorities over the next five to ten years, will enable the Department to be more responsive to the needs of Canadians. The Department will also develop improved internal approaches for better linking science, science policy and policy development.
Northern S&T has achieved significant prominence in Canada in recent years and the 2007 Speech from the Throne further strengthened the Government of Canada's commitment to this issue. Health Canada will develop a departmental plan for addressing Northern S&T issues and will identify opportunities to participate in Government of Canada activities in this area.
The Department will continue to pursue strategic partnerships and linkages with external partners/stakeholders to access the science and augment the science capacity needed to fulfill the Department's regulatory and policy responsibilities.
Health Canada will continue its efforts to support the recruitment, retention, development, and promotion of scientists. The Department will continue to enhance its Post-Doctoral Fellowship Program, as well as departmental participation in the Natural Sciences and Engineering Research Council's Visiting Fellowships Program, to ensure they remain responsive to departmental needs.
Science Promotion
Health Canada will continue its efforts to raise awareness and understanding of the science and research it conducts and uses, including through frequent updates to relevant pages on the departmental Internet site. The Department will host its annual Science Forum to showcase the work of its scientists and researchers and discuss the cutting-edge science that informs policy and regulatory decision making. The Forum will also facilitate linkages and information sharing between departmental researchers and decision makers and counterparts from across Canada.
Details of Transfer Payment Programs
| Contributions for Bigstone Non-Insured Health Benefits Pilot Project (Voted) | ||||
|---|---|---|---|---|
| Start Date: April 2005 | End Date: March 2010 | |||
| Description: Administration and delivery of benefits with Bigstone Health Commission to registered Indians and recognized Inuit | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected Results: Improved access to quality well-coordinated culturally appropriate primary health care programs and services for First Nations and Inuit individuals, families and communities | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 8.50 | 8.80 | 8.80 | 9.10 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 8.50 | 8.80 | 8.80 | 9.10 |
| Planned Evaluations: N/A | ||||
| Planned Audits: N/A | ||||
| Contributions for First Nations and Inuit Community Programs (Voted) | ||||
| Start Date: April1,2005 | End Date: March 2010 | |||
| Description: Community programs support child and maternal-child health; mental health promotion; addictions prevention and treatment; chronic disease prevention and health promotion services | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected Results: The expected results are: increased participation of First Nations and Inuit individuals, families, and communities in programs and supports and improved continuum of programs and services in First Nations and Inuit communities | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 220.30 | 235.70 | 240.30 | 168.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 220.30 | 235.70 | 240.30 | 168.00 |
| Planned Evaluations: Children and Youth Cluster evaluation to be initiated in winter 2008-09 | ||||
| Planned Audits: Recipients are required to provide year end audited financial statements. Contribution compliance audits are conducted every year for a sample of recipients. | ||||
| Contributions for First Nations and Inuit Health Benefits (Voted) | ||||
| Start date: April 2005 | End date: March 2010 | |||
| Description: A limited range of medically necessary health-related goods and services which supplement those provided through other private or provincial and territorial health insurance plans is provided to registered Indians and recognized Inuit. Benefits include drugs, dental care, vision care, medical supplies and equipment, short-term crisis intervention mental health services, and transportation to access medical services not available on reserve or in the community of residence. | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected Results: Access by eligible clients to Non-Insured Health benefits | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 140.60 | 135.40 | 139.30 | 143.40 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 140.60 | 135.40 | 139.30 | 143.40 |
| Planned Evaluations: N/A | ||||
| Planned Audits: Recipients are required to provide year end audited financial statements. Contribution compliance audits are conducted every year for a sample of recipients. | ||||
| Contributions for First Nations and Inuit Health Facilities and Capital Program (Voted) | ||||
| Start Date: April 2005 | End Date: March 2010 | |||
| Description: Provides funding to eligible recipients for the construction, acquisition, leasing, operation and maintenance of nursing stations, health centres, health stations, health offices, treatment centres, staff residences, and operational support buildings. | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected Results: Increase availability of health facilities, equipment and other moveable assets in First Nations and Inuit communities that support the provision of health services | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 55.10 | 46.50 | 47.30 | 47.30 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 55.10 | 46.50 | 47.30 | 47.30 |
| Planned Evaluations: N/A | ||||
| Planned Audits: Contribution compliance audits are conducted every year for a sample of recipients. | ||||
| Contributions for First Nations and Inuit Health Governance and Infrastructure Support (Voted) | ||||
| Start Date: April 2005 | End Date: March 2010 | |||
| Description: Governance and infrastructure support for the First Nations and Inuit Health System | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected Results: Improved health status of First Nations and Inuit through strengthened governance and infrastructure | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 179.60 | 191.50 | 201.20 | 146.60 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 179.60 | 191.50 | 201.20 | 146.60 |
| Planned Evaluations: N/A | ||||
| Planned Audits: Contribution compliance audits are completed every year for a sample of recipients. | ||||
| Contributions for First Nations and Inuit Health Protection (Voted) | ||||
| Start Date: April 2005 | End Date: March 2010 | |||
| Description: Communicable Disease and Environmental Health and Research programs facilitate preparedness to implement measures in the control, management and containment of outbreaks of preventable diseases and improve management and control of environmental hazards. | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected Results: Environmental health risk management continues to improve the health status of FirstNations individuals, families and communities and improve access to quality, well-coordinated communicable disease prevention and control programs for First Nations and Inuit individuals and communities | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 12.50 | 11.10 | 11.12 | 10.60 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 12.50 | 11.10 | 11.12 | 10.60 |
| Planned Evaluations: Communicable Disease Control Cluster Evaluation to be initiated in winter 2008. Environmental Health and Research Cluster Evaluation to be initiated in winter 2008. | ||||
| Planned Audits: Recipients are required to provide year end audited financial statements. Contribution compliance audits are conducted every year for a sample of recipients. | ||||
| Contributions for First Nations and Inuit Primary Health Care (Voted) | ||||
| Start date: April 2005 | End date: March 2010 | |||
| Description: Primary Health Care services include emergency and acute care health services, and community primary health care services which include illness and injury prevention and health promotion activities. These programs also include: the First Nations and Inuit Home and Community Care; and the Oral Health Strategy. | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected Results: Improved access to quality well-coordinated culturally appropriate primary health care programs and services for First Nations and Inuit individuals, families and communities | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 120.90 | 122.20 | 123.30 | 124.60 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 120.90 | 122.20 | 123.30 | 124.60 |
| Planned Evaluations: N/A | ||||
| Planned Audits: Recipients are required to provide year end audited financial statements. Contribution compliance audits are conducted every year for a sample of recipients. | ||||
| Contributions to the Organization for the Advancement of Aboriginal People's Health (Voted) | ||||
| Start date: April 2005 | End date: March 2010 | |||
| Description: To support the Organization for the Advancement of Aboriginal People's Health | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected results: Continued empowerment of Aboriginal peoples through advancements in knowledge and sharing of knowledge on Aboriginal health | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 5.00 | 5.00 | 5.00 | 5.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 5.00 | 5.00 | 5.00 | 5.00 |
| Planned Evaluations: N/A | ||||
| Planned Audits: N/A | ||||
| Grant for the Territorial Health Access Fund and Operational Secretariat (Voted) | ||||
| Start date: April 2005 | End date: March 2010 | |||
| Description: Grant for the territorial Health Access Fund and Operational Secretariat | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
Expected results:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 15.00 | 15.00 | 15.00 | 0.00 |
| Total Contributions | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 15.00 | 15.00 | 15.00 | 0.00 |
| Total Program Activity | 0.00 | 0.00 | 0.00 | 0.00 |
| Planned Evaluations: Evaluation planned for March 2008. Note: This is a recipient (Government of Yukon) evaluation responsibility. | ||||
| Planned Audits: N/A | ||||
| Grant for the Territorial Medical Travel Fund (Voted) | ||||
| Start date: April 2005 | End date: March 2010 | |||
| Description: To support the medical travel fund | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
Expected results:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 15.00 | 15.00 | 15.00 | 0.00 |
| Total Contributions | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 15.00 | 15.0 | 15.00 | 0.00 |
| Planned Evaluations: N/A | ||||
| Planned Audits: N/A | ||||
| Grant to the Canadian Blood Services (Voted) | ||||
| Start date: April 2000 | End date: Ongoing | |||
| Description: To support basic, applied and clinical research on blood safety and effectiveness issues through the auspices of Canadian Blood Services. | ||||
| Strategic Outcome: Access to Safe and Effective Health Products and Food and Information for Healthy Choices | ||||
| Expected results: Continued improvements to basic applied and clinical research on blood safety and effectiveness | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Access to Safe and Effective Health Products and Food and Information for Healthy Choices | ||||
| Total Grants | 5.00 | 5.00 | 5.00 | 5.00 |
| Total Contributions | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 5.00 | 5.00 | 5.00 | 5.00 |
| Planned Evaluations: Health Canada is not planning to do any evaluations of this activity | ||||
| Planned Audits: A separate audit of this grant is not planned at this time. | ||||
| Grant/Contribution in Support of the Federal Tobacco Control Strategy (Voted) (Voted) | ||||
| Start date: April1,2001 | End date: March31,2011 | |||
| Description: A transfer payment program in support of the Federal Tobacco Control Strategy designed to develop and test tobacco cessation and prevention techniques and approaches and to transfer this knowledge to stakeholders with the intention of changing behaviour. Contributions are provided to support the provinces and territories as well as key national and regional non-governmental organizations and others in order to help build a strong knowledge base and ongoing capacity for developing effective tobacco prevention and cessation interventions. The grant portion of the program is designed to support international tobacco control efforts. | ||||
| Strategic Outcome: Reduced health and environmental risks from products and substances, and safer living and working environments | ||||
Expected results:Supporting the attainment of a smoking prevalence rate in Canada of 12% by 2011 by:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Substance use and abuse | ||||
| Total Grants | 0.50 | 0.50 | 0.50 | 0.50 |
| Total Contributions | 6.50 | 15.80 | 15.80 | 15.80 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 7.00 | 16.30 | 16.30 | 16.30 |
| Planned Evaluations: Summative evaluation of FTCS including G's&C's in 2010-11 | ||||
| Planned Audits: To be determined - individual recipients' audit plans developed annually | ||||
| Contribution for Alcohol and Drug Treatment Rehabilitation Program (Voted) | ||||
| Start date: April1,1997 | End date: March31,2008 | |||
| Description: The Alcohol and Drug Treatment and Rehabilitation Contribution Program (ADTR) is an A-base component of the former Canada's Drug Strategy (CDS) which supported the federal government's efforts to reduce substance use and the harm associated with the abuse of alcohol and other drugs to individuals, families and communities. It provides cost-shared funding to participating provinces and territories through negotiated bi-lateral federal/provincial/territorial (F/P/T) contribution agreements. The ADTR Program ends March31,2008 and the funding will roll into the new Drug Treatment Funding Program (DTFP) effective April1,2008. | ||||
| Strategic Outcome: Reduced health and environmental risks from products and substances, and health, sustainable living and working environments | ||||
| Expected results: The 2005-06 review of the ADTR Program will result in a re-focussing of the Program effective April1,2008. | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Substance use and abuse | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 13.2 | 0.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 13.2 | 0.00 | 0.00 | 0.00 |
| Planned Evaluations: No further evaluations planned as this Program ends March31,2008. | ||||
| Planned Audits: No planned audits due to the low risk nature of the Program | ||||
| Contribution to the Drug Treatment Funding Program (Voted) | ||||
| Start date: April1,2007 | End date: March31,2012 | |||
| Description: The aim of the Drug Treatment Funding Program (DTFP) will be to provide the incentive (seed funding) for provinces, territories and key stakeholders to initiate projects that will lay the foundation for systemic change (reoriented ADTR funds) leading to sustainable improvement in the quality and organization of substance abuse treatment systems. At the same time that provincial and territorial governments are working to achieve these system-level efficiencies, five-year time limited funding (new funds) will be available for the delivery of treatment services to meet the critical illicit drug treatment needs of at-risk youth in high needs areas. | ||||
| Strategic Outcome: Reduced health and environmental risks from products and substances, and healthy, sustainable living and working environments | ||||
Expected results:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Substance use and abuse | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions (* NADS funding received in 2007-08 is $3.9 million. There is a vote transfer in Supps B of $500,000 from DTFP contributions to O&M.) |
3.4* | 28.7 | 29.2 | 25.8 |
| Total Other Types of Transfer Payments | 0.00 | -1.9 | -1.9 | -1.9 |
| Total Program Activity | 3.4 | 26.8 | 27.3 | 23.9 |
| Planned Evaluations: A Program level Performance Management and Evaluation Plan has been developed in collaboration with provinces and territories. An interim evaluation will be conducted by Health Canada in 2009-10. This interim evaluation will focus on DTFP program design and implementation progress and preliminary results. A final evaluation will be conducted in 2011-12. | ||||
| Planned Audits: A Program level RBAF has been developed and sets out the risks and mitigation strategy for the Program. An audit plan will be developed once the Program is implemented. | ||||
| Contributions for the Drug Strategy Community Initiatives Fund (Voted)) | ||||
| Start date: April1,2004 | End date: Ongoing | |||
| Description: The Drug Strategy Community Initiatives Fund (DSCIF) managed by Health Canada, provides support for Health Promotion and Prevention projects related to substance use and abuse. The DSCIF contributes to the achievement of the National Anti-Drug Strategy through the provision of financial assistance for initiatives that address a broad range of illicit drug use issues and the underlying factors associated with illicit drug use. | ||||
| Strategic Outcome: Reduced health and environmental risks from products and substances, and health,sustainable living and working environments | ||||
Expected results:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Substance use and abuse | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions (* Annual budget for DSCIF is $9.615 million. Due to a delay in a Call for Proposals in 2007-08, $1.789 million was reprofiled to fiscal year 2008-09 and there is a vote transfer in Supps B of $900,000 from DSCIF contributions to O and M **This amount includes the re-profiled $1.789 million from 2007-08.) |
6.9* | 11.4** | 9.6 | 9.6 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 6.9* | 11.4 | 9.6 | 9.6 |
| Planned Evaluations: A DSCIF evaluation is underway which includes case studies and validation of refocused DSCIF performance evaluation framework. This work will feed into the renewal of the Program's Terms and Conditions in 2009-10. | ||||
| Planned Audits: Audit processes for eight funded projects are underway in 2007-08 via a Memorandum of Agreement with Audit Services Canada. | ||||
| Grant to the Canadian Agency for Drugs and Technology in Health (Voted) | ||||
| Start date: April1,2008 | End date: March31,2013 | |||
| Description: The Canadian Agency for Drugs and Technologies in Health (CADTH, previously known as the Canadian Coordinating Office for Health Technology Assessment or CCOHTA) is an independent, not-for-profit agency funded by Canadian federal, provincial, and territorial governments to provide credible, impartial advice and evidence-based
information about the effectiveness of drugs and other health technologies to Canadian health care decision makers. The first 5-year Funding Agreement with CADTH ends on March31,2008. Plans are underway for a renewed five year agreement with CADTH starting April1,2008 for an annual value of up to $16.9 million. The purpose of the Named Grant is to provide financial assistance to support CADTH's core business activities, namely, the Common Drug Review (CDR), Health Technology Assessment (HTA), and the Canadian Optimal Medication Prescribing and Utilization Service (COMPUS). |
||||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||||
Expected results:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Canadian Health System | ||||
| Total Grants | 17.4 | 16.9 | 16.9 | 16.9 |
| Total Contributions | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 17.4 | 16.9 | 16.9 | 16.9 |
| Planned Evaluations: A requirement of Health Canada's funding agreement with CADTH is that an independent, comprehensive evaluation of the overall program is to be conducted approximately one year prior to the end of the agreement (March31,2013). | ||||
| Planned Audits: N/A | ||||
| Contributions for the Health Care Strategies and Policy Contribution Program (Voted) | ||||
| Start date: September 2002 | End date: March31,2008 | |||
| Description: To support the federal government's interests in achieving an accessible, high quality, sustainable and accountable health system adaptable to the needs of Canadians. The contribution program will support efforts to stimulate and facilitate health care policy analysis and development to advance strategic thinking and policy options in priority areas. Current priorities include, but are not limited to: health human resources, patient wait times and mental health. The contribution program is slated to end March31,2008 and continued funding is pending further consideration. | ||||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||||
Expected results:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Canadian Health System | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 45.1 | 51.9 | 48.0 | 34.7 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 45.1 | 51.9 | 48.0 | 34.7 |
| Planned Evaluations: A summative evaluation of the program, including all initiatives, will be completed for presentation to Treasury Board Secretariat by March31,2008, as required to support the renewal of the program's Terms and Conditions. | ||||
| Planned Audits: N/A | ||||
| Named Grant to the Canadian Patient Safety Institute (Voted) | ||||
| Start date: December10,2003 | End date: March31,2008 | |||
| Description: The Named Grant to the Canadian Patient Safety Institute (CPSI) supports the federal government's interest (in a F/P/T partnership context) in achieving an accessible, high quality, sustainable and accountable health system adaptable to the needs of Canadians. It is designed to improve the quality of health care services by strengthening system coordination related to patient safety, including promoting national collaboration among key players. The first five-year Funding Agreement with CPSI ends on March31,2008. Plans are underway for a renewed agreement with CPSI starting April1,2008. Health Canadahas ongoing funding authority of up to $8 million per year for the CPSI Named Grant. | ||||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||||
Expected results:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Canadian Health System | ||||
| Total Grants | 7.60 | 8.00 | 8.00 | 8.00 |
| Total Contributions | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 7.60 | 8.00 | 8.00 | 8.00 |
| Planned Evaluations: N/A | ||||
| Planned Audits: N/A | ||||
| Canadian Partnership Against Cancer Corporation (Voted) | ||||
| Start date: April1,2007 | End date: March31,2012 | |||
| Description: The Canadian Strategy for Cancer Control (CSCC) is a five-year plan with the following objectives: (1) to reduce the expected number of new cases of cancer among Canadians; (2) to enhance the quality of life of those dying of cancer; and (3) to lessen the likelihood of Canadians dying from cancer. Health Canada is the federal liaison with the Canadian Partnership Against Cancer, the not-for-profit organization responsible for the implementation of the CSCC, and is responsible for managing the $50 million per year grant agreement with the corporation. | ||||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||||
Expected results:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Canadian Health System | ||||
| Total Grants | 50.00 | 50.00 | 50.00 | 50.00 |
| Total Contributions | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 50.00 | 50.00 | 50.00 | 50.00 |
| Please note: A request to re-profile $20.7 million of the forecasted spending for 2007-08 is pending further consideration. | ||||
| Planned Evaluations: N/A | ||||
| Planned Audits: N/A | ||||
| Named Grant to the Health Council of Canada (Voted) | ||||
| Start date: September 2004 | End date: March31,2008 | |||
| Description: The Health Council of Canada (the Council) was established out of the 2003 First Ministers' Accord on Health Care Renewal to monitor and make annual public reports on the implementation of the Accord. In the 2004 Health Accord, First Ministers extended the role of the Council to report on the health status of Canadians and health
outcomes. Specifically, the Council has four primary roles: (1) monitoring and public reporting on health care renewal; (2) monitoring and analyzing health care renewal, barriers and health outcomes; (3) highlighting innovation; and (4) engaging Canadians. The Health Council is governed by its Corporate Members (F/P/T Ministers of Health except those of Québec and Alberta) and up to 27 governmental and non-governmental Councillors. The Council's current five-year funding agreement expires on March31,2008 and is being extended for one year, effective April1,2008. The one-year extension will provide the Council with funding certainty for 2008-09. During this extension period, Corporate Members will confirm the most appropriate direction for the future of the Council, in the context of the ongoing five-year review. |
||||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||||
| Expected results: Through monitoring and annual public reporting on the progress achieved in implementing the 2003 First Ministers' Accord and the 2004 Health Accord, the Council will contribute to enhancing accountability and transparency in health system care reform. | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Canadian Health System | ||||
| Total Grants | 6.40 | 10.00 | 10.00 | 10.00 |
| Total Contributions | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 6.40 | 10.00 | 10.00 | 0.00 |
| Planned Evaluations: The Corporate Members of the Council are undertaking a review of the Council's scope, mandate, role, objectives, effectiveness and continued relevance, as required by the Council's by-laws to occur in the fourth year of each five-year mandate. Following the review, the Corporate Members will decide the future direction of the Council. The Council is also developing an evaluation framework to measure, internally, its performance. | ||||
| Planned Audits: N/A | ||||
| Contribution Program to Improve Access to Health Services for Official Language Minority Communities (Voted) | ||||
| Start date: June 2003 | End date: March 2009 | |||
| Description: The Contribution Program to Improve Access to Health Services for Official Language Minority Communities was launched in June 2003, following the 2003 federal budget and The Action Plan for Official Languages. The Program was approved for a five-year period (2003-04 to 2007-08) with a total budget of $89 million, and with ongoing
annual funding of $23 million thereafter. The Program is managed by the Official Language Community Development Bureau, and provides funding to French-speaking and English-speaking official language minority communities in Canada under two areas: (1) Networking Support and (2) Support for Training and Retention of Health Professionals. The Networking Support component provides funds for the establishment and sustainability of networks that will mobilize the capacities of institutions, health professionals and communities to encourage health stakeholders to deliver services in the official language of their choice; foster the development of solid, durable links between health sector stakeholders; mitigate the geographic dispersal of communities; and promote greater community engagement. These networks will facilitate information-sharing and resource development, which will lead to new ways of improving access to health services for official language minority communities. Funding provided to Francophone minority communities under the Support for Training and Retention of Health Professionals component is meant to increase the number of practising Francophone health professionals in minority communities through improved access to available programs and the extension of such training across the country via participating educational institutions. This also includes media-based and distance training, and capacity-building within institutions that offer training to health professionals within Francophone minority communities. The funds directed to support the training and retention of health professionals in Anglophone minority communities will promote professional training and language training in the official language of minority communities, particularly in the regions of Quebec. It will also support regional incentive measures for the recruitment and retention of health professionals, to encourage them to move to the regions or remain there. In July 2007, the Minister of Health approved a one-year extension of the Contribution Program for 2008-09. This one-year transition period will allow current initiatives to continue while work is undertaken to renew the Program in the context of a new federal Action Plan on official languages. |
||||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||||
Expected results:
Specific Results by Program Component: 1) Networking Support component:
2) Support for Training and Retention of Health Professionals component:
|
||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: Canadian Health System | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 26.80 | 23.00 | 0.00 | 0.00 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 26.80 | 23.00 | 0.00 | 0.00 |
| Planned Evaluations: Formative (mid-term) evaluation is expected for December 2007. A summative (final) evaluation is expected in June 2008. | ||||
| Planned Audits: N/A | ||||
| Payments to First Nations and Inuit Health Services Transfer (Voted) | ||||
| Start date: April 2007 | End date: March 2012 | |||
| Description: To increase responsibility and control by Indian communities of their own health care and to effect improvement in the health conditions of Indian people | ||||
| Strategic Outcome: Better health outcomes and reduction of health inequalities between First Nations and Inuit and other Canadians | ||||
| Expected Results: Increased control or accountability by First Nations communities of health care services. | ||||
| ($ millions) | Forecast Spending 2007-08 | Planned Spending 2008-09 | Planned Spending 2009-10 | Planned Spending 2010-11 |
| Program Activity: First Nations and Inuit Health Programming and Services | ||||
| Total Grants | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Contributions | 227.90 | 233.90 | 241.20 | 248.70 |
| Total Other Types of Transfer Payments | 0.00 | 0.00 | 0.00 | 0.00 |
| Total Program Activity | 227.90 | 233.90 | 241.20 | 248.70 |
| Planned Evaluations: There are no specific Program related evaluation plans for 2007-08. However, directed Program funding and services will be evaluated within their designated cluster. In particular, any funding or services delivered through the Children and Youth, Communicable Disease Control or Environmental Health and Research clusters will be included in their 2008-09 cluster evaluation plans. | ||||
| Planned Audits: N/A | ||||
Evaluations
| Name of Evaluation | Evaluation Type | Status | Expected Completion Date |
|---|---|---|---|
| Fiscal Year 2008-09 | |||
| Gender Based Analysis Implementation Strategy | Summative Program Evaluation | Planned | March 2009 |
| Improving access to health services of Official Languages Minority Communities | Summative Evaluation (Contributions) |
Ongoing | Fall 2008 |
| Therapeutics Access Strategy | Formative Evaluation | Ongoing | Summer 2008 |
| Human Drugs | Strategic Program Evaluation | Ongoing | Fall 2009 |
| Food Safety and Nutrition Quality | Strategic Program Evaluation | Ongoing | Fall 2008 |
| Nutrition Promotion | Strategic Program Evaluation | Planned | Fall 2010 |
| Natural Health Products Program | Summative Evaluation (Grants and Contributions) |
Ongoing | March 2009 |
| Medical Devices | Strategic Program Evaluation | Planned | March 2010 |
| Access to Medicines Program | Summative Program Evaluation | Planned | Fall 2009 |
| Augmenting Health Canada's Response to Bovine Spongiform Encephalopathy (BSE) | Summative Program Evaluation | Planned | March 2010 |
| Federal Contaminated Sites Action Plan | Summative Evaluation | Planned | March 2010 |
| Drug Strategy Community Initiative Fund | Evaluation (renewal of Contributions) |
Ongoing | March 2009 |
| Federal Drinking Water Compliance Program | Evaluation of Compliance Program | Ongoing | Summer 2008 |
| Building Public Confidence in Pesticide Regulation and Improving Access to Pest Management Products | Summative Program Evaluation | Planned | December 2009 |
| First Nations and Inuit Home and Community Care Program | Summative Evaluation (Contributions) |
Ongoing | Fall 2008 |
| Children and Youth for First Nations and Inuit | Relevance/Effectiveness Evaluation (Contributions) |
Ongoing | March 2009 |
| Communicable Disease Control for First Nations and Inuit | Relevance/Effectiveness Evaluation (Contributions) |
Ongoing | March 2009 |
| Chronic Disease and Injury Prevention for First Nations and Inuit | Relevance/Effectiveness Evaluation (Contributions) |
Planned | March 2010 |
| Non-Insured Health Benefits for First Nations and Inuit | Relevance/Effectiveness Evaluation (Contributions) |
Planned | March 2010 |
| Health Facilities and Capital Program for First Nations and Inuit | Relevance/Effectiveness Evaluation (Contributions) |
Planned | March 2010 |
| Fiscal Year 2009-10 | |||
| Biologics | Strategic Program Evaluation | Planned | Fall 2011 |
| Veterinary Drugs | Strategic Program Evaluation | Planned | March 2011 |
| Pilot Heat Alert and Response Systems | Evaluation (Component of Clean Air Agenda) |
Planned | Spring 2010 |
| Air Quality Health Index | Evaluation (Component of Clean Air Agenda) |
Planned | Spring 2010 |
| Protecting Canadians and the Environment from Toxic Substances through a Chemicals Management Plan | Implementation Evaluation (Horizontal) |
Planned | March 2011 |
| Drug Treatment Funding Program of the National Anti-Drug Strategy | Formative Evaluation (Contributions) |
Planned | March 2010 |
| Continued Implementation of the Labrador Innu Comprehensive Health Strategy | Summative Evaluation (Contributions) |
Planned | March 2010 |
| Mental Health Addictions for First Nations and Inuit | Relevance/Effectiveness Evaluation (Contributions) |
Planned | March 2011 |
| Environmental Health and Research for First Nations and Inuit | Relevance/Effectiveness Evaluation (Contributions) |
Ongoing | March 2011 |
| Fiscal Year 2010-11 | |||
| Health Care Policy Contribution Program | Summative Evaluation (Contributions) |
Planned | August 2012 |
| Federal Tobacco Control Strategy | Summative Evaluation (Contributions) |
Planned | March 2011 |
| Drug Treatment Funding Program of the National Anti-Drug Strategy | Summative Evaluation (Contributions) |
Planned | January 2012 |
| Health Transfer Services, First Nations and Inuit | Relevance/Effectiveness Evaluation (Grants and Transfer Payment) |
Planned | March 2012 |
| Health Governance and Infrastructure | Relevance/Effectiveness Evaluation (Contributions) |
Planned | March 2012 |
| Primary Care for First Nations and Inuit | Relevance/Effectiveness Evaluation (Contributions) |
Planned | March 2012 |
| Electronic Link to Internal Evaluation Plan: http://www.tbs-sct.gc.ca/rma/database/newdeptview_e.asp?id=41 Treasury Board of Canada Secretariat - Audits and Evaluations by Department |
|||
Foundations (Conditional Grants)
| Canadian Institute for Health Information (CIHI) | ||
|---|---|---|
| Start Date: April 1, 2007 | End Date: March 31, 2012 | Total Funding: 404.49 million |
| Description: CIHI is an independent, not-for-profit organization supported by federal/provincial/territorial (F/P/T) governments that provides essential data and analysis on Canada's health system and the health of Canadians. CIHI was created in 1991 by the F/P/T Ministers of Health to address significant gaps in health information. CIHI's data
and its reports inform health policies, support the effective delivery of health services and raise awareness among Canadians about the factors that contribute to good health. Since 1999, the federal government has provided funding to CIHI through a series of grants and conditional grants, known as the "Roadmap Initiatives". More recently, CIHI's funding has been consolidated through the Health Information Initiative. CIHI was provided with approximately 260 million between 1999 and 2005 through the Roadmap Initiatives (I, II and II+). Budget 2005 allocated an additional 110 million over five years (2005-06 to 2009-10) to CIHI through Roadmap III. This funding has allowed CIHI to provide quality and timely health information, including the delivery of data on a variety of important health indicators and other health publications to support health sector decision-making and improve accountability. Beginning in 2007-08, the Health Information Initiative will provide ongoing conditional grant funding to CIHI replacing the previous Roadmap II, II+, and III funding and will also provide additional funds for new initiatives. This funding will allow CIHI to continue important work under the Roadmap Initiatives and to further enhance the coverage of health data systems. This will allow Canadians to obtain information about their health care system, including information on wait times, and it will support the development of comparable health indicators. The funding will also enable CIHI to respond effectively to emerging priorities. CIHI is currently in the process of completing national stakeholder consultations to renew their strategic directions and priorities. The results of the consultations will be factored into future corporate plans and directions. Under this initiative, up to 404.49 million will be delivered to CIHI over five years (2007-08 to 2011-12). |
||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||
|
Summary of Annual Plans of Recipient: The 2008-09 Operational Plan and Budget will be presented for approval to the Board of Directors at a March 2008 meeting, and subsequently submitted to the Minister of Health. At this time, the key projects and initiatives will be confirmed. A preliminary overview of key projects and initiatives includes:
|
||
| Planned Evaluations: A third-party evaluation of the first phase of the Roadmap Initiative was previously completed and CIHI was found to have met its objectives. An evaluation of the second phase was recently undertaken and the final results will soon be submitted to Health Canada. The next evaluation will be conducted after the completion of the Health Information Initiative in 2010. |
||
| Planned Audit: An audit was recently completed by Health Canada internal auditors on the Health Information Contribution Program. Auditors concluded that, in general, the internal controls in place at CIHI to manage the Contribution Agreement were sufficient to meet the terms and conditions of the Agreement with minor exceptions. Risk exposure was not serious. | ||
| URL of Recipient Site: http://secure.cihi.ca/cihiweb/splash.html The Canadian Institute for Health Information (CIHI) |
||
| Canada Health Infoway (Infoway) | ||
| Start Date: March 9, 2001 | End Date: Not applicable | Total Funding: 1.6 billion |
| Description: Canada Health Infoway is a federally-funded, independent, not-for-profit corporation with a mandate to accelerate the development and adoption of electronic health information systems with compatible standards and communications technologies on a pan-Canadian basis. Health information and communications technologies such as
electronic health records (EHRs), telehealth and public health surveillance systems will significantly improve access to health care services, patient safety, quality of care and productivity. Infoway collaborates with F/P/T governments towards a common goal of modernizing Canada's health information systems. This collaborative approach reduces overall costs by coordinating efforts, avoiding duplication, taking advantage of economies of scale, replicating successful initiatives across the country, and sharing best practices. For example, Infoway's EHR Blueprint architecture has been adopted by jurisdictions and vendors across Canada, saving time, effort and money, and helping to ensure systems are interoperable. As well, some jurisdictions have saved both time and money by acquiring vendor solutions together, rather than individually. Infoway is a strategic investor, providing a portion of system development costs and project oversight. Provincial and territorial (P/T) partners are responsible for the actual system development, implementation and overall funding, including ongoing operational costs. |
||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||
| Summary of Annual Plans of Recipient: In its Corporate Business Plan 2007-08, entitled Electronic Health Records: Transforming health care, improving lives, Canada Health Infoway indicated the following action plans, which will continue into 2008-09 and build upon Infoway's existing business strategies: 1. Participation in Healthcare Renewal: Infoway will continue to support national, jurisdictional and local initiatives to strengthen health care in Canada through a focus on improving patient access (wait times projects with P/Ts). Infoway will continue to participate in committees dealing with patient safety, heart health, cancer, mental health, primary health care, health indicators and reporting. 2. Collaboration with Partners: Infoway will continue to work with public and private sector partners to implement the pan-Canadian health infostructure. Infoway also continues to work with provinces and territories to help align their three-year plans with Infoway investments for EHRs, telehealth, public health surveillance and patient access (wait times) solutions. Infoway will continue to discuss the opportunities and challenges of EHR solutions with stakeholders, including health delivery organizations and clinician groups. 3. Targeting the Investments: As of December 31, 2007, Infoway had approved over 1.3 billion to support more than 240 projects across the country. Over the next one to two years, Infoway will focus on: continued work on EHR development and surveillance, updating of the telehealth program, implementation of the new patient access to quality care program (wait times), and adjustments to the gated funding model to help P/Ts meet their cash flow requirements for larger projects. Infoway intends to deliver 110-150 million in new project approvals by the end of March 2008. 4. Supporting Solution Deployment: Infoway will continue to provide expertise to support its partners as they successfully implement secure electronic health information solutions and will monitor projects to ensure cost and risk are well-managed. This work includes: standard development, EHR Blueprint, national solution agreements (such as pricing agreements), and collaboration with jurisdictions on risk and quality assessments and privacy. Infoway recently established a Privacy Forum where F/P/T Privacy Officers and representatives from F/P/T health departments discuss privacy governance matters as they relate to electronic health information management. 5. Promote Solution Adoption and Benefits Realization: Infoway will enhance its change management services, collaborate with P/Ts and health providers on knowledge dissemination to support clinician adoption and implement regional peer networks of physicians, nurses and pharmacists to champion and support EHR adoption and training strategies. This year Infoway will also communicate early results from Innovation projects launched over the past two years. |
||
| Planned Evaluations: Infoway will continue to coordinate jurisdictional benefits evaluation plans, and based on the framework and methodologies approved in 2007-08, will contract with both jurisdictional and research partners to conduct electronic health record benefits evaluations. The studies will measure outcomes related to quality, access and productivity. Baseline evaluations in several areas are underway, with early results being seen in the field of diagnostic imaging. During fiscal year 2008-09, Infoway will also have an external firm carry out an evaluation of its overall performance in achieving the outcomes identified in section 4.2 of the Addendum Agreement governing the pan-Canadian Health Surveillance System. | ||
| Planned Audits: An audit by Health Canada's Audit and Accountability Bureau, Chief Financial Officer Branch, began in December 2007. This audit has both an internal (Health Canada) and an external component, for which interviews with Canada Health Infoway officials are planned. The audit is intended to provide the Deputy Minister with
assurances that an appropriate Management Control Framework is in place within the Health Policy Branch and Canada Health Infoway Inc. (Infoway) for the monitoring and administration of Transfer Payment funds; and that Infoway is using funds in compliance with the terms and conditions of the funding agreements. Since the Bureau has indicated this audit is to be completed
before March 31, 2008, both Canada Health Infoway and Health Canada will respond to its recommendations in 2008-09. In addition, during fiscal year 2008-09, Infoway's regular annual financial and compliance audits will be carried out by independent audit firms. |
||
|
URL of Recipient Site: http://www.infoway-inforoute.ca/en/Home/home.aspx Canada Health Infoway |
||
| Canadian Health Services Research Foundation (CHSRF) | ||
| Start Date: 1996-97 | End Date: N/A | Total Funding: 151.5 million |
Description: Please note that CHSRF's programs also receive funding from other sources. Total federal funding for the CHSRF is as follows:
CHSRF's work contributes to Health Canada's aim of strengthening the knowledge base to address health and health care priorities. More specifically, CHSRF's programs further the development of health human resources, provide health managers with tools to improve primary and continuing care, and support nursing research from a health system perspective. |
||
| Strategic Outcome: Accessible and sustainable health system responsive to the health needs of Canadians | ||
Summary of Annual Plans of Recipient: The CHSRF will continue to pursue four strategic objectives:
Four strategic priorities, endorsed at the October 25-26, 2007 Board of Trustees meeting, will guide the CHSRF's core programming activities: Workforce and the work environment; Quality and safety; Change management; and Values-based decision-making. This is in addition to the commitments (both from a program and a financial perspective) that already exist and which
extend into the next three to four years. |
||
| Planned Evaluations: A CHSRF-commissioned independent evaluation of the NRF will be completed in 2008. The CHSRF conducts an annual evaluation of the EXTRA program. An extensive five-year review is planned for 2008. |
||
| Planned Audits: The CHSRF conducts an annual financial audit as well as an audit of its pension plan. Recommendations from a 2007 audit of funded projects will be implemented in 2008. The CHSRF will also receive recommendations from a Health Canada audit of the CHSRF, initiated in 2007-08. |
||
| URL of Recipient Site: http://chsrf.ca/ Canadian Health Services Research Foundation |
||
Green Procurement
| 1. | How is your department planning to meet the objectives of the Policy on Green Procurement? | ||
| Health Canada supports the objectives of the Green Procurement Policy, including incorporating environmental performance considerations and value for money into the procurement decision making process. Health Canada is an active participant in federal procurement reform initiatives, such as the establishment of mandatory standing offers by Public Works and Government Services Canada (PWGSC), which incorporate environmental performance and life cycle analysis. Health Canada has incorporated commitments in the Sustainable Development Strategy (SDS) 2007-10 in support of the Green Procurement Policy. | |||
| 2. | Has your department established green procurement targets? | ||
| Yes | No | In progress | |
| 3. | Describe the green procurement targets that have been set by your department and indicate the associated benefits anticipated. | ||
| Health Canada has established commitments in the SDS 2007-10 in support of the Green Procurement Policy. Health Canada is working to incorporate green procurement tracking procedures in its existing enterprise system through participation in horizontal SAP initiatives with other SAP departments. Once implemented, communications and awareness to users will facilitate reporting on green procurement. Knowledgeable functional experts raise awareness regarding sound procurement planning and green procurement opportunities with cost centre managers who are the contracting authorities. The department also promotes training of cost centre managers on green procurement by encouraging them to take the Canada School of Public Service (CSPS) on-line training, by broadcasting general awareness messages and by participating in the Environment Week in the National Capital Area. |
|||
Horizontal Initiatives
| Chemicals Management Plan | ||||||
|---|---|---|---|---|---|---|
| Lead departments: Health Canada and Environment Canada | Lead department program activity: Sustainable Environmental Health | |||||
| Start date: 2007-08 | End date: 2010-11 | |||||
| Total federal funding allocation: $299.2 million | ||||||
| Description of the horizontal initiative: The Chemicals Management Plan (CMP) is part of the Government's comprehensive environmental agenda and is managed jointly by Health Canada (HC) and Environment Canada (EC). The activities identified in this plan build on Canada's position as a global leader in the safe management of chemical substances
and products, and focus upon timely action on key threats to health and the environment. The CMP will also generate a higher level of responsibility for industry through realistic and enforceable measures, stimulate innovation, and augment Canadian competitiveness in an international market that is increasingly focussed on chemical and product safety. HC and EC will manage the CMP funding collectively and ensure that it is aligned with human health and environmental priorities. |
||||||
Shared outcomes: High-level outcomes for managing the CMP include:
|
||||||
| Governance structure: HC shares the lead on the CMP with EC. The CMP consists of five inter-related program elements (listed below) to be planned, delivered and evaluated within an integrated framework, managed jointly by Health Canada and Environment Canada. Within the federal government, governance is assured through a joint HC/EC Assistant Deputy Ministers Committee (ADM Committee) and the Interdepartmental Chemicals Management Executive Committee (CMEC). The ADM Committee was established to maximize the coordination of efforts, while minimizing duplication between the two departments. The mandate of CMEC is to ensure that all chemical management issues are optimally managed and that activities under the CMP are delivered in an integrated manner, using a suite of legislations, including the Canadian Environmental Protection Act, the Pest Control Products Act, the Food and Drugs Act and the Hazardous Products Act. Networks focusing on key CMP activities (Risk Assessment, Risk Management, Research/Science, Monitoring and Surveillance, and Policy and Program Management) will be established to support the above governance structures. |
||||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| Health Canada | Sustainable Environme-ntal Health Chemical Managem-ent (PAA Sub-Activity) |
a. Risk Assessment | 9.9 | 2.5 | Increase level of Canadian public awareness of chemical management issues and actions being taken |
|
| b. Risk Management | 50.1 | 12.4 | ||||
| c. Research | 26.6 | 5.9 | ||||
| d. Monitoring and Surveillance | 3.4 | 5.3 | ||||
| e. Program Management | 5.4 | 1.5 | Risk assessments are conducted and risk management objectives are met for regulations and other control instruments for substances and the products of biotechnology | |||
| Consumer Products | a. Risk Management | 12.6 | 2.4 | |||
| Pesticide Regulation | a. Risk Assessment | 9.9 | 2.5 | |||
| b. Risk Management | 13.6 | 3.3 | Declining trends in levels of risk, adverse reactions, illnesses and injuries from toxic chemical substances in the environment and their use and/or the risk of exposure to Canadians | |||
| Health Products | a. Risk Assessment | 3.3 | 0.8 | |||
| b. Risk Management | 12.5 | 2.4 | ||||
| c. Research | 2.5 | 0.4 | ||||
| d. Monitoring and Surveillance | 1.1 | 0.2 | ||||
| Food and Nutrition | a. Risk Assessment | 3.8 | 0.8 | |||
| b. Risk Management | 6.2 | 0.3 | ||||
| c. Research | 1.2 | 0.3 | ||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| Environment Canada | Risks to Canadians, their health and their environme-nt posed by toxic and other harmful substances are reduced | a. Research/ Science | 2.1 | 1.5 | Direction, collaboration and coordination of science and management activities Increased knowledge of the risks of toxic chemical substances through research and options to mitigate Risk assessments conducted in a timely fashion as required Risk management objectives are met, e.g. effective controls identified and implemented |
|
| b. Monitoring and Surveillance | 26.4 | 6.9 | Enhanced knowledge of stakeholders and the Canadian public regarding risk assessment/management activities, including knowledge on the effectiveness of control actions | |||
| c. Risk Assessment | 13.1 | 3.1 | Risks to Canadians' health and their environment posed by harmful chemicals are reduced | |||
| d. Risk Management | 64.9 | 16.1 | Canadians and their environment are protected from the harmful effects of chemicals | |||
| Sub-Total (Environment Canada) | 106.5 | 27.6 | ||||
| Sub-Total (Health Canada) | 192.7 | 42.0 | ||||
| Grand Total: | 299.2 | 69.6 | ||||
| Results to be achieved by non-federal partners: N/A | Contact information: Fran篩s Dignard Strategic Science and Operations, Health Canada (613) 941-0590, francois_dignard@hc-sc.gc.ca |
|||||
| Federal Tobacco Control Strategy (FTCS) | ||||||
| Lead department: Health Canada | Program activity: Substance use and abuse | |||||
| Start date: April 1, 2007 | End date: March 31, 2011 | |||||
| Total federal funding: 368.5 million | ||||||
| Description of the horizontal initiative: The FTCS establishes a framework for a comprehensive, fully-integrated, and multi-faceted approach to tobacco control. It is driven by the longstanding commitment of the Government of Canada to reduce the serious and adverse health effects of tobacco for Canadians. It focuses on four mutually reinforcing components: prevention, cessation, protection, and product regulation. | ||||||
|
Shared outcome: The long-term outcome of the FTCS will remain the same for the next four years: to reduce tobacco-related disease and death in Canada. To pursue the long-term outcome, the FTCS will contribute the following tobacco control goals and objectives for April 1, 2007 to March 31, 2011. Goal: Reduce overall smoking prevalence from 19 (2005) to 12 percent by 2011. Objectives:
|
||||||
|
Governance structures: Resources for the implementation of the FTCS were allocated to a number of departments and agencies. HC is the lead department in the FTCS and is responsible for regulating the manufacture, sale, labelling, and promotion of tobacco products as well as developing, implementing and promoting initiatives that reduce or prevent the negative health impacts associated with smoking. The partner departments and agencies are:
|
||||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation ($ M) |
Planned Spending for 2008-09 ($ M) | Expected Results for 2008-09 | |
| Health Canada (HC) | Substance use and abuse | FTCS | 287.34 | 57.46 | The FTCS has set a 4-year goal to: Reduce overall smoking prevalence from 19% (2005) to 12% by 2011. All activities are expected to contribute towards achieving this result. Details on 2008-09 HC key activities in support of this result are detailed below. | |
| Public Safety Canada (PSC) | N/A | FTCS | 3.05 | 0.61 | Expected results for 2008-09 will be reported through PSC's departmental Report on Plans and Priorities. | |
| Royal Canadian Mounted Police (RCMP) | N/A | FTCS | 8.62 | 1.72 | Expected results for 2008-09 will be reported through the RCMP's departmental Report on Plans and Priorities. | |
| Office of the Director of Public Prosecutions (ODPP) | N/A | FTCS |
12.22 | 2.44 | Expected results for 2008-09 will be reported through the ODPP's Report on Plans and Priorities. | |
| Canada Revenue Agency (CRA) | N/A | FTCS | 4.44 | 0.89 | Expected results for 2008-09 will be reported through the CRA's departmental Report on Plans and Priorities. | |
| Canada Border Services Agency (CBSA) | N/A | FTCS | 52.8 | 10.56 | Expected results for 2008-09 will be reported through the CBSA's departmental Report on Plans and Priorities. | |
| Total: | 368.47 | 73.68 | ||||
| Results to be achieved by non-federal partners: N/A | Contact information: Cathy A. Sabiston Director General, Tobacco Control Program Health Canada (613) 941-1977 |
|||||
| Early Childhood Development and Early Learning and Child Care | ||||||
| Lead Departments: Health Canada (HC) Indian and Northern Affairs Canada (INAC) Human Resources and Social Development Canada (HRSDC) Public Health Agency of Canada (PHAC) |
Program activity: First Nations and Inuit Health Programming and Services Federal Early Childhood Development (ECD) Strategy for First Nations and Other Aboriginal Children Additional funding to ECD Programs from: Enhancing Early Learning and Child Care (ELCC) for First Nations Children Living on Reserve and Working Towards the First Phase of a Single Window |
|||||
| Start date: ECD - October 2002 ELCC - December 2004 |
End date: ECD Strategy - ongoing ELCC Single Window - ongoing; |
|||||
| Total federal funding: As a result of an ECD Strategy announced in October 2002, 320 million over five years (and ongoing) is dedicated to enhancing various federal ECD programs. In December 2004, Cabinet approved an additional 45 million over three years (14 million ongoing) to improve integration and coordination of two federal ECD/ELCC programs (Aboriginal Head Start On Reserve and the First Nations and Inuit Child Care Initiative). | ||||||
| Description of the horizontal initiative (including funding agreement): The ECD Strategy for First Nations and Other Aboriginal Children was announced on October 31, 2002. The strategy provides $320 million over five years to: improve and expand existing ECD programs and services for Aboriginal children; expand ECD capacity and networks; introduce new research initiatives to improve understanding of how Aboriginal children are doing; and work towards the development of a "single window" approach to ensure better integration and coordination of federal Aboriginal ECD programming. In December 2004, as first phase of a "single window", Cabinet approved an additional 45 million over three years (14 million ongoing) to improve integration and coordination of two ECD programs, the Aboriginal Head Start on Reserve and the First Nations and Inuit Child Care Initiative, beginning in 2005-06. The objectives of these funds are to increase access to and improve the quality of ELCC programming for First Nations children on reserve, and improve integration and coordination between the two programs through joint planning, joint training and co-location. Joint planning will also include INAC-funded child/day care programs in Alberta and Ontario. | ||||||
| Shared Outcome: The federal ECD Strategy complements the September 2000 First Ministers federal, provincial and territorial (F/P/T) ECD Agreement. It seeks to address the gap in life chances between Aboriginal and non-Aboriginal children by improving the developmental opportunities to which Aboriginal children (and their families) are exposed
at an early age (0 to 6 years). The funding approved in December 2004 for ELCC for First Nations Children Living on Reserve and Working Towards the First Phase of a Single Window complements funding released to provinces and territories under the March 2003 Multilateral Framework for Early Learning and Childcare (ELCC) to improve access to ELCC programs and services. |
||||||
| Governance Structure: Interdepartmental ECD ADM Steering Committee; Interdepartmental ECD Working Group. | ||||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation ($ M) |
Planned Spending for 2008-09 ($ M) | Expected Results for 2008-09 | |
| HC Electronic Link: http://www.hc-sc.gc.ca/fnihb-dgspni/fnihb/cp/ahsor/index.htm |
First Nations and Inuit Health Programming and Services | Aboriginal Head Start on Reserve (AHSOR) | 107.595 (total for 2002-03 through to 2006-07; 21.519/ year) committed in 2002 24.000 (total for 2005-06 through to 2007-08; 7.500 in 2005-06, 8.300 in 2006-07, 8.200 in 2007-08 ) committed in 2005 |
21.519 (and ongoing) 7.500 (and ongoing) |
Program expansion and enhancement Increase integration, coordination, access and quality |
|
| Electronic Link: http://www.hc-sc.gc.ca/fnih-spni/famil/preg-gros/intro_e.html |
First Nations and Inuit Health Programmi-ng and Services | Fetal Alcohol Spectrum Disorder - First Nations and Inuit Component (FASD-FNIC) |
70,000 (total for 2002-03 through to 2006-07; 10,000 in 2002-03 and 15.000 thereafter) committed in 2002 |
15.000 (and ongoing) |
Program expansion and enhancement | |
| First Nations and Inuit Health Programmi-ng and Services | Capacity Building | 5.075 (total for 2002-03 through to 2006-07; 1.015/year) committed in 2002 | 1.015(and ongoing) | Increased capacity | ||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| PHAC Electronic Link: http://www.phac-aspc.gc.ca/dca-dea/programs-mes/ahs_main_e.html |
Child and Adolescent Health Promotion | Aboriginal Head Start in Urban and Northern Communities (AHSUNC) | 62.880 (total for 2002-03 through to 2006-07; 12.576/ year) committed in 2002 |
12.576 (and ongoing) |
Program expansion and enhancement by increasing number of special needs and parental outreach workers; enhance special needs training; serve another 1,000 children | |
| Child and Adolescent Health Promotion | Capacity Building | 2.500 (total for 2002-03 through to 2006-07; 0.500/year) committed in 2002 |
0.500 (and ongoing) |
Increased capacity |
||
| HRSDC | Lifelong Learning- Health Human Resources (HHR) | First Nations and Inuit Child Care Initiative (FNICCI) | 45.700 (total for 2002-03 through to 2006-07; 9.140/year) committed in 2002 21.000 (total for 2005-06 through to 2007-08; 7.000/year) committed in 2005 |
9.140 (and ongoing) 6.500 (and ongoing) |
Program expansion and enhancement Increase program integration, coordination, access and quality |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| Lifelong Learning- HHR |
Research and Knowledge | 21.200 (total for 2002-03 through to 2006-07; 4.240/year) committed in 2002 |
4.240 (and ongoing) | Information on the well-being of Aboriginal children through an Aboriginal Children's Survey (ACS) and the Aboriginal component of "Understanding the Early Years" | ||
| INAC | Lifelong Learning- Early Learning and Childcare | Capacity Building | 5.050 (total for 2002-03 through to 2006-07; 1.010/year)as committed in 2002 | 1.010 (and ongoing) - committed in 2002 | Increased capacity and development of "single window" | |
| Total ECD: 320.000 (60.000 in 2002-03 and 65.000 thereafter) |
Total ECD: 65.000 |
|
||||
| Total ELCC: 45.000 (14.500 in 2005-06; 15.300 in 2006-07; 15.200 in 2007-08). |
Total ELCC: 14.000 |
|||||
| Results to be achieved by Non-Federal Partners: N/A | Contact: Marcia Armstrong, Program Officer, ECD Strategy Unit, First Nations and Inuit Health Branch, Health Canada Postal Locator 1920D Tunney's Pasture, Ottawa Telephone: (613) 946-4621 Fax: (613) 952-5244 |
|||||
| Building Public Confidence in Pesticide Regulation and Improving Access to Pest Management Products | ||||||
| Lead department: Health Canada | Program Activity: Pesticide Regulation | |||||
| Start Date: 2002-03 | End Date: 2008-09 | |||||
| Total Funding Allocated:155 million | ||||||
|
Description: This initiative incorporates efforts of six federal government partners to increase public and stakeholder confidence in the pesticide regulatory system, to protect health and environment, and to increase the competitiveness of the agri-food and forestry sectors. Research and monitoring in the area of pesticides is being coordinated with their
regulation. Under this initiative, the presence and effects of pesticides in the environment, in marine and freshwater ecosystems, and in the forest environment are being monitored. The initiative enhances monitoring and enforcement of pesticide residue limits in foods, in feed, of pesticide residues in fertilizers, and pesticide guarantee verification for fertilizer-pesticide combinations. Reduced-risk pesticides and biological pesticides for forestry are being developed and their use facilitated. Commodity-based risk reduction strategies for the agriculture and agri-food sector are being developed and implemented. Programs improving access to agricultural minor-use pesticides and reduced-risk pesticides for agricultural use are being established. Research to support the introduction of minor-use pesticides that pose a reduced risk to the environment is being conducted. A reporting system to track adverse effects of pesticides has been developed, and information on these effects will be collected and recorded. Collectively, this work is being conducted to achieve public confidence in increased conservation and protection of human health and the environment while contributing to the competitiveness of Canada's agricultural sector. The information presented in this table has been organized along the following three main themes of this initiative:
|
||||||
Shared Outcomes:Immediate Outcomes:
Final Outcome:
|
||||||
Governance Structures:
|
||||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| I. Research and Monitoring | ||||||
| AAFC | Building Public Confidence in Pesticide Regulation and Improving Access to Pest Management Products (BPC) | (a) Conducting research to support the introduction of minor-use pesticides that pose a reduced risk to the environment. | 8.0 | 0.0 |
Fiscal year 2008-09 is a transition year from APF to Growing Forward policy. Discussions on how the programs will evolve will take place during the course of the year at which time associated future funding decision will be made. Although no specific funding allocation has been made at this time, the following are the expected results:
|
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| CFIA | BPC | (b) Enhanced monitoring and enforcement of pesticide residue limits in food and feed. | 2.7 | 0.25 |
|
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| CFIA | BPC | (c) Enhanced monitoring and enforcement of pesticide residues in fertilizers and pesticide guarantee verification in fertilizer-pesticide combinations. | 2.4 | 0.25 | Develop monitoring and surveillance policies and programs and advise inspectors on appropriate follow-up Increase interaction with the PMRA to obtain the most up-to-date pesticide safety and labelling information Update the Compendium of Fertilizer-Use Pesticides, which contains information regarding registration, guarantees and proper labelling Work to develop regulatory changes to facilitate updating the Compendium more regularly, and, if successful, provide Compendium updates more regularly to the producers of mixtures and to the CFIA's inspection staff Sample fertilizer-pesticide combinations to verify guarantees Sample fertilizers suspected to be contaminated with pesticides Verify fertilizer-pesticide labels Conduct investigation and compliance activities (anticipated based on sampling and inspection frequencies) Analyse samples submitted by inspectors and develop new methods, as required |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| DFO | BPC | (d) Monitor and research the presence and effects of pesticides in marine and freshwater ecosystems. | 7.9 | 1.0 | DFO will provide the PMRA with final reports on regional National Fund projects. These research projects will be focused to address key research knowledge gaps, as they were in 2007-08, after consultation with PMRA. Current DFO research focuses on the potential impacts of chlorpyrifos, chlorothalonyl and glyphosate to affect growth, reproduction, olfaction and immunocompetence in freshwater fish and salmon as well as growth and survival of model invertebrates. DFO will provide the PMRA with a yearly report from DFO's Centre for Environmental Research on Pesticides (CERP). CERP will conduct laboratory and field based studies to quantify impacts of exposure to the priority pesticides on fish and fish habitat. Impacts will be quantified in terms of reproductive success, growth and energy metabolism. Priority research will be identified in consultation with PMRA. After consultation with the PMRA, DFO will design and initiate new research projects related to the theme "Potential Impacts of Pesticides on Fisheries Resources". |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| DFO cont'd |
DFO will contribute to the Formative Evaluation of the Building Public Confidence in Pesticide Regulation and Improving Access to Pest Management Products Horizontal Initiative. | |||||
| EC | BPC | (e) Monitor and research on presence and effects of pesticides in the environment. | 7.61 | 1.0 |
EC's commitment to the BCP will continue for 2008-09. Based on Cycle 1 results and the preliminary results in the first year of Cycle 2, EC will set out to deliver on a schedule of research and monitoring projects on the presence and impacts of pesticide in the environment. The Pesticide Program Coordinating Committee (which has PMRA membership) will continue to assess
priorities for pesticide science at EC. Specifically, EC will:
|
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| EC cont'd |
|
|||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| EC cont'd |
|
|||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| EC cont'd |
|
|||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| EC cont'd |
Terrestrial effects: There will be a concerted effort to focus the current terrestrial deliverables for birds and mammals on Species at Risk Act (SARA) -listed species and other species of concern, as well as on areas of perennial conflict between wildlife and pesticide use such as the lower BC Mainland. Priority will be given to pesticides causing acute intoxications (e.g. cholinesterase-inhibiting insecticides) or broad contamination of terrestrial food chains (second generation anticoagulant rodenticides). Efforts will be focused also on comparing dietary and non-dietary (dermal, preening and inhalation) routes of exposure to pesticides in order to improve our ability to assess new pesticides and better predict impacts. For terrestrial life-stages of amphibians, the effort will be to continue work on an amphibian test protocol. For plants, the efforts will focus on improvement of routine phytotoxicity tests in greenhouses and on risk assessment with validation through in-situ research. | |||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| EC cont'd |
EC will also continue to work with the PMRA in the implementation of the existing EC/PMRA Memorandum of Understanding (MOU) in order to better integrate and coordinate research with regulation. The MOU has four components, Science Policy, Knowledge Generation, Issue Management and Compliance Promotion and Enforcement. EC will continue working on providing leadership in the development and implementation of a federal, coordinated pesticides science strategy for research and monitoring through the Interdepartmental Committee. Furthermore, EC will participate in the proposed 6NR MOU. EC will continue to contribute to PMRA's pesticide assessments where appropriate, will coordinate with PMRA on the development of environmental quality guidelines and will continue to provide science/policy advice on key Government of Canada policies as they relate to pesticide management and use in Canada. |
|||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| HC (PMRA) | Pesticide Regulation | (f) Linking pesticide regulation and research. | 4.2 | 0.8 | Identify and communicate annually PMRA's research and monitoring priorities to 6NR partners through regular meetings and other avenues, as needed. Continue facilitating discussions to identify linkages between 6NR research results and their use in regulatory decision making by PMRA, as well as opportunities for collaborative research among the 6NR. Facilitate the two-way communication and coordination between regulation and research between governments within Canada (through PMRA's F/P/T Committee) and internationally as well as with the private and academic sectors, through presentations linking research and regulation at regional, national and international meetings (e.g., through Society of Environmental Toxicology and Chemistry (SETAC), Canadian Standards Association (CSA), International Union of Pure and Applied Chemistry (IUPAC). Improve risk assessment procedures particularly in the area of environmental fate prediction (e.g., water modelling and exposure assessment). Strengthen PMRA's capacity to implement probabilistic risk assessments where appropriate. |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| HC(PMRA) | BPC | (g) Conducting research to support the introduction of minor-use pesticides that pose a reduced risk to the environment. | 3.5 | 0.0 |
Fiscal year 2008-09 is a transition year from APF to Growing Forward policy. Discussions on how the programs will evolve will take place during the course of the year at which time associated future funding decisions will be made. Advance the risk assessment methodologies through:
Update the database on environmental toxicology and fate to guide decisions, internally and externally, on comparative risk and reduced risk products. |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| HC (PMRA) cont'd |
g) Conducting research to support the introduction of minor-use pesticides that pose a reduced risk to the environment. |
Health Evaluation Directorate
|
||||
| NRCan | BPC | (h) Research and monitor pesticides in the forest environment. | 3.5 |
0.5 | Coordination of the Enhanced Pest Management Program (EPM). Review of final reports of two projects funded for one year only. Review of proposals, and fund selected projects including: Environmental impact assessment of alternative pest control options, Modeling wildlife exposure and risk relative to systemic insecticides, and Updating the Edaphic Indicator Research Tool. Sponsor and provision of input into the 2008 National Forest Pest Management Forum. Continuation, integration and coordination of NRCan work with the activities of the PMRA and other members of the 6NR Working Group on Pesticides. Provision of input in the 2008 Summative Evaluation. |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| II. Developing and Implementing Commodity Specific Risk Reduction Strategies | ||||||
| AAFC | BPC | (a) Commodity based risk reduction strategies. | 19.3 | 2.5 | Fiscal year 2008-09 is a transition year from APF to Growing Forward policy. Discussions on how the programs will evolve will take place during the course of the year at which time associated future funding decision will be made. Pesticide risks to health and the environment assessed for grower identified priority pest management issues and priority risk issues selected for support in coordination with PMRA Development and implementation of up to 4 risk reduction strategies based on pesticide risk priorities Stakeholders engaged in pesticide risk solution development for priority pesticide risk issues Engage stakeholders to develop a review and updating program for published crop profiles and update up to 6 previously published profiles Collection of data for up to 6 crops through regional focus groups for the purpose of updating profiles and tracking success of the program Continued funding for ongoing research and implementation projects |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| AAFC cont'd |
Request proposals throughout the fiscal year to address solutions developed through pesticide risk reduction strategies, with up to 10 new projects supported Communicate results of completed projects Continued joint communications with PMRA, including the publication of cooperative pesticide risk reduction strategies Regulatory support to biopesticide companies for the submission of up to 5 biopesticides for registration |
|||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| HC (PMRA) | BPC | (a) Commodity based risk reduction strategies (RR). | 25.7 | 4.0 | Planned staffing actions in 2008-09, indeterminate positions Ongoing consultations with stakeholders to assist PMRA in determining priority regulatory needs. Work share with other government departments and 6NR. Work on pesticide risk indicator: consult, build and validate database Together with AAFC and other Directorates at PMRA, assess the risk reduction potential of stakeholder identified pest management priorities. Workshare with AAFC on new crop profiles and issue documents and finalising existing documents. Work with AAFC to define areas where cross commodity work can be accomplished. Risk reduction strategies will be developed for issues that have a demonstrated potential to reduce pesticide risk. Where possible, strategies will be developed based on common themes rather than be restricted to single pest-crop combinations. Stakeholder consultations will be held throughout the year as required. |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| HC (PMRA) cont'd |
A number of strategies will be initiated this year to transition to lower risk products where key pesticide uses are being lost through the re-evaluation program. This will include strategies to address the loss of azinphos methyl, terbufos and phorate in a number of sectors. National consultations will be held to inform stakeholders of these transition strategies and
begin development of the strategies. In addition to work on commodity based risk reduction strategies, PMRA is working with stakeholders to develop strategies to address issues in a number of nonagricultural sectors, including forestry, the heavy duty wood preservatives industry, ornamental and landscaping, structural pest control, food processing, storage pest control and honey production. |
|||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| III. Generation of Data to Support the Registration of Reduced-Risk and Minor-Use Pesticides for the Agricultural and Agri-Food Sector and Reduced-Risk Pesticides and Biopesticides for Forestry | ||||||
| AAFC | BPC | (a) Improving access to agricultural minor-use pesticides, and reduced-risk pesticides for agricultural use. | 33.7 12.0M (A-base) |
0.0 2.0 (A-base) |
Fiscal year 2008-09 is a transition year from Agricultural Policy Framework (APF) to Growing Forward policy. Discussions on how the programs will evolve will take place during the course of the year at which time associated future funding decision will be made. AAFC will hold a national minor use priority setting workshop with stakeholders to select 36 Minor Use research priorities. AAFC will select up to an additional 20 joint AAFC/Inter-regional 4 (IR-4) research priorities for the 2009 research season. AAFC will consult with and solicit written support from the pesticide manufactures whose pesticides are chosen for these crop-pest research priorities. AAFC will complete and forward presubmission consultation requests to PMRA. AAFC will conduct or contract out research trials to generate the required data. Data generation will take place during the 2009 growing season. |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| AAFC cont'd |
Analysis of data from previous years research will occur throughout the year followed by the writing of final reports and submissions to PMRA. AAFC is targeting the completion of 40 Minor Use (MU) submissions during the year. AAFC will participate in the NAFTA Technical Working Group on Pesticides. AAFC will meet with IR-4 and Industry representatives to promote Joint Reviews to the PMRA and Environmental Protection Agency (EPA) and promote NAFTA submissions. |
|||||
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| HC (PMRA) | BPC | (a) Improving access to agricultural minor-use pesticides, and reduced-risk pesticides for agricultural use. | 20.8 | 0.0 | Fiscal year 2008-09 is a transition year from APF to Growing Forward policy. Discussions on how the programs will evolve will take place during the course of the year at which time associated future funding decisions will be made. Product evaluation work, such as the review of presubmission proposals from AAFC and provincial coordinators and issue data requirements Register new minor crop uses, including minor use and reduced-risk products and uses Harmonization work and regulatory projects Joint Reviews in collaboration with the U.S. EPA, AAFC and U.S. Department of Agriculture IR-4 Program, further work on crop groupings and on Maximum Residue Levels (MRL) promulgation Complete 10 AAFC/IR-4 joint minor use reviews (PMRA/EPA) Increase communication and provide feedback to AAFC to improve the quality and use of scientific rationales |
|
| Federal Partners | Federal Partner Program Activity (PA) | Names of Programs for Federal Partners | Total Allocation (millions) |
Planned Spending for 2008-09 (millions) | Expected Results for 2008-09 | |
| NRCan | BPC | (b) Develop and facilitate the use of reduced-risk pesticides and biological pesticides for forestry. | 4.1 | 0.5 |
If first year progress reports are satisfactory, fund the following three Enhanced Pest Management Method (EPMM) proposals for a second year 2008-09:
Coordination and facilitation of registration of minor use pesticides for uses in woody ornamental, urban forest, and woodlands. Liaison with the PMRA and other government departments on pest management issues Input into Implementation of the 6NR Work Plan on Pesticides |
|
| Total | 154.96 million | |||||
| Results to be achieved by non-federal Partners: N/A | Contact Information: Richard Aucoin Chief Registrar and Director General Pest Management Regulatory Agency 2720 Riverside Drive Ottawa, Ont K1A 0K9 |
|||||
Internal Audits
| Name of Internal Audit | Audit Type | Status | Expected Completion date | Electronic Link to Report |
|---|---|---|---|---|
| Dental Benefits | Program | In progress | 2008-09 | N/A |
| Health Protection - Communicable Disease | Program | Planned | 2008-09 | N/A |
| Tobacco Control - Research | Program | Planned | 2008-09 | N/A |
| Healthy Environments and Consumer Safety Branch - Quality Assurance Process for Recipient Audits | Transfer Payment | In progress | 2008-09 | N/A |
| Biologic and Genetic | Program | In progress | 2008-09 | N/A |
| Electronic Health Information System (Infoway) | Transfer Payment | In progress | 2008-09 | N/A |
| Canadian Health Services Research Foundation (CHSRF) | Transfer Payment | In progress | 2008-09 | N/A |
| Respendable Revenues | Risk-Based Controls | In progress | 2008-09 | N/A |
| Risk-Based Controls-Issuance of contracts to former public servants (Health Products and Food Branch) | Program | In progress | 2008-09 | N/A |
| Data Integrity - Feeder systems to SAP - Phase II (consists of 4 separate projects) | Support Function | In progress | 2008-09 | N/A |
| Audit of HR Strategies for Recruitment and Retention | Support Function | In progress | 2008-09 | N/A |
| Risk-Based Controls - Leave | Risk-Based Controls | In progress | 2008-09 | N/A |
| Risk Management | Initiative | In progress | 2008-09 | N/A |
| Governance and Accountability | Initiative | In progress | 2008-09 | N/A |
| Values and Ethics | Initiative | In progress | 2008-09 | N/A |
| Electronic Link to Internal Audit plan: N/A | ||||
Progress Toward the Department's Regulatory Plan
| Regulators | Expected Results |
|---|---|
| Program Activity: Canadian Assisted Human Reproduction | |
| Regulations under the Assisted Human Reproduction Act (Subsection 14(2)b), Counselling) |
The Assisted Human Reproduction Act requires that counselling services are made available to the donor before donating human reproductive material or in vitro embryos. The regulations will provide the details respecting the counselling services. |
| Regulations under the Assisted Human Reproduction Act (Section 40 to Section 43, Licensing) |
The Assisted Human Reproduction (AHR) Act requires that all persons undertaking controlled activities (i.e., assisted human reproduction procedure such as in vitro fertilization) must obtain a license to undertake those activities and undertake those activities in a licensed premises. The AHR regulations will detail the requirements for obtaining a license such as the administrative framework for the activities and the premises, and qualifications. |
| Regulations under the Assisted Human Reproduction Act (Section 10, Clinical and Laboratory Activities) |
Section 10 of the Assisted Human Reproduction Act specifies that controlled activities must be performed in accordance with a license and regulations. The regulations, with respect to controlled activities, will define the conduct of core activities involved in the practice of assisted human reproduction (e.g., in vitro fertilization, embryo transfer). These regulations will address the protection and promotion of human health, safety, dignity and rights related to the conduct of the controlled activities. |
| Regulations under the Assisted Human Reproduction Act (Section 14 to Section 16, Health Reporting Information for Controlled Activities) |
These regulations will specify the health reporting information that Assisted Human Reproduction (AHR) clinics will need to collect prior to each AHR procedure. These regulations will also address retention, use, disclosure and destruction of health reporting information. |
| Regulations under the Assisted Human Reproduction Act (Section 12, Reimbursement of Expenditures) |
Section 12 of the Assisted Human Reproduction (AHR) Act allows for the reimbursement of expenditures to donors of human reproductive material (i.e., semen, oocytes) and surrogate mothers. These AHR regulations will specify the reasonable expenditures that can be reimbursed. |
| Program Activity: Food and Nutrition | |
| Food and Drug Regulations (Addition of Vitamins and Minerals to Foods) |
Facilitation of greater consumer choice and industry innovation by revision of regulations on the addition of vitamins and mineral nutrients to foods taking into account the role of nutrient addition to foods, consumer needs and expectations, and industry requests. |
| Food and Drug Regulations (Enhanced Labelling) |
The proposed amendment will enhance protection of allergic consumers through mandatory labelling, on the labels of pre-packaged food products, of specific food allergens, gluten sources and sulphites when present at 10 parts per million or more, whether they have been added directly or indirectly. |
| Food and Drug Regulations (Mandatory Labelling of Raw Ground Meat and Ground Poultry) |
The proposed regulation will reduce the risk of food-borne illness as a result of providing safe handling information on the labels of these products which, due to their raw state, can introduce disease-causing bacteria to the food preparation environment. |
| Program Activity: Health Products | |
| Food and Drug Regulations (Health Claims) |
The proposed amendment allows the addition of two diet-related health claims to the list of claims manufacturers can use to promote healthy foods regarding: fruits, vegetables and whole grains and reduced risk of heart disease; and folic acid and reduced risk of neural tube defects. |
| Food and Drug Regulations (Progressive Licensing Framework) |
A new regulatory framework based on sound science and risk management is being developed, including revised requirements for initial licensing of new products, provisions allowing for conditions of licensing (e.g., post-market commitments), regulations for the content and revisions of product monographs, provisions for continual evaluation and re-evaluation of licenses, new enforcement and compliance tools, regulations for the issuance of risk communication tools, and provisions for public involvement in the regulatory process. |
| Food and Drug Regulations (Regulations amending Division 1 requirements for Non-Medicinal Ingredients in Drug Product Formulation) |
The proposed regulatory amendment is to require the submission of complete qualitative and quantitative formulation data, including a list of all non-medicinal ingredients (NMIs) in a drug product, as well as the source of any human or animal derived NMIs or medicinal ingredients in products currently regulated under Division 1 of the Regulations. This information would also be required to be submitted whenever there are updates or changes to the formulation. |
| Food and Drug Regulations (Non-Medicinal Ingredient Labelling) |
The proposed regulatory amendment to Division 1 is to require the labelling of non-medicinal ingredients on the labels of non-prescription drugs for human use (with some exceptions). This may result in a positive impact on consumer health and safety, by allowing individuals to avoid life-threatening allergens and agents for which they have a history of adverse reactions. Furthermore, this would enable consumers to make informed choices when purchasing non-prescription medications. |
| Medical Devices Regulations (Investigational Testing) | The proposed amendment to the Medical Devices Regulations will further address risks to health associated with investigational testing by requiring conformance to Good Clinical Practices, reporting to Health Canada for reviewing/monitoring, and introducing an inspection program. |
| Medical Devices Regulations (Amendment to Schedule 2) |
With the addition of breast implants to Schedule 2, manufacturers of breast implants will be required to comply with the Implant Registration requirements. This will enable manufacturers of breast implants to notify implant recipients of any new information concerning the safety, effectiveness or performance of the breast implant, and any corrective action that is required to address any problems associated with the breast implant. |
| Natural Health Products Regulations/Medical Devices Regulations / Food and Drug Regulations (Schedule A Revision and Claims) |
The proposed amendment allows industry to label and advertise Schedule A preventative claims approved by Health Canada for non-prescription drugs and Natural Health Products, and the Modernization of Schedule A list of disorders and diseases. |
| Food and Drug Regulations (Clinical Trial Registration) |
Registration and disclosure of clinical trial information, including results, would provide the public with access to information about ongoing, completed and published clinical trials. Public access to such information would further good research practices, assist in making treatment decisions, and help increase public trust in clinical research. |
| Fees in Respect of Medical Devices Regulations/Authority to Sell Drugs Fees Regulations/ Drug Evaluation Fees Regulations/ Establishment Licensing Fees Regulations (Cost Recovery Initiative) |
The Health Products and Food Branch (HPFB) Cost Recovery Initiative (CRI) was established to analyze and improve upon the current cost-recovery regime that covers the regulation, licensing, compliance, enforcement, and post market surveillance of health products by providing a long-term stable funding source for HPFB. The current focus of the CRI is on human drugs and medical devices. Four (4) sets of current fee regulations will be revised in 2008-09 to reflect the revised fees as developed by the CRI. |
| Food and Drug Regulations (Regulations amending the Special Access Program) |
The regulations provide for emergency access to drugs that do not have a Canadian market authorization on a patient-by-patient basis. Amendments to the regulations will allow for the block release of such drugs to a population and/or for drug stockpiling to address public health emergencies. |
| Food and Drug Regulations (Revisions to Division 12 - Pre-packaged Water and Ice) |
The proposed amendment provides for revision and updating of the safety and labeling requirements for prepackaged water and ice products. |
| Food and Drug Regulations (Amendments to Division 3 with respect to Positron Emitting Radiopharmaceuticals) |
Amendment of Division 3, Part C of the Food and Drug Regulations with respect to the use of positron-emitting radiopharmaceuticals (PERs) in basic clinical research, in recognition that the application of the current regulations for clinical trials under Division 5, Part C of the FDR to PERs basic clinical research studies is placing an undue regulatory burden on the researchers in this field and may be impeding basic clinical research involving PERs in Canada. |
| Food and Drug Regulations (Safety of Blood and Blood Components Regulations) |
Replacement of current requirements governing human blood and blood components in the Food and Drug Regulations with new comprehensive safety and quality of blood and blood components regulations. The objectives of the new regulations include outlining clear and intelligible requirements; allowing for timely updating of the requirements as new technologies/products/issues emerge; and achieving greater harmonization in Canada related to the collection, handling and post-approval surveillance of blood and blood components. |
| Food and Drug Regulations (Amendment to C.01.016 to require notification of results of annual summary reports) |
Manufacturers will be required to inform the Minister of significant safety issues arising from their annual assessment of Adverse Drug Reaction Reports. |
| Amendment to Division 8 of the Food and Drug Regulations and Guideline to fully implement International Conference on Harmonization (ICH) E2C Guideline on Drug Safety Reporting i.e. Periodic Safety Update Reporting | Canadian requirements for periodic reporting of drug safety will be harmonized with other leading international regulators (US Food and Drug Administration (FDA), European Agency for the Evaluation of Medicinal Products (EMEA), Japan, etc.) |
| Amendments to the Natural Health Products Regulations, the Food and Drug Regulations and the Establishment Licensing Fees Regulations | The proposed dual licensing regulatory amendment will alleviate the exporting challenges arising from the coming into force of the Natural Health Products Regulations (NHPR). It will allow, on a voluntary basis, natural health product companies to hold an establishment license pursuant to the Food and Drug Regulations, in addition to the required site license under the NHPR. |
| Natural Health Products Regulations (Natural Health Products Regulatory Review) |
The Natural Health Products Directorate launched a review of the Natural Health Products Regulations (NHPR) in consideration of the challenges and issues identified in the first three years of regulating natural health products. Potential amendments which have been identified for the NHPR relate to the development of a new risk-based approach for product and site licensing, surveillance and compliance and enforcement activities. |
| Food and Drug Regulations (Extraordinary Use of New Drug Regulations) |
Under the Food and Drug Regulations, a drug is approved only if substantial evidence of its efficacy is available. Amendment of the Food and Drug Regulations will allow the approval of certain drugs the efficacy of which cannot be tested on humans in clinical trials for ethical reasons. These drugs may be required in emergency situations such as the treatment of pandemic influenza virus. The availability of these drugs is necessary to meet emergency preparedness measures which require the stockpiling of some of these drugs. |
| Food and Drug Regulations (Amendments to Division 15 Maximum Residue Limits for Veterinary Drugs) |
New or revised Maximum Residue Limits for veterinary drugs in foods to ensure the safety and quality of food products derived from treated food-producing animals. |
| Food and Drug Regulations (Prohibition of Importation for Personal Use) |
This proposed amendment will further restrict the importation of drugs to include the personal importation of drugs intended to be used in food-producing animals to avoid potentially harmful residues in food products from animals treated with these drugs. These regulatory amendments will better address the health risks to human and animal health and the safety of Canada's food supply associated with the use of unauthorized veterinary drugs. |
| Program Activity: Substance Use and Abuse | |
| Marihuana Medical Access Regulations (Phase III amendments) | These amendments will address a series of issues raised by both external stakeholders and those involved in administering the Marihuana Medical Access Program, e.g., the proposed modifications would strengthen the tools available for use in ensuring compliance. |
| Promotion of Tobacco Products and Accessories Regulations (Display and Signs at Retail) (Preparation of Proposed Regulations to Restrict the Display of Tobacco Products, Accessories and Signs at Retail) |
The proposed regulations are to reduce visibility of tobacco promotion at retail. Achievements will be measured through surveys at retail. |
| Tobacco Products Information Regulations (Preparation of Amendments to the Tobacco Products Information Regulations) |
The amendment is to provide clarification and update of the applicable requirements of the regulations, correction of minor grammatical errors, redundancies and ambiguities and discrepancies between the English and French version and to remove the obligation to list numerical values for toxic emissions. |
| Promotion of Tobacco Products and Accessories Regulations (Prohibited Terms) |
The proposed regulations are to prohibit the promotion of tobacco products and accessories which display the terms "light" or "mild". The purpose is to reduce confusion among smokers regarding these descriptors. Greater awareness that no class of cigarettes is a "safer" alternative. Achievements will be measured through surveys. |
| Tobacco Products Information Regulations (Renewal of Tobacco Product Labelling Regulations) |
The proposed regulations are to increase awareness of tobacco-related hazards through mandating of new health-related information on tobacco product labels. Awareness will be measured through surveys. |
| Warnings on Advertisements Regulations | The proposed regulations are to ensure that all Canadians, particularly youth and adult non-smokers, are provided with appropriate information when they see or read tobacco advertisements, which would include messages about the health hazards associated with tobacco use. |
| Regulations Amending the Tobacco Reporting Regulations | The proposed amendments to the Tobacco Reporting Regulations are to provide clarification and update the applicable requirements of the regulations, including prescribed laboratory methods, correction of minor grammatical errors, redundancies and ambiguities in regulation and discrepancies between the English and French versions. |
| Narcotic Control Regulations (Inclusion of tramadol in Schedule I to the Controlled Drugs and Substances Act and in the Schedule of the Narcotic Control Regulations) |
Tramadol would be regulated in the same way as other opioid analgesics, thereby decreasing the potential for it to be misused while also ensuring availability to health professionals for use in treating their patients. |
| New Classes of Practitioners Regulations | The proposed regulation is to increase flexibility in the health care system by allowing midwives, nurse practitioners and doctors of podiatric medicine to conduct certain activities, such as prescribing, with controlled substances in the treatment of their patients, when authorized to do so in their scopes of practice, as determined by individual provincial/ territorial Ministries of Health. Currently, the only health professionals who are authorized to prescribe controlled substances under the Controlled Drugs and Substances Act are doctors, dentists and doctors of veterinary medicine. |
| Program Activity: Consumer Products | |
| Cribs and Cradles Regulations (Amending Cribs, Cradles and Bassinets Regulations) |
The proposed amendment is to reduce risks of injury to children posed by cribs, cradles and bassinets. |
| Corded Window Coverings Regulations | The proposed regulation is to reduce risks to children's health (strangulation) posed by cords on window covering products such as mini-blinds and curtains. |
| Regulations on Consumer Products Containing Lead (Group 1): products intended to be or likely to be placed in the mouth | The proposed regulations are to reduce health risks to children related to lead exposure. Examples include toys intended for children under 3 years of age and mouthpieces used in sports equipment and musical instruments. Performance will be based on the removal of such hazardous products from the Canadian marketplace and the level of public awareness. |
| Regulations on Consumer Products Containing Lead (Group 2): products intended for use by a child or by an adult caring for a child | The proposed regulations are to reduce health risks to children related to lead exposure. Examples include play equipment, strollers and children's clothing and accessories. Performance will be based on the removal of such hazardous products from the Canadian marketplace and the level of public awareness. |
| Prohibition of di(2-ethyhexyl) phthalate (DEHP) (Prohibition of di(2-ethylhexyl) phthalate (DEHP) from products used by children under 3 years) |
The proposed regulations are to reduce health risks to young children from exposure to di(2-ethylhexyl) phthalate in products made of soft vinyl plastics. These types of products are likely to be mouthed by children under 3 years of age. |
| Program Activity: Pesticide Regulation | |
| Pest Control Products Regulations (The amendment to the Pest Control Products Regulations is to reflect the Grower Requested Own-use Import Program under the Pest Control Products Act) |
A grower-requested own use import program in place for price discipline for pesticides available in Canada. |
| Pest Control Products Regulations Regulations pertaining to Material Safety Data Sheets for pesticides under the Pest Control Products Act |
Requirements in place for registrants to provide pesticide Material Safety Data Sheets to workplaces. |
Services Received Without Charge
| (millions of dollars) | 2008-09 |
|---|---|
| Accommodation provided by Public Works and Government Services Canada (PWGSC) | 38.4 |
| Contributions covering employers' share of employees' insurance premiums and expenditures paid by Treasury Board of Canada Secretariat (excluding revolving funds) | 50.7 |
| Worker's compensation coverage provided by Human Resources and Skills Development Canada | 0.6 |
| Salary and associated expenditures of legal services provided by the Department of Justice Canada | 3.6 |
| Total services received without charge | 93.3 |
Sources of Respendable and Non-respendable Revenue
| Program Activity (millions of dollars) |
Forecast Revenue 2007-08 |
Planned Revenue 2008-09 |
Planned Revenue 2009-10 |
Planned Revenue 2010-11 |
|---|---|---|---|---|
| Health Products | 39.9 | 39.9 | 39.9 | 39.9 |
| Food and Nutrition | 1.3 | 1.3 | 1.3 | 1.3 |
| Sustainable Environmental Health | 1.4 | 1.4 | 1.4 | 1.4 |
| Consumer Products | 0.5 | 0.5 | 0.5 | 0.5 |
| Workplace Health | 13.8 | 14.1 | 14.1 | 14.1 |
| Pesticide Regulation | 7.0 | 7.0 | 7.0 | 7.0 |
| First Nations and Inuit Health Programming and Services | 5.4 | 5.5 | 5.5 | 5.5 |
| Total Respendable Revenue | 69.3 | 69.6 | 69.6 | 69.6 |
| Program Activity (millions of dollars) |
Forecast Revenue 2007-08 |
Planned Revenue 2008-09 |
Planned Revenue 2009-10 |
Planned Revenue 2010-11 |
|---|---|---|---|---|
| Health Products | 3.1 | 3.1 | 3.1 | 3.1 |
| Food and Nutrition | 0.8 | 0.8 | 0.8 | 0.8 |
| Sustainable Environmental Health | 0.1 | 0.1 | 0.1 | 0.1 |
| Consumer Products | 0.1 | 0.1 | 0.1 | 0.1 |
| Workplace Health | 1.5 | 1.5 | 1.5 | 1.5 |
| Pesticide Regulation | 1.0 | 1.0 | 1.0 | 1.0 |
| First Nations and Inuit Health Programming and Services | 2.3 | 2.3 | 2.3 | 2.3 |
| Total Non-respendable Revenue | 8.9 | 8.9 | 8.9 | 8.9 |
| Total Respendable and Non-respendable Revenue | 78.2 | 78.5 | 78.5 | 78.5 |
Status Report on Major Crown Projects
Description
Health Information and Claims Processing Services (HICPS) Major Crown Project.
HICPS is the key delivery mechanism for the payment of pharmacy, medical supplies and equipment, and dental benefits under Health Canada's Non-Insured Health Benefits (NIHB) Program.
The HICPS Project was established to conduct a competitive procurement to replace the existing HICPS contract, to manage the implementation of the new service contract and ensure a smooth transition from the current incumbent to the new contractor.
Project Phase
Project Implementation: The HICPS Major Crown Project entered the project implementation phase with the December 4, 2007 award of the HICPS contract to ESI Canada.
HICPS supports the delivery of much-needed health benefits for over 790,000 eligible First Nations and Inuit clients. The HICPS Project Implementation phase will terminate in December 2009 with a project evaluation. The new Health Information and Claims Processing Services will be put into production, taking over service provision on December 1, 2009.
| Lead Department or Agency: | Health Canada |
| Contracting Authority: | Public Works and Government Services Canada |
| Participating Departments and Agencies: | Indian and Northern Affairs Canada |
| Prime Contractor: | ESI Canada, Mississauga, Ontario, Canada |
| Major Subcontractors: | Resolve Corporation, Toronto, Ontario, Canada |
| Major Milestones | Date |
|---|---|
| Initial meetings with Contractor, coordination of implementation phase project plan | Contract Award (December 4, 2007 through January 2008) |
| Business Requirements Gathering and Design | February 2008 to August 2008 |
| HICPS Development | September 2008 to April 2009 |
| HICPS Testing and Acceptance | May to September 2009 |
| Documentation, Simulations, Validation, Data Conversion and Training | September 2009 to November 2009 |
| HICPS Implementation (ESI Canada officially takes over real-time service provision) | December 1, 2009 |
| Evaluation of the HICPS Project and lessons learned | December 2010 to March 2011 |
Progress Report and Explanations of Variances
The definition phase of the HICPS project (including the request for proposals process through bid evaluation and ultimately contract award) was concluded on budget and the project implementation phase is now underway as of December 4, 2007.
Industrial Benefits
The Industrial Regional Benefits (IRB) model was modified to focus on benefiting the Aboriginal economic community, rather than a specific industry or region of Canada, resulting in an Aboriginal benefit requirement (ABR) which is unique to the HICPS Project.
The development of the ABR approach for the HICPS Project was informed by industry feedback through two Requests for Information (RFI) consultation processes, and approved by Treasury Board. As HICPS' Prime Contractor, ESI Canada will be required to ensure a mandatory and substantial Aboriginal benefits requirement representing direct or indirect benefits to Aboriginal businesses or individuals.
User Fees
| Name of User Fee | Fee Type | Fee-setting Authority | Reason for Planned Introduction of or Amendment to Fee | Effective Date of Planned Change | Consultation and Review Process Planned |
|---|---|---|---|---|---|
| Authority to Sell Drugs Fees - Amended | Regulatory Service (R) | Financial Administration Act (FAA) |
Fees are being amended because:
|
2008-09 | Building on consultations, the Independent Advisory Panels and the Parliamentary Review of the Fee Proposals conducted in 2007-08, publication of proposed fees is planned in Canada Gazette in 2008-09. |
| Certificates of Pharmaceutical Product (Drug Export) Fees - Amended | Other Goods and Services (O) | Ministerial authority to enter into contract | |||
| Drug Establishment Licensing Fees - Amended | R | FAA | |||
| Drug Master File Fees - Amended | O | Ministerial authority to enter into contract | |||
| Drug Submission Evaluation Fees - Amended | R | FAA | |||
| Medical Device Licence Application Fees - Amended | R | FAA | |||
| Fees for Right to Sell a Licensed Medical Device - Amended | R | FAA | |||
| Medical Device Establishment Licensing Fees - Amended | R | FAA | |||
| Veterinary Drug Evaluation Fees - Amended | R | FAA |
Fees are being reviewed because:
|
2010 | Fees related to veterinary drug product activities will be undergoing revision, and existing service standards may also be revisited, but no specific proposals or time lines are yet targeted for stakeholders. Consultations with stakeholders will take place prior to implementation. |
| Natural Health Product Site Licensing Fee - New | R | FAA | Currently charging fees for similar activities for other product lines; will support program delivery; provides direct benefit to licence holder. | 2008-09 | Building on consultations conducted in late 2007-08, an official fee proposal will be published in early 2008-09, and Independent Advisory Panels will be arranged as required before presenting the proposal to Parliament. |
| Active Pharmaceutical Ingredient Establishment Licensing Fees - New | R | FAA | |||
| Good Clinical Practice Clinical Trial Site Registration Fee - New | R | FAA |
Additional information is available on the Health Products and Food Branch Cost Recovery Initiative website (http://www.hc-sc.gc.ca/dhp-mps/finance/costs-couts/index_e.html)